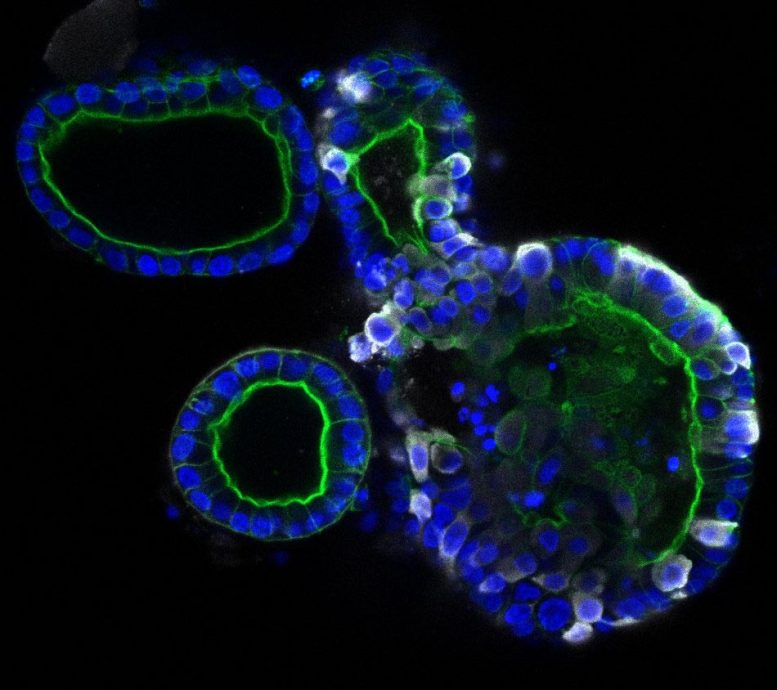
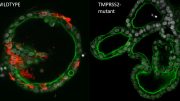
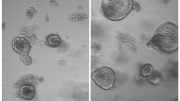
Intestinal organoids, the right one infected with coronavirus SARS-CoV-2. The coronavirus is colored white, the organoids themselves are colored blue and green. Credit: Joep Beumer, copyright Hubrecht Institue
Researchers from the Hubrecht Institute in Utrecht, Erasmus MC University Medical Center Rotterdam, and Maastricht University in the Netherlands have found that the coronavirus SARS-CoV-2, which causes COVID-19, can infect cells of the intestine and multiply there. Using state-of-the-art cell culture models of the human intestine, the researchers have successfully propagated the virus in vitro, and monitored the response of the cells to the virus, providing a new cell culture model for the study of COVID-19. These findings could explain the observation that approximately one third of COVID-19 patients experience gastrointestinal symptoms such as diarrhea, and the fact that the virus often can be detected in stool samples. The results of this study were published in the scientific journal Science on the 1st of May 2020.
Patients with COVID-19 show a variety of symptoms associated with respiratory organs – such as coughing, sneezing, shortness of breath, and fever – and the disease is transmitted via tiny droplets that are spread mainly through coughing and sneezing. One third of the patients however also have gastrointestinal symptoms, such as nausea and diarrhea. In addition, the virus can be detected in human stool long after the respiratory symptoms have been resolved. This suggests that the virus can also spread via so-called “fecal-oral transmission.”
