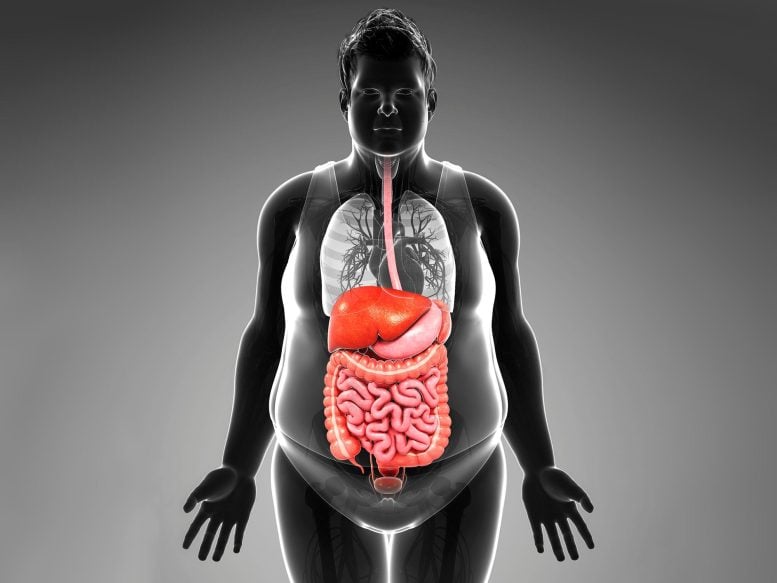
Obesity’s detrimental health effects, such as hypertension and diabetes, stem not merely from an excess of fat, but rather from the loss of fat’s plasticity — its ability to respond to changes — according to a review published in the Cell journal. The researchers explain that fat, besides storing energy, plays crucial roles in immune response, insulin sensitivity regulation, and body temperature maintenance. As fat loses its plasticity due to aging and obesity, it can’t respond to bodily cues, leading to insulin resistance, inflammation, and cell death due to the fast growth of adipose tissue that outstrips its blood supply.
A review in the Cell journal reveals that the negative health impacts of obesity are not merely due to excess fat, but rather a loss of fat’s plasticity, its ability to respond to changes. This decline leads to health issues such as insulin resistance and inflammation. The researchers highlight the potential of altering fat tissue phenotypes for therapeutic benefits, pointing to an exciting avenue for future medical advancements.
Obesity is known to cause cardiometabolic diseases like hypertension and diabetes but attributing these diseases to merely an overabundance of fat is a simplification. On a basic level, fat acts as a receptacle to store energy, but upon a closer look it is an essential actor in vital bodily processes like the immune response, the regulation of insulin sensitivity, and maintenance of body temperature. In a review published in the journal Cell on February 3rd, 2022, researchers argue that the negative health effects of obesity stem not simply from an excess of fat but from the decline in its ability to respond to changes, or in other words, its plasticity.
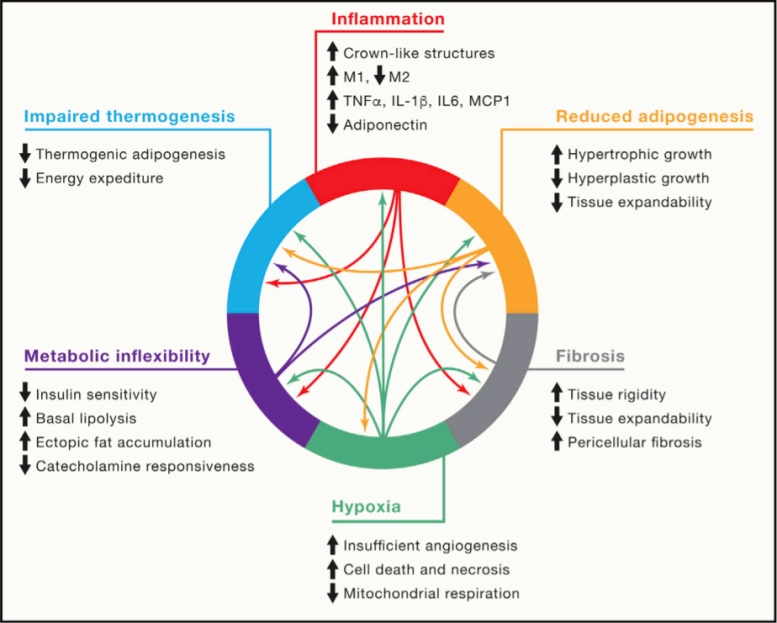
The makeup and functioning of this tissue changes in response to weight fluctuations and aging. As fat declines in plasticity due to aging and obesity, it loses its ability to respond to bodily cues. In the current model of this phenomenon, the rapid growth of adipose tissue outpaces its blood supply, depriving the fat cells of oxygen and causing the accumulation of cells that no longer divide. This leads to insulin resistance, inflammation, and cell death accompanied by the uncontrolled spill of lipids from these cells.
