
The strong chemical bonds in biomass, or plant matter, can be broken by electricity and water thanks to the researchers’ newly developed chemical method.
These chemical tools can access plant-based renewable energy, which might reduce our reliance on fossil fuels.
There has never been a more pressing need for scientists to discover paths to products and fuels that are truly renewable given the growing prices of energy and the quickly emerging impacts of burning fossil fuels on the global climate.
“We use 20 million barrels of oil a day in the U.S.; that’s about a fifth of the world’s usage,” said Ned Jackson, a professor of organic chemistry in the College of Natural Science at Michigan State University. “All our liquid fuels and nearly all of our manufactured materials, from gasoline and gallon jugs to countertops and clothes, start with petroleum — crude oil.”
It is vital to provide the means to switch from fossil fuels to renewable sources of carbon for all of these aspects of everyday living. According to even the most optimistic estimates, however, Jackson said, “What we could harvest annually from biomass in the U.S. only has about two-thirds as much carbon in it as the crude oil that the nation uses.”
Jackson and Yuting Zhao, a former graduate student who is now a postdoctoral researcher at the University of Illinois, have discovered a chemical process that uses electricity and water to break the strong chemical bonds in biomass or plant matter. This “electrocatalytic” approach could be applied to lignin, a carbon-rich biomass component that is often wasted or burnt as a byproduct of paper production. This new technique may potentially be used to destroy environmental pollutants.
The study was recently published in the journal Nature Communications.
A global goal is to tap into both the carbon and the energy stored in biomass to enable it to replace petroleum. But new, efficient methods are needed to break this complex, tough, low-energy material down into the building blocks for fuels and products. Specifically, tools are needed to disconnect the strong chemical bonds that bind it together, while retaining — and even enhancing — as much of the carbon and energy content as possible.
“One of the things that drives us is the idea that our main use of petroleum is fuel that is burned to produce energy, adding greenhouse gases to the atmosphere,” Jackson said. “The new science is a step toward extracting useful carbon compounds to displace some fraction of the fossil petroleum that we use today.”
Reference: “Skeletal Ni electrode-catalyzed C-O cleavage of diaryl ethers entails direct elimination via benzyne intermediates” by Yuting Zhou, Grace E. Klinger, Eric L. Hegg, Christopher M. Saffron, and James E. Jackson, 19 April 2022, Nature Communications.
DOI: 10.1038/s41467-022-29555-3
This study was partly funded by the Great Lakes Bioenergy Research Center (GLBRC).


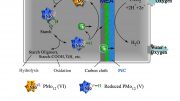





“What we could harvest annually from biomass in the U.S. only has about two-thirds as much carbon in it as the crude oil that the nation uses.”
Which means that it has a lower energy density. And, in turn, a larger volume of material must be harvested and processed to obtain the same amount of energy available from coal or crude oil. Furthermore, that means after the lignin has been converted to a simple carbon-poor chemical, it will have to be polymerized to create denser, more complex molecules. That will take energy.
Once again, these chemists are only looking at theoretical approaches to produce energy with less CO2. They need to look at the whole picture of energy inputs and outputs, waste-stream cycle, and environmental impacts. This is another example of specialists looking at a narrow part of what is considered a problem. Might the lignin be of greater value as a soil conditioner, or the basis for making compost to enhance the fertility of soils? There needs to be guidance from the NSF or DOE of how these kinds of proposals fit into the big picture.
Good stuff. Science rules!🅾️🅱️