
University of Delaware researchers have broken new ground that could bring more environmentally friendly fuel cells closer to commercialization. Credit: Graphic illustration by Jeffrey C. Chase
University of Delaware researchers’ carbon capture advance could bring environmentally friendly fuel cells closer to market.
University of Delaware engineers have demonstrated a way to effectively capture 99% of carbon dioxide from air using a novel electrochemical system powered by hydrogen.
It is a significant advance for carbon dioxide capture and could bring more environmentally friendly fuel cells closer to market.
The research team, led by UD Professor Yushan Yan, reported their method in Nature Energy on Thursday, February 3.
Game-changing tech for fuel cell efficiency
Fuel cells work by converting fuel chemical energy directly into electricity. They can be used in transportation for things like hybrid or zero-emission vehicles.
Yan, Henry Belin du Pont Chair of Chemical and Biomolecular Engineering, has been working for some time to improve hydroxide exchange membrane (HEM) fuel cells, an economical and environmentally friendly alternative to traditional acid-based fuel cells used today.
But HEM fuel cells have a shortcoming that has kept them off the road — they are extremely sensitive to carbon dioxide in the air. Essentially, the carbon dioxide makes it hard for a HEM fuel cell to breathe.
This defect quickly reduces the fuel cell’s performance and efficiency by up to 20%, rendering the fuel cell no better than a gasoline engine. Yan’s research group has been searching for a workaround for this carbon dioxide conundrum for over 15 years.
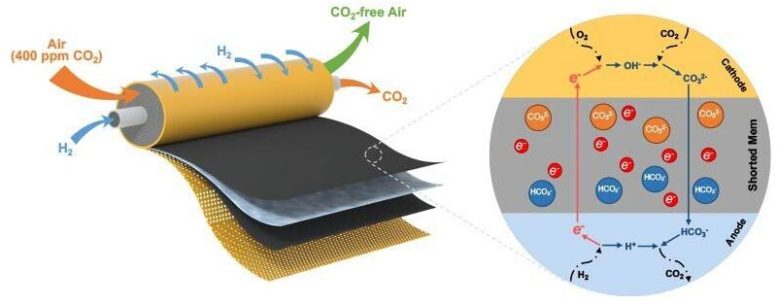
The UD research team’s spiral wound module takes in hydrogen and air through two separate inlets (shown on the left) and emits carbon dioxide and carbon dioxide-free air (shown on the right) after passing through two large-area, catalyst-coated shorted membranes. The inset image on the right shows, in part, how the molecules move within the short-circuited membrane. Credit: University of Delaware
A few years back, the researchers realized this disadvantage might actually be a solution — for carbon dioxide removal.
“Once we dug into the mechanism, we realized the fuel cells were capturing just about every bit of carbon dioxide that came into them, and they were really good at separating it to the other side,” said Brian Setzler, assistant professor for research in chemical and biomolecular engineering and paper co-author.
While this isn’t good for the fuel cell, the team knew if they could leverage this built-in “self-purging” process in a separate device upstream from the fuel cell stack, they could turn it into a carbon dioxide separator.
“It turns out our approach is very effective. We can capture 99% of the carbon dioxide out of the air in one pass if we have the right design and right configuration,” said Yan.
So, how did they do it?
They found a way to embed the power source for the electrochemical technology inside the separation membrane. The approach involved internally short-circuiting the device.
“It’s risky, but we managed to control this short-circuited fuel cell by hydrogen. And by using this internal electrically shorted membrane, we were able to get rid of the bulky components, such as bipolar plates, current collectors or any electrical wires typically found in a fuel cell stack,” said Lin Shi, a doctoral candidate in the Yan group and the paper’s lead author.
Now, the research team had an electrochemical device that looked like a normal filtration membrane made for separating out gases, but with the capability to continuously pick up minute amounts of carbon dioxide from the air like a more complicated electrochemical system.

This picture shows the electrochemical system developed by the Yan group. Inside the highlighted cylindrical metal housing shown is the research team’s novel spiral wound module. As hydrogen is fed to the device, it powers the carbon dioxide removal process. Computer software on the laptop plots the carbon dioxide concentration in the air after passing through the module. Credit: University of Delaware
In effect, embedding the device’s wires inside the membrane created a short-cut that made it easier for the carbon dioxide particles to travel from one side to the other. It also enabled the team to construct a compact, spiral module with a large surface area in a small volume. In other words, they now have a smaller package capable of filtering greater quantities of air at a time, making it both effective and cost-effective for fuel cell applications. Meanwhile, fewer components mean less cost and, more importantly, provided a way to easily scale up for the market.