“The central role of adipose tissue dysfunction in disease and the incredible plasticity of fat tissue supports the promise of modulating fat tissue phenotypes for therapeutic purposes,” write the authors, led by Claudio J. Villanueva from the College of Life Sciences/David Geffen School of Medicine and Patrick Seale from Perelman School of Medicine at the University of Pennsylvania. “Many questions and opportunities for future discovery remain, which will yield new insights into adipose tissue biology and hopefully lead to improved therapies for human disease.”
Reference: “Adipose tissue plasticity in health and disease” by Alexander Sakers, Mirian Krystel De Siqueira, Patrick Seale and Claudio J. Villanueva, 3 February 2022, Cell.
DOI: 10.1016/j.cell.2021.12.016
Research reported in this publication was supported by NIDDK at the National Institutes of Health, the UCLA Life Sciences Fund, and UCLA Graduate Council Diversity Fellowship. The authors declare no competing interests.






a sensitive society will tell itself anything to make fat people feel well about themselves, while neglecting that their bodies might shut down at any moment
Spoken like a typical body shaming bigot. Mounting scientific evidence clearly points to obesity having genetic origin as well as pathogenic causes. An exhaustive study by the World Health Organization concluded obesity isn’t as simple as poor habits. No indigenous animals on the Earth deliberately over except for survival. Even those animals resume their normal eating habits when hyperphagia (gorging)is no longer necessary. I suggest it’s you with the problem and it’s called superiority complex. It’s a psychological disorder where someone is compelled to demean others in order to feel good about themselves and you have it in spades. Seek help.
We shame criminals so that they shape up.
We shame presidents so that they shape up.
We shame other countries so that they shape up.
Shame gets sh*t done. If you really care about human life, you need to speak up. Give them the truth or you are complicit in the silent murder.
We shame criminals so that they shape up. And most reoffend.
We shame presidents so that they shape up. And they never do.
We shame other countries so that they shape up. And it caused WWII among other problems.
We shame mental illnesses. And they commit suicide.
Shame gets sh*t done is a bad excuse for poor behavior not a solution.
Actually, no. That’s extremely ignorant and misinformed. Shame is PROVEN to be ineffective at “getting sh*t” done. There’s an enormous body of research showing that it’s actually counterproductive to change. I can actually prove it. Easily. You are currently being shamed in the comments for being a a**hole. Do you feel motivated to change your veiwpoint and behavior? Because if shaming bad behavior is effective at producing change, right now you should be rethinking your stance on this. So tell me, how effective is shame?
“we” shame people because we are self righteous and passive aggressive, not to to get them to shape up. People shame themselves with their own actions, and society punishes criminals. Twitter bullies and morality enforcers weaponize shame.
Nah… shaming me is just your immature response to seeing something you find offensive. Thing is… I don’t give two craps what you think.
Dan here finished off a whole bag of Chips and a liter of soda while whining about intelligent fit people.
Dan has a point, all you bigots out their need to get off your high horses and stop shaming people for their bad health practices like smoking, heavy drinking, and eating too many calories.
Also some percent, maybe 40%, might be a disposition towards obesity, which means people can’t help it that they eat too much pizza and don’t exercise, and if they can’t help it, why try? True, obesity rates have increased 27-47% worldwide over the past 30 years, and while our genetics don’t mutate that quickly, those people 30+ years ago who were genetically predisposed just didn’t realize it. Now that we are informed, we know it’s not our fault and we can choose our own unhealthy lifestyles. Enough already with medical professionals and society trying to get us healthy, it’s obviously not going to work, we’re also genetically predisposed to resisting critiques, even if it can make us healthier. “Healthy” is probably a made up word without any scientific basis. Mental health is also important, and eating potato chips while binging shows on my couch makes me happy, so leave me alone!!!
Dan, Reality is correct. Anyone can lose weight. Literally anyone. I assume, Dan, you are fat and want to justify it by shaming Reality for speaking the truth? There is a lot of that going around these days. Truth shaming. A lot. But truth is thruth, and you’re not speaking truth. You are enabling…fat behavior. And it has its foundation in marxism (everyone is the SAME-fat people are thin too….whine). No Dan we are NOT like you. We don’t OVEREAT junk food. We don’t overeat. No matter how hard you try to justify your own lack of good eating hygiene, it won’t get you normo-weight. Put down the ho-hos Dan!
Overeat, that is.
While I agree that society is not only turning a blind eye to obesity (ostensibly trying to fight it), it seems in many ways certain powers that be want obesity worse.
However, this article does say that greater fat quantity is bad. The article also claims that greater fat quantity contributes, with age, to a loss of fat plasticity, which compounds the negative effects of having too much fat.
From what I can tell, the article isn’t diminishing the negative effects of fat quantity, it illuminates how fat becomes more dangerous the faster one is.
That same sensitive society probably told you that your reading comprehension is great and that you’re a smart boy while handing you your participation trophy.