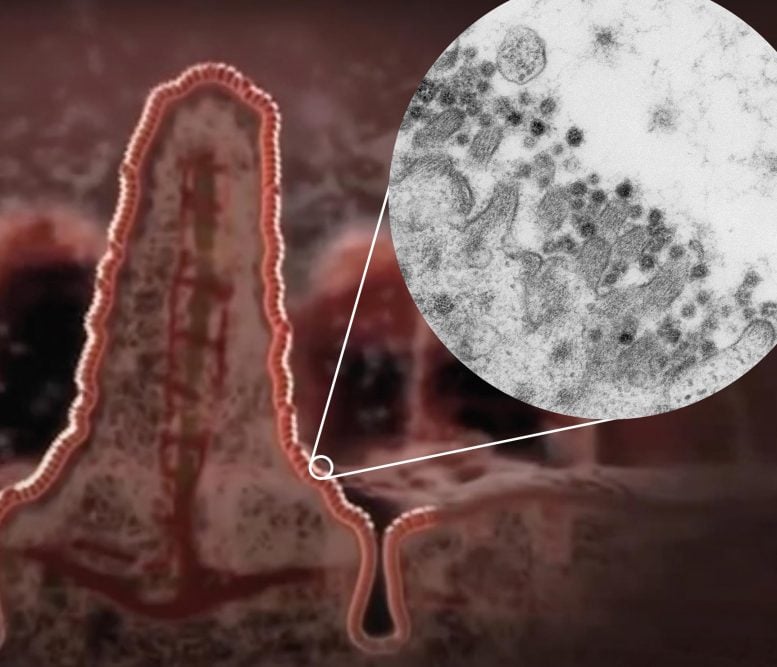
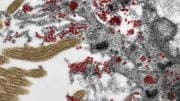
Illustration of a villus in the intestine with a zoom-in to an electron microscopy image of coronavirus SARS-CoV-2 (dark circles) at the edge of an intestinal cell. Credit: Kèvin Knoops, Raimond Ravelli and Maaike de Backer, copyright: Maastricht University
Though the respiratory and gastrointestinal organs may seem very different, there are some key similarities. A particularly interesting similarity is the presence of the ACE2 receptor, the receptor through which the COVID-19 causing SARS-CoV-2 virus can enter the cells. The inside of the intestine is loaded with ACE2 receptors. However, until now it was unknown whether intestinal cells could actually get infected and produce virus particles.
Intestinal organoids
Researchers from the Hubrecht Institute, Erasmus MC, and Maastricht University set out to determine whether the SARS-CoV-2 virus can directly infect the cells of the intestine, and if so, whether it can replicate there as well. They used human intestinal organoids: tiny versions of the human intestine that can be grown in the lab. Hans Clevers (Hubrecht Institute): “These organoids contain the cells of the human intestinal lining, making them a compelling model to investigate infection by SARS-CoV-2.”
Infection of intestinal cells
When the researchers added the virus to the organoids, they were rapidly infected. The virus enters a subset of the cells in the intestinal organoids, and the number of cells that are infected increases over time. Using electron microscopy, an advanced way to visualize different components of the cell in great detail, the researchers found virus particles inside and outside the cells of the organoids. Peter Peters (Maastricht University): “Due to the lockdown, we all studied virtual slides of the infected organoids remotely from home.”
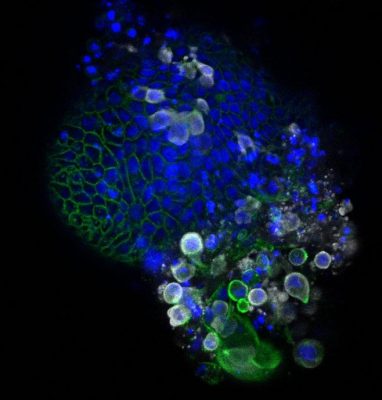
Intestinal organoid infected with coronavirus SARS-CoV-2. The coronavirus is colored white, the organoids themselves are colored blue and green. Credit: Joep Beumer, copyright: Hubrecht Institute
The researchers investigated the response of the intestinal cells to the virus with RNA sequencing, a method to study which genes are active in the cells. This revealed that so-called interferon-stimulated genes are activated. These genes are known to combat viral infection. Future work will focus on these genes more carefully, and on how they could be used to develop new treatments.
The researchers also cultured the organoids in different conditions that result in cells with higher and lower levels of the ACE2 receptor, through which SARS-CoV-2 can enter the cells. To their surprise, they found that the virus-infected cells with both high and low levels of the ACE2 receptor. Ultimately, these studies may lead to new ways to block the entry of the virus into our cells.
Implications
Bart Haagmans (Erasmus MC): “The observations made in this study provide definite proof that SARS-CoV-2 can multiply in cells of the gastrointestinal tract. However, we don’t yet know whether SARS-CoV-2, present in the intestines of COVID-19 patients, plays a significant role in transmission. Our findings indicate that we should look into this possibility more closely.” The current study is in line with other recent studies that identified gastrointestinal symptoms in a large fraction of COVID-19 patients and virus in the stool of patients free of respiratory symptoms. Special attention may be needed for those patients with gastrointestinal symptoms. More extensive testing using not only nose and throat swabs, but also rectal swabs or stool samples may thus be needed.