The research team’s results showed that an electrochemical cell measuring 2 inches by 2 inches could continuously remove about 99% of the carbon dioxide found in air flowing at a rate of approximately two liters per minute. An early prototype spiral device about the size of a 12-ounce soda can is capable of filtering 10 liters of air per minute and scrubbing out 98% of the carbon dioxide, the researchers said.
Scaled for an automotive application, the device would be roughly the size of a gallon of milk, Setzer said, but the device could be used to remove carbon dioxide elsewhere, too. For example, the UD-patented technology could enable lighter, more efficient carbon dioxide removal devices in spacecraft or submarines, where ongoing filtration is critical.
“We have some ideas for a long-term roadmap that can really help us get there,” said Setzler.
According to Shi, since the electrochemical system is powered by hydrogen, as the hydrogen economy develops, this electrochemical device could also be used in airplanes and buildings where air recirculation is desired as an energy-saving measure. Later this month, following his dissertation defense, Shi will join Versogen, a UD spinoff company founded by Yan, to continue advancing research toward sustainable green hydrogen.
Reference: “A shorted membrane electrochemical cell powered by hydrogen to remove CO2 from the air feed of hydroxide exchange membrane fuel cells” by Lin Shi, Yun Zhao, Stephanie Matz, Shimshon Gottesfeld, Brian P. Setzler and Yushan Yan, 3 February 2022, Nature Energy.
DOI: 10.1038/s41560-021-00969-5
Co-authors on the paper from the Yan lab include Yun Zhao, co-first author and research associate, who performed experimental work essential for testing the device; Stephanie Matz, a doctoral student who contributed to the designing and fabrication of the spiral module, and Shimshon Gottesfeld, an adjunct professor of chemical and biomolecular engineering at UD. Gottesfeld was principal investigator on the 2019 project, funded by the Advanced Research Projects Agency-Energy (ARPA-E), that led to the findings.








You do realize that plants need CO2 to live, yes? The lower limit for plant life is around 180 ppm; the current levels are actually still too low for optimal growth for some species.
Don’t worry Paul. There are no technologies, even scaled up globally, that can take even one ppm of CO2 from the atmosphere. We are subsidizing a carbon capture and storage industry that can’t possibly work. One ppm of CO2 is 7,800 million metric tons.
This is Ken M. level trolling right here
OK. Sounds good. Does the hydrogen production offset the carbon dioxide process?!! How’s the power production to input ratio?!
Too bad the production of hydrogen is so energy intensive, and still requires the burning of fossil fuels to power the process. The experimental approaches always wind up failing to reduce the consumption of fossil fuel, which is ALWAYS being used at some point in the process. Hydrogen is not an energy “source” (and neither is solar or wind power). Hydrogen must first be created in order to be used in this proposal. So far, every fuel cell known to man has these same problems, and thus will never be brought to scale.
JR is right. There are no alternative technologies or renewable energies that can be manufactured, distributed and installed without using vehicles that require biofuels that are 90% fossil fuel. Any serious attempt to make a cost-benefit analysis would have to include that.
Wow. So many people fail to understand the bigger picture. Yes, there cannot be ANY process on Earth that humans require that won’t rely on fossil fuels somewhere in the causal chain. What an amazing insight.
The point is that research must be done to get us all anywhere close to a final goal of at least carbon neutral. Of COURSE the tech won’t be truly carbon neutral at first. That is because we can’t just design and replace the entire paradigm at once. Pieces of the paradigm have to be replaced and over time you eventually get to a place of carbon neutral.
And you don’t have to replace the entire paradigm to make a dent. If we just cut our co2 in half we would make a difference. That still allows for commercial transport via trains and trucks, excavation of raw materials, and manufacturing.
This isn’t even CO2 capture as a solution to CO2 in the atmosphere. This article was about finding a way to keep the degrading CO2 out of a battery cell. This tech results in raising the battery efficiency by 20%.
The point is that you can start with the small bits and develop technology gradually. We had low range ev cars quite a while ago. Then they figured out how to make them reasonably long range. Then they figured out how to make them affordable, partially with the government assisting with tax subsidies. Then they figured out how to make reasonably powerful ev trucks. Then ev semi trucks. Ev aircraft. Ev boats. You have to start somewhere and it won’t be truly CO2 neutral any time soon. But that doesn’t mean you don’t START or push the tech ever harder.
