New study by a team of Harvard-led researchers rebuts 75-year-old belief in reptile evolution.
Challenging a 75-year-old notion about how and when reptiles evolved during the past 300 million-plus years involves a lot of camerawork, loads of CT scanning, and, most of all, thousands of miles of travel. Just check the stamps in Tiago R. Simões’ passport.
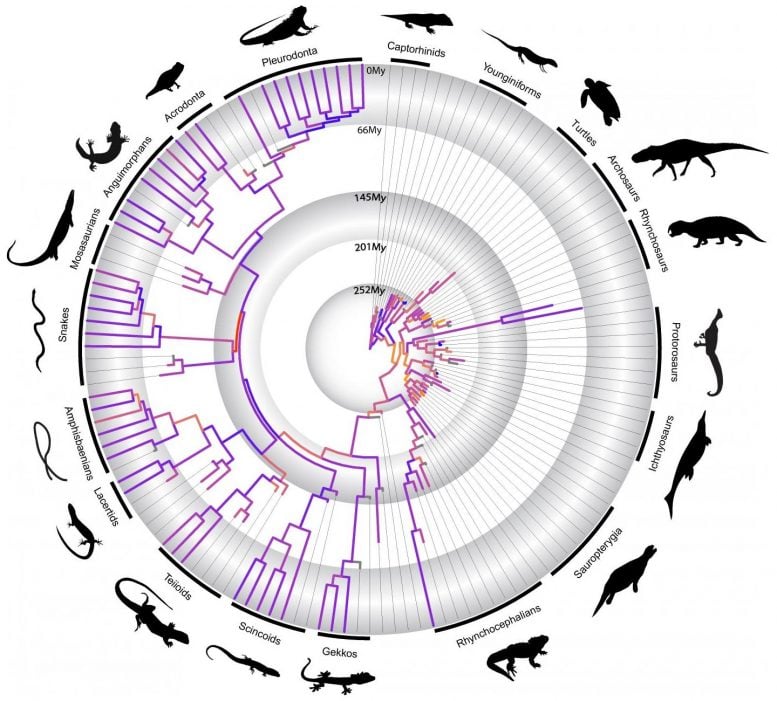
Simões is the Alexander Agassiz Postdoctoral Fellow in the lab of Harvard paleontologist Stephanie Pierce. From 2013 to 2018, he traveled to more than 20 countries and more than 50 different museums to take CT scans and photos of nearly 1,000 reptilian fossils, some hundreds of millions of years old. It amounted to about 400 days of active collection, helping form what is believed to be the largest available timeline on the evolution of major living and extinct reptile groups.
Now, a statistical analysis of that vast database is helping scientists better understand the evolution of these cold-blooded vertebrates by contradicting a widely held theory that major transitions in evolution always happened in big, quick (geologically speaking) bursts, triggered by major environmental shifts. The findings are described in a recently published paper in Nature Communications.
In it, researchers show that the evolution of extinct lineages of reptiles from more than 250 million years ago took place through many small bursts of morphological changes, such as developing armored body plans or wings for gliding, over a period of 50 million years instead of during a single major evolutionary event, as previously thought. They also show that the early evolution of most lizard lineages was a continuously slower and more incremental process than previously understood.

Tiago R. Simões gathered information from 50 museums, including Harvard’s Museum of Comparative Zoology. Credit: Tiago R. Simões
“It wasn’t a sudden jump that kind of established the wide diversity that we see today in reptiles,” Simões said. “There was an initial jump, but relatively small, and then a sustained increase over time of those rates [of evolution] and different diversity values.”
Evidence of this has been seen in other types of animals, but this is the first time it’s been seen in reptiles — one of the most diverse animals on the planet, with more than 10,000 different species and a dizzying variety of abilities and traits. Consider how some lizard species can freeze solid overnight then thaw the next morning, or how turtles grow protective armor.
The findings run contrary to the evolutionary theory of adaptive radiation that Harvard paleontologist George G. Simpson popularized in the 1940s, which sought to explain the origins of the planet’s biological diversity. Adaptive radiation has been the focus of intense investigation for decades, but wasn’t until recent years that the technology, methods, and data have existed to precisely measure rapid rates of evolution in the fossil record in terms of different animal species, morphologies, and at the molecular level using DNA.
Researchers of this study also included Pierce, the Thomas D. Cabot Associate Professor of Organismic and Evolutionary Biology and curator of vertebrate paleontology in the Museum of Comparative Zoology; Oksana Vernygora, a graduate student from the University of Alberta in Canada; and Professor Michael Wayne Caldwell at Alberta.
Simões traveled to almost all of the world’s major natural history museums to collect the data for the study, including the national natural history museums in London, Paris, Berlin, Ottawa, Beijing, and Tokyo. In the U.S., he visited the Smithsonian National Museum of Natural History, the Carnegie Museum of Natural History, and Harvard’s Museum of Comparative Zoology.
The scientists believe that by understanding how animals evolve over longer periods of time, they can glean a number of lessons on ecology and how organisms are affected by environmental changes. Using the database, researchers can determine when major reptile lineages or morphologies originated, see how those changes affected reptile DNA, and learn important lessons about how species were impacted by historical events.