In the meantime, the researchers are continuing their collaboration to learn more about COVID-19. They are studying the differences between infections in the lung and the intestine by comparing lung and intestinal organoids infected with SARS-CoV-2.
Reference: “SARS-CoV-2 productively Infects Human Gut Enterocytes” by Mart M. Lamers, Joep Beumer, Jelte van der Vaart, Kèvin Knoops, Jens Puschhof, Tim I. Breugem, Raimond B.G. Ravelli, J. Paul van Schayck, Anna Z. Mykytyn, Hans Q. Duimel, Elly van Donselaar, Samra Riesebosch, Helma J.H. Kuijpers, Debby Schipper, Willine J. van de Wetering, Miranda de Graaf, Marion Koopmans, Edwin Cuppen, Peter J. Peters, Bart L. Haagmans and Hans Clevers, 1 May 2020, Science.
DOI: 10.1126/science.abc1669
This study was a collaboration between the Hubrecht Institute in Utrecht, the Erasmus MC University Medical Center Rotterdam, Maastricht University, the UMC Utrecht, and Single Cell Discoveries in the Netherlands. The microscopy data are publicly available via the Image Data Resource (idr0083 – with help from the University of Dundee and the European Bioinformatics Institute) and the genomic data are publicly available via the Gene Expression Omnibus (GSE149312), to ensure efficient sharing of data related to COVID-19 between researchers all across the world.
Hans Clevers is principal investigator at the Hubrecht Institute and the Princess Máxima Center for Pediatric Oncology, professor of Molecular Genetics at the UMC Utrecht and Utrecht University, and Oncode Investigator.
Bart Haagmans is a principal investigator at the Viroscience department at the Erasmus MC University Medical Center Rotterdam.
Peter Peters is director and principal investigator at the Maastricht Multimodal Molecular Imaging Institute (M4i) and professor of Nano Biology at the Maastricht University and Maastricht University Medical Center.



Do this intestinal organoids contain gut bacteria?
Yes it does contain bacteria, but that’s irrelevant to the virus attacking epithelium cells of the gut lining. I haven’t seen any indication microbiome alters the virus binding.
Could this be how to “vaccinate” (not a real vaccine) against COVID-19? If an infection starts in the gut (either ingested or introduced through the other end) that gives your immune system a head start on recognizing and fighting it before it ever makes it to the respiratory tract. It may not be pleasant, but unlike having it in your lungs it doesn’t sound like this kills people, so maybe it’s a way to give people protective immunity that will last a few years until we come up with another less-unpleasant vaccination method?
Turn healthy people into COVID spreaders.. Not a great idea, also people would still die from the virus that way, just perhaps slightly fewer would die than otherwise..
I’m absolutely sure I had this virus in my intestines for sure ! Long story short! Went grocery shopping on March 12 Thursday! A lady in line was talking to me I actually felt her breath go down my throat. Not a sneeze or a cough just her breath! 2 weeks to the day I suddenly had the worst diarrhea of my life that lasted 10 days. Couldn’t sleep eat drink I paced the floors all night then after the 10 days still had some diarrhea but stomach was gurgling out of control I was deathly sick didn’t want to go to a doctor because we were in lock down. It lasted a total of just about 5 weeks ! After reading this I really think I maybe had the virus !! Any thoughts ??
The activation of interferon stimulated genes in the infected cells in the absence of typical interferon-producing cells (is this true in your organoid system?) provides evidence of these cells as additional sources for the extreme cytokine release seen in many COVID-19 patients.
Test these, forget the drugs.
The hunt for BIGPHARMA PROFIT MAKING treatments is on and pockets of governments and supposed medical authorities. I just gave my PERSONAL plan which is
All these may be taken together: zinc 30-40mg + quercitin 500-1,000mg and/or EGCG 600-700mg – these were shown as effective as quinine drugs in 2014; BHT which disturbs lipid coats of such viruses rendering them open to the immune system; mega doses of Vitamin C – I take 5,000mg a day but would take up to 20,000 or more if ill. Linus Pauling had done extensive initial research; and in a medical office since some simple equipment is used by a nurse would be photoluminescence (UV light) therapy. Deadly and quite effective on microbes. These would deal quite nicely with the manifesting illness. Oh, with an advanced medical office if they had hyperbaric oxygen chambers, I would be pleased to lie in one for 45 minutes as oxygen is deeply saturated into the body. Such levels help with other conditions but are deadly to viruses. All this is more than enough.
