
This huge development, in the opinion of the researchers, will eventually help to fully realize the enormous potential of hydrogen fuel cells.
The commercialization of eco-friendly fuel has been stalled for decades by the high cost of platinum, but a study indicates that the low-cost catalyst may be a feasible substitute.
For many years, researchers have been looking for a catalyst that would significantly lower the price of producing hydrogen fuel cells.
A green energy revolution could result from such a breakthrough, with laptops and trains alike using fuel that only produces water as a byproduct. Researchers may be getting closer to achieving this objective, according to recent findings from the University at Buffalo (UB).
The U.S. Department of Energy (DOE) has identified efficiency, durability, and affordability as the three main goals for fuel cell research. In a study that was recently published in Nature Energy, scientists explain how iron can be combined with nitrogen and carbon to produce a catalyst that meets all three criteria.
“This has been years in the making,” says the study’s lead author Gang Wu, Ph.D., professor of chemical and biological engineering at the UB School of Engineering and Applied Sciences. “We believe this is a significant breakthrough that will eventually help unleash the tremendous potential of hydrogen fuel cells.”
The promise of fuel cells
According to DOE, fuel cells operate similarly to batteries but don’t lose power or need recharging. They generate heat and power so long as fuel, such as hydrogen, is supplied.
Because they produce less or no emissions as compared to combustion engines, they have long intrigued scientists, environmentalists, and others. Additionally, they have a broad variety of uses, including powering buildings, power plants, automobiles, and other systems.
However, the lack of widespread commercialization of fuel cells is due, among other things, to the high cost of the expensive catalysts needed to accelerate key fuel cell processes.
A set of six precious metals known as the platinum-group metals have shown to be the most effective catalysts. While these metals are efficient and long-lasting, they are exceedingly costly due to their scarcity. As a consequence, researchers are looking for less expensive options.
Overcoming barriers
One such alternative has been iron-based catalysts. Iron is appealing because it is abundant and inexpensive. But it does not perform as well as platinum, especially because it lacks the durability to withstand the highly corrosive and oxidative environments inside fuel cells.
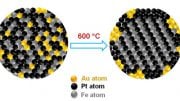
To overcome this barrier, the research team bonded four nitrogen atoms to the iron. Researchers then embedded the material in a few layers of graphene “with accurate atomic control of local geometric and chemical structures,” Wu says.
The resulting structure is a vastly improved catalyst. For example, the research team reported the catalyst:
- Is believed to be the most efficient iron-based catalyst produced to date, exceeding the DOE’s 2025 target for electric current density.
- Achieved a durability rating that approaches platinum group catalysts.
All this, Wu says, points to the iron-based catalyst’s potential to make fuel cells, particularly hydrogen fuel cells, much more affordable for commercial use. Researchers are planning follow-up studies to further improve the catalyst.
Reference: “Atomically dispersed iron sites with a nitrogen–carbon coating as highly active and durable oxygen reduction catalysts for fuel cells” by Shengwen Liu, Chenzhao Li, Michael J. Zachman, Yachao Zeng, Haoran Yu, Boyang Li, Maoyu Wang, Jonathan Braaten, Jiawei Liu, Harry M. Meyer III, Marcos Lucero, A. Jeremy Kropf, E. Ercan Alp, Qing Gong, Qiurong Shi, Zhenxing Feng, Hui Xu, Guofeng Wang, Deborah J. Myers, Jian Xie, David A. Cullen, Shawn Litster, and Gang Wu, 7 July 2022, Nature Energy.
DOI: 10.1038/s41560-022-01062-1
In addition to UB, the collaborative research team included members from the following organizations: Argonne National Laboratory; Carnegie Mellon University; Giner Inc.; Indiana University–Purdue University Indianapolis; Oak Ridge National Laboratory; Oregon State University; Purdue University; and the University of Pittsburgh.
The study was funded by the U.S. Department of Energy and the U.S. National Science Foundation. Wu and two co-authors have filed joint patent applications through the University at Buffalo and Giner Inc.







“Because they produce less or no emissions as compared to combustion engines,”
All fuel cells produce water as a by-product. If exhausted to the air, they will increase the relative humidity (RH). Increased RH will increase the Heat Index, and promote the growth of various fungi. Certainly having moist air, or condensed droplets of water in and around one’s laptop, is inviting problems. Moist air will cause corrosion of most metals and shorten the life span, including the contacts of a laptop keyboard.
