Researchers found phospholipids to be essential in epithelial cell adhesion.
Phosphatidylinositol bisphosphate (PIP2), a phospholipid, is essential for epithelial cell-cell adhesion and maintaining cellular identity.
Body cells in multicellular organisms adhere to one another to form tissues that perform various physiological functions. Epithelial cells form our skin and lining surfaces, such as the gut and other ducts, and protect our internal organs.
Epithelial cells are a type of cell that lines the surfaces of your body. They are found on your skin, blood vessels, urinary tract, and organs.
To maintain the integrity of an organism and function properly, it is important for these cells to remain attached to each other. They do so through specific types of cellular junctions. These junctions are characterized by proteins, which also help in maintaining cellular identity.
The loss of these proteins from cell surfaces causes them to lose their identity as epithelial cells, prompting their transformation into mesenchymal cells (through a process known as epithelial-mesenchymal transformation, or EMT), and subsequently, their progression towards cancer and fibrosis.
These cancerous cells are only loosely adherent to each other (given that the proteins that helped maintain cellular adhesion are now lost), so they may separate from each other, migrate into the bloodstream, and cause the cancer to metastasize (spread to other parts of the body).
Now, while the role of proteins in maintaining cellular identity is well-researched, we can’t help but wonder–do lipids (fatty molecules) also play a role in characterizing cells and preventing EMT?
Under the guidance of Dr. Yoshikazu Nakamura and Dr. Kaori Kanemaru, scientists from Tokyo University of Science (TUS), Tokyo University of Pharmacy and Life Sciences, Tokyo Medical and Dental University, Akita University, Hokkaido University, and Kobe University have tried to find an answer to this question.
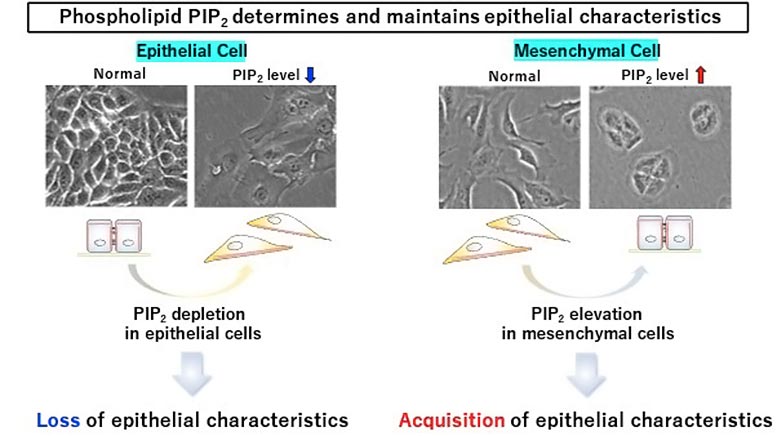
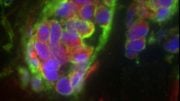
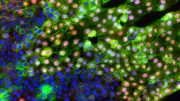
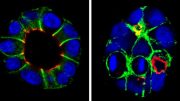
The phospholipid PIP2 plays a critical role in maintaining the characteristics of epithelial cells. Credit: Yoshikazu Nakamura from Tokyo University of Science
“We know lipids are an important class of biomolecules, necessary for certain cellular functions. One such lipid, a phosphatidylinositol, forms a phospholipid called phosphatidylinositol bisphosphate (PIP2),” Associate Professor Dr. Nakamura from TUS dives into the topic. He tells us that PIP2 is important because it is crucial for the formation of signaling molecules that regulate cell proliferation, survival, and migration. “We had evidence that higher amounts of PIP2 were found in the epidermal layer of skin, so we hypothesized that this phospholipid contributed to the properties and characterization of epithelial cells.”
The findings from their study will be published in the journal Nature Communications today (May 9, 2022). The paper describes how the team used a battery of analytical techniques including chromatography, mass spectroscopy, immunofluorescence, retroviral expression, and real-time quantitative PCR to confirm that PIP2 plays a critical role in the determination of epithelial identity.
“We saw that epithelial cells lost their properties when PIP2 was depleted from their cell membranes. On the other hand, osteosarcoma cells (which are cancerous, non-epithelial cells) gained epithelial cell-like properties when PIP2 was produced in their plasma membranes,” says Dr. Nakamura, with a look of excitement. The group was also able to show that PIP2 regulates these epithelial properties by recruiting Par3—a protein that guides vesicles intracellularly—to the plasma membrane. Once in the plasma membrane, Par3 facilitates the formation of adherens junctions (one of the cellular junctions discussed above) which anchor neighboring cells together. This partially prevents EMT, and hence, progression of cancer.
“So,” Dr. Nakamura explains, “In theory, PIP2’s partial inhibition of EMT could halt cancer progression, making this phospholipid an attractive target molecule for anti-cancer treatment.”
TUS’ research has opened a new avenue for the development of anti-cancer drug development, possibly giving us a solution that will “stick.”
Reference: “Plasma membrane phosphatidylinositol (4,5)-bisphosphate is critical for determination of epithelial characteristics” by Kaori Kanemaru, Makoto Shimozawa, Manabu Kitamata, Rikuto Furuishi, Hinako Kayano, Yui Sukawa, Yuuki Chiba, Takatsugu Fukuyama, Junya Hasegawa, Hiroki Nakanishi, Takuma Kishimoto, Kazuya Tsujita, Kazuma Tanaka, Toshiki Itoh, Junko Sasaki, Takehiko Sasaki, Kiyoko Fukami and Yoshikazu Nakamura, 9 May 2022, Nature Communications.
DOI: 10.1038/s41467-022-30061-9
Funding: Takeda Science Foundation, Sumitomo Foundation, Terumo Life Science Foundation, Mochida Memorial 1 Foundation for Medical and Pharmaceutical Research, Ichiro Kanehara Foundation, Hamaguchi Foundation for the Advancement of Biochemistry



Be the first to comment on "Getting Sticky With It: PIP2 Discovered To Play a Key Role in Epithelial Cell Adhesion"