
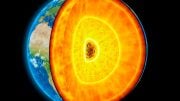
Below Earth’s thin crust lies the mantle, enveloping the core about 1,800 miles beneath us. Around 410 miles deep, seismic signals reveal a significant transition where the upper and lower mantle meet, causing a notable change in seismic wave speed at this boundary. It’s unclear if this is just a physical boundary or if the rock’s chemical composition changes here. The prevailing notion is that the Earth’s mantle has a relatively uniform chemical composition. Nonetheless, experiments performed by ETH researchers reveal this perspective to be overly simplistic.
The current understanding is that the chemical composition of the Earth’s mantle is relatively homogeneous. However, experiments conducted by ETH researchers now show that this view is too simplistic. Their results solve a key problem facing the geosciences – and raise some new questions.
There are places that will always be beyond our reach. The Earth’s interior is one of them. But we do have ways of gaining an understanding of this uncharted world. Seismic waves, for instance, allow us to put important constraints about the structure of our planet and the physical properties of the materials hidden deep within it. Then there are the volcanic rocks that emerge in some places on the Earth’s surface from deep within and provide important clues about the chemical composition of the mantle. And finally, there are lab experiments that can simulate the conditions of the Earth’s interior on a small scale.
A new publication by Motohiko Murakami, Professor of Experimental Mineral Physics, and his team was featured recently in the journal PNAS and shows just how illuminating such experiments can be. The researchers’ findings suggest that many geoscientists’ understanding of the Earth’s interior may be too simplistic.

ETH researchers use a complex test array to examine the behavior of rock deep in the Earth’s interior. The specimen is in the block in the center of the photograph. Credit: M. Murakami, ETH Zurich
Dramatic change
Below the Earth’s crust, which is only a few kilometers thick, lies its mantle. Also made of rock, this surrounds the planet’s core, which begins some 2,900 kilometers (1,800 miles) below us. Thanks to seismic signals, we know that a dramatic change occurs in the mantle at a depth of around 660 kilometers (410 miles): this is where the upper mantle meets the lower mantle and the mechanical properties of the rock begin to differ, which is why the propagation velocity of seismic waves changes dramatically at this border.
What is unclear is whether this is merely a physical border or whether the chemical composition of the rock also changes at this point. Many geoscientists presume that the Earth’s mantle as a whole is composed relatively consistently of magnesium-rich rock, which in turn has a composition similar to that of peridotite rock found on the Earth’s surface. These envoys from the upper mantle, which arrive on the Earth’s surface by way of events like volcanic eruptions, exhibit a magnesium-silicon ratio of ~1.3.
“The presumption that the composition of the Earth’s mantle is more or less homogeneous is based on a relatively simple hypothesis,” Murakami explains. “Namely that the powerful convection currents within the mantle, which also drive the motion of the tectonic plates on the Earth’s surface, are constantly mixing it through. But it’s possible that this view is too simplistic.”
Where’s the silicon?
There really is a fundamental flaw in this hypothesis. It is generally agreed that the Earth was formed around 4.5 billion years ago through the accretion of meteorites that emerged from the primordial solar nebula, and as such has the same overall composition of those meteorites. The differentiation of the Earth into core, mantle, and crust happened as part of a second step.
Leaving aside the iron and nickel, which are now part of the planet’s core, it becomes apparent that the mantle should actually contain more silicon than the peridotite rock. Based on these calculations, the mantle should have a magnesium-silicon ratio closer to ~1 rather than ~1.3.
This moves geoscientists to ask the following question: where is the missing silicon? And there is an obvious answer: the Earth’s mantle contains so little silicon because it is in the Earth’s core. But Murakami reaches a different conclusion, namely that the silicon is in the lower mantle. This would mean that the composition of the lower mantle differs from that of the upper mantle.

The two diamonds that compress the specimen are located in the center of this specimen caddy. Tightening the screws generates pressure similar to that found within the lower mantle. Credit: M. Murakami, ETH Zurich
Winding hypothesis
Murakami’s hypothesis takes a few twists and turns: First, we already know precisely how fast seismic waves travel through the mantle. Second, lab experiments show that the lower mantle is made mostly of the siliceous mineral bridgmanite and the magnesium-rich mineral ferropericlase. Third, we know that the speed the seismic waves travel depends on the elasticity of the minerals that make up the rock. So if the elastic properties of the two minerals are known, it is possible to calculate the proportions of each mineral required to correlate with the observed speed of the seismic waves. It is then possible to derive what the chemical composition of the lower mantle must be.
While the elastic properties of ferropericlase are known, those of bridgmanite are as yet not. This is because this mineral’s elasticity depends greatly on its chemical composition; more specifically, it varies according to how much iron the bridgmanite contains.

During the experiment, the rock specimens are clamped between two diamond tips. The diamond tips measure approx. 0.1 mm. Credit: M. Murakami, ETH Zurich
Time-consuming measurements
In his lab, Murakami and his team have now conducted high-pressure tests on this mineral and experimented with different compositions. The researchers began by clamping a small specimen between two diamond tips and using a special device to press them together. This subjected the specimen to extremely high pressure, similar to that found in the lower mantle.
Silicon discovered
Murakami then used the measurement values to model the composition that best correlates with the dispersal of seismic waves. The results confirm his theory that the composition of the lower mantle differs from that of the upper mantle. “We estimate that bridgmanite makes up 88 to 93 percent of the lower mantle,” Murakami says, “which gives this region a magnesium-silicon ratio of approximately 1.1.” Murakami’s hypothesis solves the mystery of the missing silicon.
But his findings raise new questions. We know for instance that within certain subduction zones, the Earth’s crust gets pushed deep into the mantle – sometimes even as far as the border to the core. This means that the upper and lower mantles are actually not hermetically separated entities. How the two areas interact and exactly how the dynamics of the Earth’s interior work to produce chemically different regions of mantle remains to be seen.
Reference: “Experimental evidence for silica-enriched Earth’s lower mantle with ferrous iron dominant bridgmanite” by Izumi Mashino, Motohiko Murakami, Nobuyoshi Miyajima and Sylvain Petitgirard, 22 October 2020, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.1917096117







God poured water on a burnt out star to create Earth and other planets.
” … this view is *too* simplistic.”
Is there are correct amount of “simplistic”?
No, there is not. Something that’s simplistic is too simple.
No, there isn’t.
How could the minerals not separate into different regions? As the molten material flows away to colder regions the pressure differences would separate the elements as they cooled at different rates.
Oh ok then. Mmm.