
Animal form is shaped not only by genetics but also by mechanical forces and self-organization. This principle has implications for understanding tumor formation and animal development
Researchers at the RIKEN Center for Biosystems Dynamics Research (BDR) in Japan have identified a new mechanism that helps animals to develop with precise and constant form.
The reproducibility of form, shape, and characteristic appearance is a key feature of our development that is made possible because their instructions are coded in our DNA. What is perplexing, however, is how this reproducibility is achieved despite genetic variation and developmental “noise” resulting from environmental, physical and chemical fluctuations. Recent work in fruit flies has suggested that “noise-canceling” mechanisms in the embryo rely on a detailed and highly reproducible genetic “blueprint” with specific instructions down to the single-cell level.
Now, in research published in Developmental Cell, an international team led by Yu-Chiun Wang at RIKEN BDR asked whether this blueprint is sufficient to explain developmental consistency, or whether it is helped by alternative noise-canceling mechanisms. Their findings indicate a previously overlooked role for the mechanical forces that sculpt the embryo, as they turn out to be the noise-producing culprit as well as the key to ensuring precision – a true double-edged sword!
In their work, the team investigated a structure called the cephalic furrow in the fruit fly embryo, in which the surface of the embryo folds along a straight line in an origami-like fashion. To make this fold, the cells deploy a molecule called myosin to exert mechanical forces that shorten the cells making up the fold. What was surprising, however, was that on average 20% of the cells did not receive the instructions to become part of the cephalic furrow. It turned out that the information of where to make a fold was precise, but the reading of this information was unexpectedly sloppy. As a result, myosin distribution was highly variable, resulting in a discrepancy between the blueprint information and cell behavior. “These results were very puzzling as generations of developmental biologists were in awe of how the genetic blueprint could instruct machine-like precision of development,” says Wang.
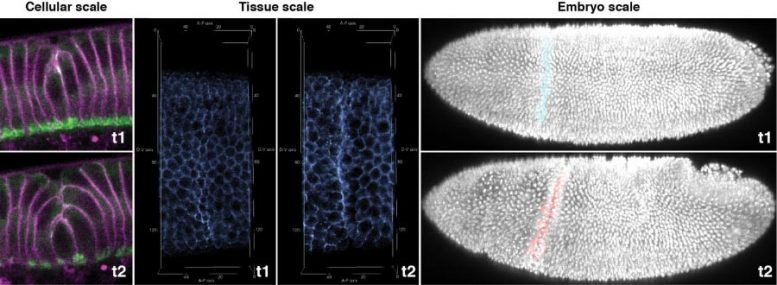
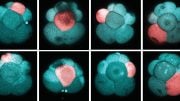
The origami of the fruit fly embryo – the cephalic furrow – shown at three different scales at an early (t1) and a late (t2) timepoints. The forces that shorten the cells (cellular scale) have a fuzzy distribution across the tissue (tissue scale, left), from which a straight “ribbon” emerges as a result of mechanical self-organization (tissue scale, right) that in turn ensures a straight crease (right, embryo scale). Credit: RIKEN
To find out how the embryo folds along a straight crease as in an origami despite this “noise”, the researchers looked more broadly across the tissue and realized that myosin is polarized along the direction parallel to the forming crease. They hypothesized that, powered by myosin, the cell membranes pull on each other, creating a form of mechanical communication that allows a straightened ribbon-like structure to emerge out of a fuzzy zone of stochastic membrane contraction. It appeared that this myosin-dotted ribbon is the crease of the developing cephalic furrow.
To show that this was indeed the case, the scientists cut the ribbon used a sharply focused laser beam to inactivate myosin in a small number of cells. They found that the cephalic furrow developed a kink, indicating that the straightness of the folding requires an intact ribbon of contractile membranes. Computer simulations also confirmed that tissue folding based on polarized contractile forces indeed can overcome the noise in contractile forces.
The conclusion of this study is that the constancy of animal form requires more than just the deterministic process of genetic inheritance and genetic networks, but also relies on the stochastic and emergent behaviors of mechanical forces. “This work taught us that constancy in biology stems not only from its regulatory complexity, but also from the unique noise-and-self-correction principle of self-organization. This is a missing chapter in developmental biology textbooks,” Wang says. Wang also thinks that by the same token, pathological processes that involve growth, reorganization and changing cell and tissue shapes, such as tumor formation and cancer metastasis, must also contain an element of mechanical self-organization, alongside the well-known factor of genetic susceptibility. “The cephalic furrow is a very pronounced structure,” he continues, “yet it forms and disappears about one hour after its initiation. This mysterious, beautiful and yet ephemeral structure of “epithelial origami” continues to mesmerize us and teach us things that we haven’t yet understood about animal development.”
Reference: “Tissue-Scale Mechanical Coupling Reduces Morphogenetic Noise to Ensure Precision during Epithelial Folding” by Anthony S. Eritano, Claire L. Bromley, Antonio Bolea Albero, Lucas Schütz, Fu-Lai Wen, Michiko Takeda, Takashi Fukaya, Mustafa M. Sami, Tatsuo Shibata, Steffen Lemke and Yu-Chiun Wang, 12 March 2020, Developmental Cell.
DOI: 10.1016/j.devcel.2020.02.012



Be the first to comment on "Mechanical Forces Shape Animal “Origami” Precisely Despite “Noise” and Genetic Variation"