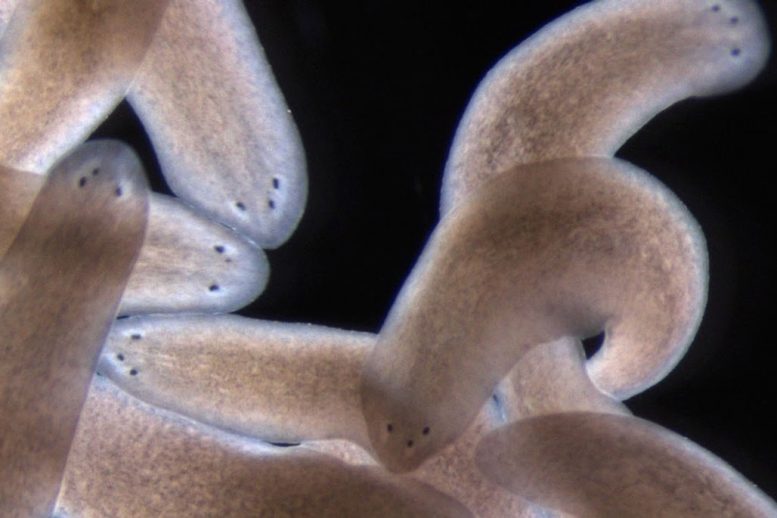
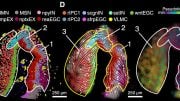
Three-eye planarians created by removing one of the flatworm’s eyes during regeneration of its head. Image: Kutay Deniz Atabay/Whitehead Institute
Researchers at Whitehead Institute have uncovered a framework for regeneration that may explain and predict how stem cells in adult, regenerating tissue determine where to form replacement structures.
In a paper that appeared online March 15 in the journal Science, the researchers describe a model for planarian (flatworm) eye regeneration that is governed by three principles acting in concert, which inform how progenitor cells behave in regeneration. The model invokes positional cues that create a scalable map; self-organization that attracts progenitors to existing structures; and progenitor cells that originate in a diffuse spatial zone, rather than a precise location, allowing flexibility in their path. These principles appear to dictate how progenitor cells decide where to go during regeneration to recreate form and function, and they bring us closer to a systems-level understanding of the process.
From previous work, the researchers knew that stem cells are likely reading out instructions from neighboring tissues to guide their path, and it became clear that the process faces some serious challenges in regeneration. “We realized that positional information has to move; it needs to change during regeneration in order to specify the new missing parts to be regenerated. This revised information can then guide progenitor cells that are choosing to make new structures to differentiate into the correct anatomy at the correct locations,” says the paper’s senior author Peter Reddien, a Whitehead Institute researcher, an MIT professor of biology, and a Howard Hughes Medical Institute (HHMI) investigator. “There is a puzzle that emerges, however. Since positional information shifts after injury during regeneration, there is a mismatch between the positional information pattern and the remaining anatomy pattern. Realizing this mismatch exists was a trigger for our study. We wanted to understand how stem cells making particular tissues decide where to go and differentiate. Is it based on anatomy, or is it based on positional information? And when those two things are not aligned, how do they decide?”
Reddien and his lab have spent over a decade unraveling the mysteries of regeneration using a small flatworm, called the planarian. If a planarian’s head is amputated, or its side is removed, each piece will regenerate an entire animal. In order to understand how progenitors decide where to go in the noisy environment of animal regeneration, the researchers used the planarian eye, a visible organ that is small enough to be removed without serious injury and has the added advantage of having defined progenitor cell molecular markers.
The researchers devised a simple experiment to resolve the question of how the progenitors decide where to go: amputate the animal’s head, then after three days remove one of the eyes from the head piece. What they found was that progenitor cells would nucleate a new eye in a position anterior (closer to the tip) to the remaining eye, rather than in the “correct” position specified by the anatomy, symmetrical with the current eye. However, if the same experiment is done, except with one of the eyes removed from the head piece earlier — the same day as the amputation, rather than three days later — there is a different result: The new eye is nucleated at a position symmetrical to the remaining eye, at the “correct” position according to the anatomy, suggesting that when the choices are conflicting, anatomical self-organizing dynamics win.
These simple rules guide the system to successful regeneration and also yield surprising outcomes when the system is pushed to its limits, producing alternative stable anatomical states with three-, four-, and five-eyed animals. Resecting both the side of an animal and amputating the head can place progenitors far enough from existing, self-organizing attractors that they miss them, allowing them to nucleate a new attractor — a third eye — in the head fragments. “If the migrating progenitors are too close to the attractor that is the existing eye, it will suck them in, just like a black hole; if they are far enough, they can escape,” says Kutay Deniz Atabay, first author on the paper and a graduate student in the Reddien lab.
When the researchers did the same surgery on a three-eyed animal, the progenitors miss the attraction of existing eyes and form a five-eyed animal. In each of these cases, all of the eyes are functionally integrated into the brain and exhibit the same light-avoidance response of the planarian eye. The researchers also observed that these alternative anatomical states endure, revealing rules for maintenance of existing and alternative anatomical structures. “This recipe helps provide an explanation for a fundamental problem of animal regeneration, which is how migratory progenitors make choices that lead the system to regenerate and maintain organs,” Atabay says.
“These studies contribute to a proposed framework for regeneration in which competing forces, self-organization, and extrinsic cues, are the guideposts impacting the choice of progenitor targeting in regeneration; and those two forces together determine the outcome,” Reddien says.
This work was supported by the National Institutes of Health (NIH) and the HHMI. Additional funding was provided by the MIT Presidential Fellowship Program and a National Defense Science and Engineering Graduate Fellowship.
Reference: “Self-organization and progenitor targeting generate stable patterns in planarian regeneration” by Kutay Deniz Atabay, Samuel A. Locascio, Thom De Hoog and Peter W. Reddien, 15 March 2018, Science.
DOI: 10.1126/science.aap8179




Be the first to comment on "Whitehead Institute Biologists Uncover a Blueprint for Regeneration"