Tiago R. Simões’ research has provided an extensive database that is helping scientists better understand the evolution of reptiles. Credit: Tiago R. Simões
Reptiles, for instance, have survived three major mass extinction events. The biggest was the Permian-Triassic mass extinction about 250 million years ago that killed about 90 percent of the planet’s species, earning it the moniker the Great Dying. It’s believed to have been caused by a buildup of natural greenhouse gases.
The timeline researchers created found that the rates at which reptiles were evolving and the anatomical differences among them before the Great Dying were nearly as high as after the event. However, it was only much after the Great Dying that reptiles became dominant in many ecosystems and extremely diverse in terms of the number of different species.
That finding cemented that fast rates of anatomical change don’t need to coincide with genetic diversity or an abundance of species (called taxonomic diversity), and further rebutted adaptive radiation as the only explanation for the origin of new animal groups and body plans. The researchers also note that it took reptiles almost 10 million years to recover to previous levels of anatomical diversity.
“That kind of tells you on the broad scheme of things and on a global scale how much impact, throughout the history of life, sudden environmental changes may have,” Simões said.
Further evidence that contradicted adaptive radiation included similar but surprising findings on the origins of snakes, which achieved the major aspects of their skinny, elongated body plans early in their evolution about 170 million years ago (but didn’t fully lose their limbs for another 105 million years). They also underwent rapid changes to their skulls about 170 to 165 million years ago that led to such powerful and flexible mouths that today they can swallow whole prey many times their size. But while snakes experienced the fastest rates of anatomical change in the history of reptile evolution, these changes did not coincide with increases in taxonomic diversity or high rates of molecular evolution as predicted by adaptive radiations, the researchers said.
The scientists weren’t able to pinpoint why this mismatch happens, and suggested more research is needed. In particular they want to understand how body plans evolve and how changes in DNA relate to it.
“We can see better now what are the big changes in the history of life and especially in the history of reptile life on Earth,” Simões said. “We will keep digging.”
Reference: “Megaevolutionary dynamics and the timing of evolutionary innovation in reptiles” by Tiago R. Simões, Oksana Vernygora, Michael W. Caldwell and Stephanie E. Pierce, 3 July 2020, Nature Communications.
DOI: 10.1038/s41467-020-17190-9
This work was supported by an Alexander Agassiz Postdoctoral Fellowship from the Harvard Museum of Comparative Zoology and by the National Science and Engineering Research Council of Canada.








Very Interesting.
Had always beleived that just like the electron could behave both as a partile and wave at the quantum level , Mother Nature too could both behave as a long drwan out gradual process and may also behave as a sudden process (existing radiation theory) on a different time scale. Never had the proof for this theory. Thanks for providing the proof for the Reptile Evolution based on actual data and time scale which are much larger.
Have always joked about the people living in highly polluted and perpetually major metros like New Delhi and Mumbai ( where I live), growing Gills for breathing , like fishes in the Oceans! Ofocurse this assumes that the human species will survive the foul air and adapt, to breathing what the population has been experiancing in the Western States in USA!
Evolution of the Human Species and Humanoid branches, based on such data would be interesting. Also, the impact on the species in both the short term and longer term would also be extremely interesting research , based on data. Dont know if fossil samples exist which can enable such an analysis, based on data rather than assumptions. Ofcourse the evolution of the many other creatures of God would also be very interesting, as long as it is based on data with minimum amount of assumptions.
So basically there is still no evidence to evolution.
Evolution Is a belief system, like a religion and the definition of science (secular science) excludes other religions just as Judaism, Christianity, Islam and other religions exclude other belief systems. It takes allot of faith to believe evolution and all the circular reasoning, changing evidence, and lack of evidence. I choose to have faith In God.