Researchers continue to look at just the technical details of making the device work without consideration of the spectrum of problems that will need to be addressed before the devices will be commercially acceptable.
The affordability will be greatly impacted by the necessity of re-designing and replacing everything that will be negatively impacted by water or high humidity.
“Because they produce less or no emissions as compared to combustion engines,”
Clyde: The assumption is fuel cells would positively impact AGW. (AGW is also supposed to raise humidity.) Can you quantify how much more fuel cells would raise humidity in, say, the Los Angeles basin if all the cars emitted only water, compared to increased humidity due to a 1.5C increase in global temps?
The bit that seems to suggest laptops use internal combustion engines is of course just sloppy writing/editing. Everyone knows they are powered by caged squirrels (which do emit water but don’t appreciably increase local humidity).
I’m not going to take the time to actually do the calculations. However, as a first-order approximation, for the same amount of derived energy, all the CO2 will be replaced by water, so there should be a substantial increase in absolute humidity. Los Angeles is an interesting case because it has a Mediterranean climate, with most of the rain in the Winter. The Summers tend to be dry inland from the Summer fogs. So, one can expect a substantial increase in relative humidity and Heat Index because of the typical Summer Heat.
What is more problematic is cold Winter weather, such as New York, Chicago, or Duluth. I don’t think it unreasonable to expect that those locations will experience icing of not only the pavement, but also of sidewalks and power lines along the roads. Road salt is only useful down to about 15 deg F. Winter air is usually dry because the water vapor freezes. With new water vapor continuously being added, one can expect it to promptly freeze out onto all nearby surfaces.
Always thought the tiniest element was the most difficult to store and transport because it could sneak materials with “gaps”
Hydrogen is difficult to contain, and it also has a nasty habit of making the container or pipe walls brittle.
This article failed to mention that the search for a lower cost fuel cell catalyst has been going on for over a half-century. This article also failed to mention the cost of this new demonstration catalyst. Graphene has been promoted for decades and yet there is no mass production of the material.
It is extremely bad idea to use hydrogen as fuel for land/sea/air transportation because it is pretty much explosive!
Imagine a future world w/ all kinds of hydrogen vehicles, tanker trucks, gas stations everywhere!
Are we seriously thinking that there will be never any accidents/leaks/ruptures/mishandling to trigger massive explosions?
Not to mention, there is actually no need at all to use hydrogen as fuel!
All light/small vehicles are already becoming fully electric & all heavy/big land/sea/air vehicles just need us to start producing biodiesel/biofuel at large scales!
(From all possible industrial/agricultural/forestry waste/biomass & trash & sewage!)
Hopefully the additional testing that this theory goes through involves checking for palladium or other pgm traces…
If cella beads used behaves like a liquid at a reasonable cost can it solve hydrogen levels can it be pumped for miles along hydrogen pipe s.it can replace fossil fuels at pump trials and solve some of hydrogen s problem s.iron in pem cells could be great, cella hydrogen storage.any where can be posted, safely.and theoretical ly pump Rd miles along pipes.
The production of hydrogen from either renewable energy or petroleum is still not cheap. Also the transportation and storage of hydrogen presents many difficult problems that have not been resolved. hydrogen requires very low temperature store as a liquid. Hydrogen is very explosive and hard to handle.
And, a hydrogen flame is very difficult to detect.
A lot of people in the comments are putting forward really weak arguments against hydrogen without first thinking through the details. Firstly, if we’re able to scientifically show that the rise in humity caused by these cells is actually problematic, which we haven’t, machinery could simply include a condenser unit and a water tank to store the produced water. This could later be: emptied at the filling station; pumped into a vehicle’s wiper resovoir; reused in the cabin of larger transportation (such as trains, boats, and planes); or a combination of multiple.
Secondly, the facts that most light crafts have transitioned to full electric doesn’t affect hydrogen development, since hydrogen fuel cells also produce electricity. But the real reason why this argument is weak, is because it implies that battery technology; specifically, Li-Ion batteries – which are first in class – are somehow better than hydrogen fuel cells. They are not. We’re almost at the limit of what we can achieve with batteries and their energy density is still extremely lacking for 21st century requirements; not to mention the length of time it takes to recharge them in comparison to filling a fuel tank.
