An artist’s rendering of a new electrocatalytic process that can convert carbon dioxide and water into ethanol. Credit: Argonne National Laboratory
A research team led by scientists from Argonne National Laboratory, the University of Chicago’s Pritzker School of Molecular Engineering and Northern Illinois University has discovered a new electrocatalyst that can consistently convert carbon dioxide and water into ethanol with very high energy efficiency and low cost.
Because carbon dioxide is a stable molecule, transforming it into a different molecule is normally energy intensive and costly.
But the new process can electrochemically convert the carbon dioxide emitted from industrial processes—such as fossil fuel or alcohol fermentation plants—into a valuable commodity at a reasonable cost. Ethanol is an ingredient in nearly all U.S. gasoline and is widely used as an intermediate product in the chemical, pharmaceutical, and cosmetics industries.
“The process resulting from our catalyst would contribute to the circular carbon economy, which entails the reuse of carbon dioxide,” said Di-Jia Liu, a senior chemist in Argonne’s Chemical Sciences and Engineering division and a UChicago CASE scientist in the Pritzker School of Molecular Engineering at the University of Chicago.
The findings were published recently in the journal Nature Energy.
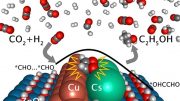
The team’s catalyst consists of atomically dispersed copper on a carbon-powder support. By an electrochemical reaction, this catalyst breaks down carbon dioxide and water molecules and selectively reassembles the broken molecules into ethanol under an external electric field.
“The process resulting from our catalyst would contribute to the circular carbon economy, which entails the reuse of carbon dioxide.” — Di-Jia Liu, senior chemist in Argonne’s Chemical Sciences and Engineering division and a UChicago CASE scientist
Previous attempts at this process often aren’t very good at fully converting the carbon dioxide. But the electrocatalytic selectivity, or “Faradaic efficiency,” of the new method is over 90%—much higher than any other reported process. What’s more, the catalyst operates stably over extended operation at low voltage.
“We could couple the electrochemical process of carbon dioxide-to-ethanol conversion using our catalyst to the electric grid and take advantage of the low-cost electricity available from renewable sources like solar and wind during off-peak hours,” Liu said.
Because the process runs at low temperatures and pressure, it can start and stop rapidly in response to the intermittent supply of renewable electricity.
“We have prepared several new catalysts using this approach and found that they are all highly efficient in converting carbon dioxide to other hydrocarbons,” said Liu. “We plan to continue this research in collaboration with industry to advance this promising technology.”
The team’s research benefited from the Advanced Photon Source and Center for Nanoscale Materials at Argonne, as well as its Laboratory Computing Resource Center.
“Thanks to the high photon flux of the X-ray beams at the APS, we have captured the structural changes of the catalyst during the electrochemical reaction,’’ said Tao Li, an assistant professor in the Department of Chemistry and Biochemistry at Northern Illinois University and an assistant scientist in Argonne’s X-ray Science division
“With this research, we’ve discovered a new catalytic mechanism for converting carbon dioxide and water into ethanol,” said co-author Tao Xu, a professor in physical chemistry and nanotechnology from Northern Illinois University. “The mechanism should also provide a foundation for the development of highly efficient electrocatalysts for carbon dioxide conversion to a vast array of value-added chemicals.”
For more on this research, read New Electrocatalyst Turns Carbon Dioxide Into Liquid Fuel.
Reference: “Highly selective electrocatalytic CO2 reduction to ethanol by metallic clusters dynamically formed from atomically dispersed copper” by Haiping Xu, Dominic Rebollar, Haiying He, Lina Chong, Yuzi Liu, Cong Liu, Cheng-Jun Sun, Tao Li, John V. Muntean, Randall E. Winans, Di-Jia Liu and Tao Xu, 27 July 2020, Nature Energy.
DOI: 10.1038/s41560-020-0666-x
In addition to Liu and Xu, authors include Haiping Xu, Dominic Rebollar, Haiying He, Lina Chong, Yuzi Liu, Cong Liu, Cheng-Jun Sun, Tao Li, John V. Muntean and Randall E. Winans.
