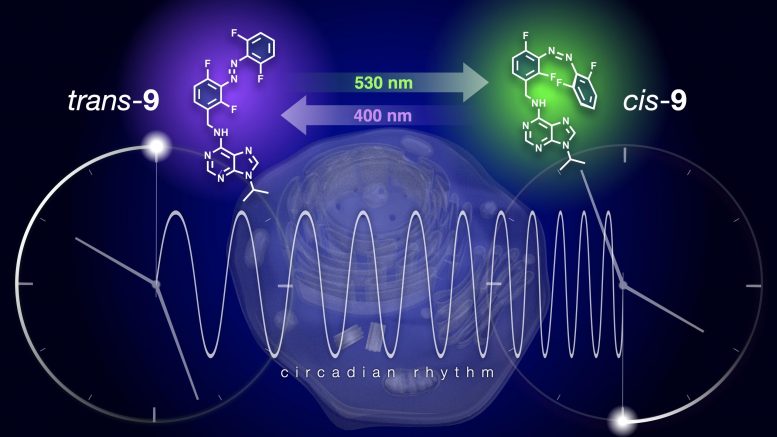
Reversible modulation of the circadian clock using chronophotopharmacology. Using light to interconvert two isomers of a photo-responsive small molecule, it is possible to pace cellular time. While irradiation with violet light extends the normal 24-hour clock to 28-hour, green light switches off this effect and brings the clock back to normal. Credit: Issey Takahashi
The biological clock is present in almost all cells of an organism. As more and more evidence emerges that clocks in certain organs could be out of sync, there is a need to investigate and reset these clocks locally. Scientists from the Netherlands and Japan introduced a light-controlled on/off switch to a kinase inhibitor, which affects clock function. This gives them control of the biological clock in cultured cells and explanted tissue. They publish their results today (May 26, 2021) in Nature Communications.
Life on Earth has evolved under a 24-hour cycle; of light and dark, hot and cold. “As a result, our cells are synchronized to these 24-hour oscillations,” says Wiktor Szymanski, Professor of Radiological Chemistry at the University Medical Center Groningen. Our circadian clock is regulated by a central controller in the suprachiasmatic nucleus, a region in the brain directly above the optic nerve, but all our cells contain a clock of their own. These clocks consist of an oscillation in the production and breakdown of certain proteins.
Light switch
“It is becoming increasingly clear that these clocks can be disrupted in organs or tissues, which may lead to disease,” adds first author Dušan Kolarski, a PhD student from the group led by Ben Feringa, Professor of Organic Chemistry. And, of course, we all know about jet lag, which is caused by travel across time zones, or problems that are caused by the switch to or from daylight saving time. “We know very little about how our cells coordinate these oscillations, or how it affects the body if, for example, one kidney is out of phase with the rest of the body,” he adds.

This picture shows first author Dusan Kolarski (back row, left) with the team from the Institute of Transformative Bio-Molecules at Nagoya University, Japan, including co-authors Tsuyoshi Hirota (back row, middle), Akiko Sugiyama (front, second from left) and Yoshiko Nagai (front, fourth from left). Credit: Institute of Transformative Bio-Molecules, Nagoya University
To study these effects, it would be useful to have a drug that affects the clocks and that can be activated locally. The latter is something that the groups of Szymanski and Feringa have done before. They created several compounds, such as antibiotics or anticancer drugs, that could be switched on and off with light. Previously, circadian biologist Tsuyoshi Hirota, associate professor at the Institute of Transformative Bio-Molecules at Nagoya University, Japan, developed a kinase inhibitor, longdaysin, which slows down the circadian clock to a cycle that lasts up to 48 hours. Kolarski fitted this longdaysin with a light switch that allowed him to activate or deactivate the compound with violet and green light, respectively.
Time zone
Developing this adaptation took Kolarski several years, but the result was well worth the effort. “It was a real scientific ‘Tour de Force’ and a beautiful example of interdisciplinary cooperation,” adds Feringa. Together with their Japanese colleagues at Nagoya University, the scientists from the University of Groningen showed how the cycle of cultured cells was extended from 24 to 28 hours by treatment with the longdaysin derivative. Deactivation with green light brought the cycle back to just over 25 hours and subsequent reactivation with violet light returned it to 28 hours.
“We also used it in tissue slices from the mouse suprachiasmatic nucleus,” says Kolarski. “The oscillations slowed to a 26-hour cycle after treatment for several days with the longdaysin derivative and returned to a 24-hour cycle after deactivation with green light.”
“This reversible regulation will provide a new approach to analyzing how the clock in each cell is organized at the tissue level to gain a deeper understanding of the complex circadian clock system,” Hirota adds.
The scientists also adjusted the phase of the cycles in cultured cells: a three-day activation of the longdaysin derivative followed by deactivation caused a shift in the 24-hour cycle by up to six hours. This is as if the cells were synchronized with a different time zone. The experiments are a proof of principle and will allow scientists to study the circadian clock in much more detail. A next step would be to use longdaysin in animals. Kolarski: “The original longdaysin, without the switch, has been used before in zebrafish. We would very much like to test it in mice. The aim is not to fix jet lag but to study the effect of longdaysin on physiology.”
Organs
A light-activated drug such as longdaysin will probably only be used to treat serious conditions. “We can actually reach quite a few organs with light, for example with an endoscope. The gastrointestinal tract and the respiratory system are easily reached, while other tissues may require small incisions to insert optic fibers,” comments Szymanski. There are also several emerging options to generate light inside organs or tissues, through techniques such as bioluminescence or sonoluminescence. Although these levels of light are still several orders of magnitude below what we need to flick a switch. We will work hard to increase sensitivity in the coming years, emphasize both Szymanski and Feringa. Kolarski adds: “We have now opened a new field of study. Eventually, all this will allow us to locally disrupt or repair the circadian oscillations.”
Simple Science Summary
The cells in our body follow a 24-hour cycle, the circadian clock. Disruptions of this cycle, for example by working night shifts, can cause disease. In recent years, it has become clear that the clock can be disrupted in individual organs or tissues. To study and potentially cure problems with the clocks inside our cells, Dutch and Japanese scientists created a compound that will elongate the 24-hour cycle and that can be activated or deactivated using light. They showed that it is possible to change the 24-hour cycle in cells or tissues to a 28-hour cycle by activating the compound. After deactivation, the cells and tissues returned to a near-normal cycle. The compound can be used to investigate the clocks inside our cells and may eventually be used to treat diseases that are caused by a disrupted clock.
Reference: “Reversible modulation of circadian time with chronophotopharmacology” by Reference: Dušan Kolarski, Carla Miró Vinyals, Akiko Sugiyama, Ashutosh Srivastava, Daisuke Ono, Yoshiko Nagai, Mui Iida, Kenichiro Itami, Florence Tama, Wiktor Szymanski, Tsuyoshi Hirota and Ben L. Feringa, 26 May 2021, Nature Communications.
DOI: 10.1038/s41467-021-23301-x







Treating the human body with light to affect circadian rhythms as well as other aspects of physiology was developed by Dr. Dinshah Ghadiali. No need to wait years for research, as anyone can use this method in their own home. Check out the book “LetThere Be Light” by Darius Dinshah.