It’s like the use of tax dollars for NASA. Yes, we ain’t colonizing Mars any time soon. But that doesn’t mean we just don’t go to space. We benefit because inventions and discoveries from one field can give us wd40, CD players and even immaterial things like inspiration and hope.
Thank you for being the voice of sanity.
These academics are (mostly) well-intentioned, but they simply have no clue about conducting process operations at scale.
Let’s do the math. The CO2 concentration is ~420 ppm. So. even if you get ALL of the CO2 out of every bit of air you process, you must process 1/420th of the entire atmosphere in order to remove 1 ppm. How much is that?
The Earth’s atmosphere weighs a little over 11 QUINTILLION pounds. After a few calculations to get it into common industrial units of gas processing, that’s about 138 Quintillion standard cubic feet (SCF). 1/420th of that is about 330 QUADRILLION SCF. That’s how much air we need to process to remove 1 ppm. Is that a lot?
In 2019, the most productive year ever for the natural gas processing industry, they processed 800 Billion SCF/day of gas. Let’s say we work just as hard to remove CO2. To process enough air to remove 1 ppm of CO2, we’d have to work at that rate for 330 Quadrillion SCF / 800 Billion SCF/day = 411,000 days = 1,126 YEARS.
And bear in mind the gas processing industry goes to all that trouble because the gas they produce has VALUE in the marketplace. The CO2 these guys plan to capture is worse than worthless, because once they’ve captured it, they have to put it somewhere (sequestration) or convert it to something valuable (utilization). Doing either of those things will also be very costly, and it will require operating at the same daunting scale as the capture process itself.
These are clueless academics, greedy entrepreneurs, and gullible investors combining to waste resources on a MASSIVE scale that could instead be used productively. We DO need to reduce the CO2 content of our atmosphere. The trolls talking about healthy plants are just as dangerous as these naïve Professors — doing nothing about it is just as bad doing something incredibly inefficient.
We need to drive fossil CO2 emissions as close to zero as possible and leverage natural processes to remove the CO2 that has already been emitted (reforestation, afforestation, geo-mineralization, etc.). There are near-, middle-, and long-term ways to achieve that, but this CO2 capture idea makes no sense on ANY time scale.
This article mentions applications such as more efficient fuel cells and removing CO2 from spacecraft, submarines, airplanes, and buildings. Nowhere does it mention removing CO2 from the entire atmosphere! For fuel cells and scrubbing the air in these smaller spaces this could be a significant advance.
Very interesting.
Here is a thought for consideration.
Nested IF.
Two hydroxyl fuel cells integrated as one. Replicated …
One for filtering out carbon di oxide and then use the 98 percent Carbon di Oxide free Air to power the fuel cell generating power for the automobile.
The first fuel cell which has absorbed the carbon di Oxide can be washed out as carbonic acid and used for plant growth.
A closed loop with alternating hydroxyl Fuel cells to be designed with source of power being solar power. Superconducting material stable at room temperature to minimise extraneous power losses may help efficiency reach new heights.
Views expressed are personal and not binding on anyone.
“… carbon capture advance could bring environmentally friendly fuel cells closer to market.”
When using hydrogen as a feedstock for the fuel cells, the byproduct will be water. There will be a strong temptation to vent the water vapor to the atmosphere, rather than condense it, carry it around all day, and then drain it at the end of the day. Water vapor is a greenhouse gas in its own right, actually more important than CO2. It will also increase the relative humidity of cities in the Summer, increasing the heat index. In the Winter, it will condense and create fog, or settle on roads making them slippery and dangerous.
Most of these researchers have a myopic view of the problem they claim to be trying to solve.
Now that you’ve separated out the carbon dioxide what are you going to do with it
In this instance, they dump it back into the air. The immediate objective is to deliver de-carboxied air to the fuel cell, so it works better. But, if you want, you could use that CO2 for something, from root beer on up, and instead dump the 5ppm CO2 air into the air.
… why let it to an atmosphere after all, you trow things around, then you make mess, and then you keep it clean…
When you grow vegetables you can put then in a greenhouse. If you add co² to the greenhouse the veggies grow faster.
put a clic article a techno to help puluting fuel cell to work more ( so to polute more) .That cant be use to fifht climat change…have a minimum thinking before lie to people by your greewashing you are complice of crime to humanity. “rendez vous” at NUREMBERG…