Funding: Argonne’s Laboratory Directed Research and Development (U.S. Department of Energy Office of Science).







Drinking our way to CO2 reduction? 🙂
The title of the article includes the words, “Highly Selective.” That is quite appropriate because the researchers seem to be looking at only a small portion of the system necessary to make fuels.
The advantage of fossil fuels is that solar energy is ‘baked’ into them over millions of years. They have what is called “high energy density.” To convert CO2 into a hydrocarbon, a tremendous amount of energy is necessary to build the components of the system. CO2-capture retrofitting of sources, delivery of clean water (including pumping), the manufacture and installation of so-called ‘renewable energy sources, and the manufacture of the catalyst. All the energy used to put the system into action needs to be amortized to determine the cost of energy input, and determine if there is a net gain or loss, and how that compares to alternatives. Energy is necessary to do work. If the CO2 conversion is a net loss of energy to do the same thing that was previously done with fossil fuels, then it becomes a situation like a family borrowing on their credit cards to make ends meet, and never paying off the debt. It is an unsustainable financial plan.
Have the researchers done a beginning-to-end analysis of the energy requirements and determined how much acreage will be required for photocells? If it is more than the area of the roof of the plant, then it probably results in a loss of potential agriculture, infrastructure, or wild lands. Just because something CAN be done, it doesn’t mean it SHOULD be done!
One would think that the designers would see that the design was, as you said, borrowing on your credit card to pay off your debts, in which case they would not continue the design.
But the scientists did continue with the design of adding ethanol to gasoline and requiring car makers to make some quantity of cars as “Flex Fuel” ready to use ethanol as fuel. I won’t get into the details, but it has recently come to light that the scientists left out important factors that now show the use of ethanol in gasoline is exactly the wasteful boondoggle, not to mention that it causes other problems like making the prices of other foods go higher. SMH…
Every thing starts at a minimum of efficiency, Ford Model A or Bi-plane and it progresses to the most efficient. This is a great start and there will be a Tesla, who will do something that has not even thought possible.
In 1972 my organic chem professor had worked out the the bond energies and mechanisms for polymerization of CO2, he was trying to find the catalyst. Maybe
Back in the 1970s we didn’t have the scientific tools we now have. Seems so simple that the catalyst could be just copper on carbon. But there’s a lot more to it than what it’s made from.
This seems like pretty cool technology.
What cost electricity is needed to commercial cost 95mole% ethanol? This needs to include voncentrating the ethanol and deionizing the water to protect the catalyst?
I never would’ve believed it and electric powered drag racing automobile!
https://youtu.be/LE0BhrciMuk
What do the materials cost? Is a retrofitted closed loop system for automobiles a possibilty?
Not exactly impressive to make a device or machine that creates something that needs the machine… And requires high amounts of energy.
“The process resulting from our catalyst would contribute to the circular carbon economy, which entails the reuse of carbon dioxide.” There’s the problem. We can never get off the merry-go-round otherwise all the CO2 sequestered will be back in the atmosphere on the next cycle. That’s the risk of biofuels (which are 90% fossil fuels).
Please, Please check out to see if this was already discussed and discovered by a black scientist. We don’t want to hear about it,like electricity was really discovered by Thomas Edison’s black slave shop cleaning boy….
ok good for smug la for free fuel for ev hybrid cars and for the open road not needing a recharge/refill for minimum 5000 miles before quick 15 second recharge to 95 percent on ev battery backs. makes air cooling on laptops and desktops convert dirty hot air into free battery juice like with water cooling systems.
Interesting but this isn’t new and they aren’t pioneers in this..serveral months ago I read an article of a university group in Australia doing exactly what is described here
1 volt DC is fine for water decomposition electrolysis.
I think CO2 might take a bit more than a volt . Looks like a sweet job for low voltage PV.