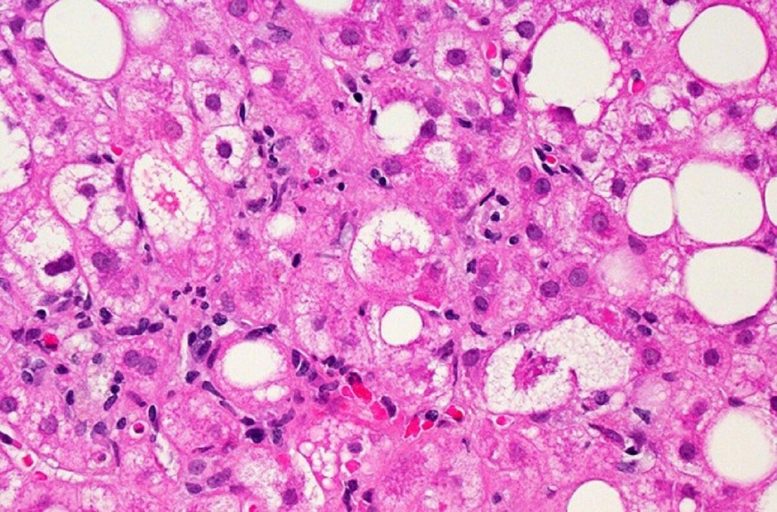
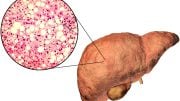
A microscopic image of liver tissue affected by non-alcoholic fatty liver disease (NAFLD). The large and small white spots are excess fat droplets filling liver cells (hepatocytes). Credit: Dr. David Kleiner, NCI
Tesamorelin prevented progression to liver fibrosis in NIH study.
Researchers at the National Institutes of Health and their colleagues at Massachusetts General Hospital (MGH) in Boston report that the injectable hormone tesamorelin reduces liver fat and prevents liver fibrosis (scarring) in people living with HIV. The study was conducted by the National Institute of Allergy and Infectious Diseases (NIAID) and the National Cancer Institute, both parts of NIH. The findings were published online on October 11, 2019, in The Lancet HIV.
“Many people living with HIV have overcome significant obstacles to live longer, healthier lives, though many still experience liver disease,” said NIAID Director Anthony S. Fauci, M.D. “It is encouraging that tesamorelin, a drug already approved to treat other complications of HIV, may be effective in addressing the non-alcoholic fatty liver disease.”
Non-alcoholic fatty liver disease, or NAFLD, frequently occurs alongside HIV, affecting as many as 25% of people living with HIV in the developed world. However, no effective treatments currently exist to treat the condition, which is a risk factor for progressive liver disease and liver cancer. Investigators led by Colleen M. Hadigan, M.D., senior research physician in NIAID’s Laboratory of Immunoregulation, and Steven K. Grinspoon, M.D., Chief of the Metabolism Unit at MGH, tested whether tesamorelin could decrease liver fat in men and women living with both HIV and NAFLD. Among the participants enrolled, 43% had at least mild fibrosis, and 33% met the diagnostic criteria for a more severe subset of NAFLD called nonalcoholic steatohepatitis (NASH). Thirty-one participants were randomized to receive daily 2-mg injections of tesamorelin, and 30 were randomized to receive identical-looking injections containing a placebo. Researchers provided nutritional counseling to all participants, as well as training in self-administering the daily injections. Researchers then compared measures of liver health in both groups at baseline and 12 months.
After one year, participants receiving tesamorelin had better liver health than those receiving placebo, as defined by a reduction in hepatic fat fraction (HFF)—the ratio of fat to other tissue in the liver. The healthy range for HFF is less than 5%. Thirty-five percent of study participants receiving tesamorelin achieved a normal HFF, while only 4% of those on placebo reached that range with nutritional advice alone. Overall, tesamorelin was well-tolerated and reduced participants’ HFF by an absolute difference of 4.1% (corresponding to a 37% relative reduction from the beginning of the study). While nine participants receiving placebo experienced onset or worsening of fibrosis, only two participants in the tesamorelin group experienced the same. Additionally, levels of several blood markers associated with inflammation and liver damage—including the enzyme alanine aminotransferase (ALT)—decreased more among those taking tesamorelin compared to those on a placebo, particularly among those with increased levels at the beginning of the study.
Given these positive results, investigators suggest expanding the indication for tesamorelin to include people living with HIV who have been diagnosed with NAFLD. They also recommend additional research to determine if tesamorelin could contribute to long-term protection against serious liver disease in people without HIV.
“Our hope is that this intervention may help people living with HIV, as well as benefit HIV-negative people with liver abnormalities,” said Dr. Hadigan. “Further research may inform us of the potential long-term benefits of this approach and develop formulations that can benefit everyone with liver disease, regardless of HIV status.”
Egrifta (tesamorelin) was approved in 2010 by the U.S. Food and Drug Administration to reduce excess abdominal fat in HIV patients with lipodystrophy—a complication characterized by an abnormal distribution of body fat initially associated with older classes of HIV medications. The most commonly reported side effects in previous clinical trials evaluating Egrifta included joint pain (arthralgia), skin redness and rash at the injection site (erythema and pruritis), stomach pain, swelling, and muscle pain (myalgia). Worsening blood sugar control occurred more often in trial participants treated with Egrifta than with placebo.
“Because tesamorelin proved effective in treating abnormal fat build-up in the abdomens of people in the context of HIV and related medication use, we hypothesized that the drug might also reduce fat that accrues in the liver and causes damage in a similar population,” said Dr. Grinspoon.
While liver disease is often associated with heavy alcohol use, NAFLD occurs when excess fat builds up in the liver without alcohol as a contributing factor. This condition may progress to liver damage, cirrhosis, or cancer that could be life-threatening and necessitate liver transplantation.
Previous studies have found that vitamin E supplements, weight loss, and other lifestyle changes can improve outcomes among HIV-negative people with NASH. However, treatment options for NASH and NAFLD are often not tested in people with HIV and none are available for this group. Obesity and type 2 diabetes raise the risk of developing NAFLD regardless of HIV status, and people with HIV are at increased risk of NAFLD because some HIV medications and HIV itself are associated with gaining abdominal fat and may contribute to liver fat build-up.
Reference: “Effects of tesamorelin on non-alcoholic fatty liver disease in HIV: a randomised, double-blind, multicentre trial” by Takara L Stanley, MD; Lindsay T Fourman, MD; Meghan N Feldpausch, ANP; Julia Purdy, CRNP; Isabel Zheng, BS; Chelsea S Pan, BA; Julia Aepfelbacher, BS; Colleen Buckless, MS; Andrew Tsao, BS; Anela Kellogg, MSN; Karen Branch, RN; Hang Lee, PhD; Chia-Ying Liu, MD; Kathleen E Corey, MD; Raymond T Chung, MD; Martin Torriani, MD; Prof David E Kleiner, MD; Colleen M Hadigan, MD and Prof Steven K Grinspoon, MD, 11 October 2019, The Lancet HIV.
DOI: 10.1016/S2352-3018(19)30338-8
This research was supported through NIAID grant U01 AI115711.





Be the first to comment on "FDA Approved Drug Reverses Signs of Liver Disease in People Living With HIV"