Thirdly, I’m not convinced that the production process for gasonline, diesel, and Li-Ion batteries are any more efficient than the production process for hydrogen. We’ve just had more time to optimise the production process and streamline delivery. Hydrogen as a fuel is still a novelty, which means that: the supporting infrastructure is limited, there’s little incentive to innovate, and the production volume is small. All of those things in tandem make is appear inviable but it is not.
And lastly, the explosiveness of hydrogen is not unique as far as fuels go. Gasoline and diesel are also very explosive. What I would concede to, is that hydrogen flames are more difficult to detect and thus pose a greater risk. That is a valid concern.
What I haven’t seen anyone highlight though, is the environmental impact of CO2 vs H2O. We’ve already seen the effect of carbon emissions. While water vapor might pose a few inconveniences I think we’re better off with water emissions. At this point in human civilization, we haven’t found the holy grail of fuel, so you have to pick your poison. Do you prefer CO2 which rises global temperatures and can’t be scrubbed out of the atmosphere at the same rate we produce it, or hydrogen which produces water and can washed out if the atmosphere in a couple hours?
“Firstly, if we’re able to scientifically show that the rise in humity caused by these cells is actually problematic, which we haven’t, …”
An observant person would notice that the exhaust for an ICE engine running on hydrocarbon fuels, immediately condenses into a small cloud as it exits the tailpipe in cold weather; even in warm weather, one can observe water dripping from the tailpipe when the engine first starts. From chemistry (science!), one can deduce that the only product from oxidizing hydrogen is water. If the CO2 produced by burning hydrocarbon fuels creates problems, why would you expect copious quantities of water vapor to be any different?
“… machinery could simply include a condenser unit and a water tank to store the produced water.”
There are potentially engineering solutions to dealing with the water by-product. Stationary power plants are most amenable to such solutions, economically. However, the issue really seems to be transportation. Even today, condensation trails from aircraft are a minor problem. Addressing the problem of fuel cells producing water carries a weight penalty that is even more severe for aircraft than for autos. There is also the problem of preventing the stored water from freezing when flying at 30,000 feet, and rupturing the storage tank. Cold climates (or seasons) are more problematic when dealing with water.
“Gasoline and diesel are also very explosive.”
Yes, but the range of air-to-fuel ratio explosivity is much wider for hydrogen. Also, the ignition temperature for hydrogen is lower than for gasoline, and especially diesel. One time I had visited a friend about an hours drive away. When I got home, I observed that I had gotten terrible gas mileage on the trip. I looked under the hood and found that the inline gas filter, which had a glass envelope, was cracked, and had been spewing gasoline onto the intake manifold, just above the hot exhaust manifold. There was no fire! I’m not sure I would have been so lucky if it had been hydrogen escaping.
“What I haven’t seen anyone highlight though, is the environmental impact of CO2 vs H2O.”
It is well known that water vapor is a more potent greenhouse gas than CO2 (absorption spectroscopy; science!). It is usually dismissed because any particular water molecule usually precipitates out in a few days. What is overlooked, is that evaporation and transpiration continually replenish the water vapor. Humans have increased the relative humidity by such things as building dams and using the reservoir water for pivotal irrigation, and relying on fossil fuels for transportation. Moving to a hydrogen economy will exacerbate the issue of replenishing precipitable water, particularly in arid and semi-arid climates with big cities.
One has to look at the big picture, and not just cherry pick the desirable characteristics of a proposed change in energy production.
Notice that I didn’t say we need to “scientifically prove there would be a rise in humidity”. I said we need to scientifically prove that this rise is “problematic”. Steam would obviously increase humidity, but that doesn’t mean it would ultimately pose a problem. In case you’ve forgotten, we’ve had steam engines before, so we’ve dealt with steam as a byproduct before. Obviously the volume would be much more now, but we don’t have evidence to claim that it would be problematic.
And secondly, I agree that a condenser unit isn’t practical for all use case in the same way mufflers aren’t practical for aircrafts. But it is a potential solution. And machinery doesn’t always need to store the produced water. For example boats could simply pump that water back out.
Also the autoignition temperature of hydrogen is higher than that of gasoline vapor.
I’m not the one cherry picking characteristics. All I’m saying is that the majority of the arguments that have been put forward, including yours, is based on conjecture, bias, or flat out ignorance.