In Feb I came back from a business trip to Singapore. At the time it was still not so serious here in Malaysia. I had diarrhea for about 4 days after and I was wondering what happened. Then I watched a prominent Dr from Singapore on tv saying that if you have diarrhea, this could be a symptom but not many people have said this. After that I felt fine and I still wonder if I had caught the virus while I was in Singapore without realizing it.
@ George DeCarlo
You’re correct about Linus Pauling research. However, everyone should keep the following in mind, especially now with the corona scamdemic going on: there are many bogus voices around who strive to distract the public from the value of vitamin C therapy and the fact that Pauling’s VALID work with vitamin C supplementation has been “falsified” by data distortions and lies, and he as a person (a double Nobel laureate) has been slandered as some deluded idiot by the criminal medical establishment and its countless quackwatch shills, lackeys, ignoramuses, and trolls for decades and it continues today — search for the scholarly report “2 Big Lies: No Vitamin Benefits & Supplements Are Very Dangerous” by Rolf Hefti (a published author of the Orthomolecular Medicine News organization).
But you can’t discredit the facts with lies. That only exposes and discredits the liars (see citation above).
Read https://www.facebook.com/ron.mccune.3 to see what more I have to say about the coronavirus and other subjects of concern. I believe that when one gets diarrhea it may be caused by many things like what you ate. When one gets a virus and diarrhea I believe that the body is going into a defensive mode and trying to clear out the intestines of any molecules of any kind that may be harmful to you as you experience diarrhea episodes. The body clears out your intestines to get whatever is in your intestines out of you. However diarrhea only cleans out so much of your intestines at such a pace that the coronavirus can still harbor in the intestines maybe and multiply as it lives in your intestines. So what if we were to give as much of edemas as we can to flush out the intestines as much as we can. If there is a way to after send water mixed with medicines that may help kill the coronavirus through the mouth so that the intestines are flushed out several times and loaded with medicines that fight coronavirus. Then send a ultraviolet light in rectum procedure and throat procedure where they look at your lower and upper digestive system to kill the virus. Do the nose, throat, lung air waves with ultraviolet light too if possible. Don’t know if that’s possible but if it works then do it! So why don’t we try to help along the body’s strategy of cleaning out the digestive system and do what we can to kill the coronavirus in the respiratory tract then maybe the coronavirus infected person will have an easier time dealing with this coronavirus or other illnesses if it have. When you look at those that got ill in more severe ways that others you see that just about all were overweight. That meant that the coronavirus have forever to multiply and an abundance of molecules to attach it’s offsprings onto so to say. I don’t know how to say all this medical stuff but you get the picture. So completely flushing out the intestines would certainly empty out many coronavirus molecules out of a body! Then if we were to give a large amount of this recipe called Adelynn’s Miracle Beans recipe and diet plan only we would be able to also induce the body to keep it’s intestines empty as the body then starts to get its energy from the energy stored in one’s veins! However in the recipe ONLY use beans and not noodles, rice or other filler foods. You don’t want to introduce energy food at this time into the intestines. You want the body to starve itself into making your bodies vascular system to have to go into overdrive to create energy because you have successfully dried up the stored energy capabilities in the stomach and vascular system. The blood system will then have to get to work getting energy from a vein network with less fats which should cause the patient to get better because the intestines and veins will have less of the coronavirus in it! Once that happens then the coronaviruses in the vein network will have to be used and will result I believe in less coronavirus in the bloodstream. I believe that as the bloodstream pumps blood throughout your body it goes through your cleaning biological systems such as your liver. Maybe as the blood goes through these blood cleaning system the coronavirus strength in the body is lessen somehow. That seems to be so because most of the people who died or got seriously sick from the coronavirus were overweight. So the amount of the virus in the body, the weight of the body fat content and the time it sits in the blood system and digestive systems are tell tale signs of many! The fact that others that aren’t overweight are getting sick too may be caused by the coronavirus entering the nose area and easily making its way into the brain area where it causes ills of different sorts. Also others that aren’t overweight and also are young that get seriously sick seem to me to be mostly thin people. That has to say something! Maybe this virus is highjacking it’s way around the bodies many parts and somehow latched onto an area in the body that allowed the coronavirus to do harm. Perhaps also giving sick patients massive amounts of vitamins along with the Adelynn’s Miracle Beans would give their bodies more ability to get better because Adelynn’s Miracle Beans recipe has all of the 9 different amino acids to make all the proteins of many different kinds of many different human cells of all kinds! Give the body all the complete protein and vitamins in large quantities and let’s see what the human body can do to save us from the miseries we face!
I believe that when one gets diarrhea it may be caused by many things like what you ate. When one gets a virus and diarrhea I believe that the body is going into a defensive mode and trying to clear out the intestines of any molecules of any kind that may be harmful to you as you experience diarrhea episodes. The body clears out your intestines to get whatever is in your intestines out of you. However diarrhea only cleans out so much of your intestines at such a pace that the coronavirus can still harbor in the intestines maybe and multiply as it lives in your intestines. So what if we were to give as much of edemas as we can to flush out the intestines as much as we can. If there is a way to after send water mixed with medicines that may help kill the coronavirus through the mouth so that the intestines are flushed out several times and loaded with medicines that fight coronavirus. Then send a ultraviolet light in rectum procedure and throat procedure where they look at your lower and upper digestive system to kill the virus. Do the nose, throat, lung air waves with ultraviolet light too if possible. Don’t know if that’s possible but if it works then do it! So why don’t we try to help along the body’s strategy of cleaning out the digestive system and do what we can to kill the coronavirus in the respiratory tract then maybe the coronavirus infected person will have an easier time dealing with this coronavirus or other illnesses if it have. When you look at those that got ill in more severe ways that others you see that just about all were overweight. That meant that the coronavirus have forever to multiply and an abundance of molecules to attach it’s offsprings onto so to say. I don’t know how to say all this medical stuff but you get the picture. So completely flushing out the intestines would certainly empty out many coronavirus molecules out of a body! Then if we were to give a large amount of this recipe called Adelynn’s Miracle Beans recipe and diet plan only we would be able to also induce the body to keep it’s intestines empty as the body then starts to get its energy from the energy stored in one’s veins! However in the recipe ONLY use beans and not noodles, rice or other filler foods. You don’t want to introduce energy food at this time into the intestines. You want the body to starve itself into making your bodies vascular system to have to go into overdrive to create energy because you have successfully dried up the stored energy capabilities in the stomach and vascular system. The blood system will then have to get to work getting energy from a vein network with less fats which should cause the patient to get better because the intestines and veins will have less of the coronavirus in it! Once that happens then the coronaviruses in the vein network will have to be used and will result I believe in less coronavirus in the bloodstream. I believe that as the bloodstream pumps blood throughout your body it goes through your cleaning biological systems such as your liver. Maybe as the blood goes through these blood cleaning system the coronavirus strength in the body is lessen somehow. That seems to be so because most of the people who died or got seriously sick from the coronavirus were overweight. So the amount of the virus in the body, the weight of the body fat content and the time it sits in the blood system and digestive systems are tell tale signs of many! The fact that others that aren’t overweight are getting sick too may be caused by the coronavirus entering the nose area and easily making its way into the brain area where it causes ills of different sorts. Also others that aren’t overweight and also are young that get seriously sick seem to me to be mostly thin people. That has to say something! Maybe this virus is highjacking it’s way around the bodies many parts and somehow latched onto an area in the body that allowed the coronavirus to do harm. Perhaps also giving sick patients massive amounts of vitamins along with the Adelynn’s Miracle Beans would give their bodies more ability to get better because Adelynn’s Miracle Beans recipe has all of the 9 different amino acids to make all the proteins of many different kinds of many different human cells of all kinds! Give the body all the complete protein and vitamins in large quantities and let’s see what the human body can do to save us from the miseries we face!