Quit body shaming others that isn’t skinny. It is ALSO HEREDITARY! I have a 13yr old daughter, who has had medical problems since birth. She is also boarder line diabetic and takes Metformin. She is over weight ever since she was a toddler and yes she had seen a dietitian for a Cple of years. On her father’s side of the family, every single one of her father’s immediate family is obese and has been for generations,that is including her father. She also has fatty liver disease (found out in an ultrasound they ran on her back during the summer). Her father has heart failure and really high blood pressure (also runs in his family). My 18yr old daughter isn’t as big as her younger sister, she also has medical problems. High Blood pressure, high cholesterol, she doesn’t get periods very often (2-3 times a yr but lasts only 1 day), she has OCD and has been in and out of therapy since she was 8. She was also been verbally abused by her father until she was 14, when I got the courage to leave their dad and move back home. I also went thru the same thing on getting verbally abused by her father also off and on from not long after I gotten married in 2001 and got worse in Feb 2017, when his mom had a major heart attack and passed. He became a heavy drinker and bar hop every weekend and not come home til next morning. He was also cheating on me by a so called “lesbian”(his excuse hanging out with his lesbian friend on Friday and Saturdays). Well… Maybe there are others that is obese and CAN NOT help it. Maybe they went thru a similar situation like mine and my kids. Depression can cause obesity, being hereditary can cause obesity, stress can also cause it. Heart problems runs in BOTH my family’s and my kids dad’s family. So does becoming diabetic (type 2, kids dad, my dad, my oldest brother, kids dad’s mom and grandmother and biological father, 2 of my uncle’s. Type 1 runs in my family also, my mom’s cousin was type 1).
NOT everyone that is obese can NOT help it! Some can yes, but some can not. The same with super skinny ppl.. My late step father in law was type 1 diabetic and he could NOT gain weight at all. He was under 130lbs. I have a friend in MI who can not help being really skinny, she eats all of the time and alot of ppl body shames her cause you can see her ribcage. She can NOT help it(she’s also type 1)…
Quit being a male version of a Karen and stop that body shaming nonsense. As long as the person is happy about their body and tries to take care of their body is all that matters. Your nonsense crap is the reason why ppl ends up having medical issues and mental issues from constantly having others body shame them… ESPECIALLY when a 38yr old ADULT starts body shaming a 13yr old and she’s big herself.
Body shaming = BULLYING
An anecdotal account of “my daughter is obese” or “all the immediate family members are obese” by itself proves nothing. It must be hard with your situation and your daughter, lots of challenges to be sure. But as a whole, the “They can’t help it” examples are very rare and don’t inform the whole.
With personal examples, please also provide their exact daily activity, amount of calories eaten, when they eat, what kind of food, how much sleep they get, and how much water they drink. This is important to make an informed conclusion, and most of the time this is the reason people are overweight, and even in rare cases where there’s an underlying genetic issue, that and their weight can still be managed if these other things are addressed properly with that in mind.
If we begin with, “I can’t help it,” or, “I’ve been to a dietician and I’m still overweight,” then we will quit trying. Will you quit, or will you take what you know and strive to make it better?
I speak from experience also, I too have underlying genetic issues, I too have been to a dietician, but for me, if I’m overweight it’s mostly a combination of these other things, NOT my generltics, only I can’t eat the same things others can … but it’s about what I can do to be healthy based on my body & genetics, not someone else’s
Arrogant prick. Shut it
Good luck getting doxxed.
This is so dumb, can’t believe people still spout this nonsense. Even if obesity is not exacerbated by genes which it in many cases is. The fact that society is set up to make people fat is always completely neglected. There is a reason people weren’t as fat decades ago and it’s not because people nowadays lack willpower or don’t practise self control.
If you REALLY want to stop people from becoming obese, tackle the CAUSE. Want to guess what it is? It’s corporations designing foods that are addictive and bad for our health. Not only that but using smart strategies to advertise said “food” so that people become influenced into buying more of it. It’s all so clearly obvious that that is at heart of the issue but people just don’t see it. Because muh choice and muh freedom. Get the f*ck out of here and fight against the real problem and stop shaming the victims.
Agreed.
So, according to you someone who’s fat because of a pinhead sized tumor on their pituitary gland has only themselves to blame? (The name for such a tumor is called Cushing’s disease)
Is telling a bipolar person they are unfixable helpful?
Is telling a slow person they’ll never get faster helpful?
>Muh genetics tho
A perfect example of when very rare medical problems are given as the reason for, “I can’t help being overweight.”
Let’s not forget that having Cushing’s does not guarantee obesity, it only makes it more of a challenge, AND it can be cured in most.
What’s harder to cure? Unhealthy habits like not eating well and insufficient exercise.
We live in a world where the habits of those with anorexia are given as weight loss tips for those fighting obesity. Science is starting to catch up to the reality that losing weight is a whole lot more than willpower, starvation, and working out until you drop while being ridiculed for “allowing yourself to look like that.” The world is moving on. Maybe you should update your programming before you get left behind.
Fiber.
Turn off AC and heater.
Toss any pills with side effects.
Limit calorie and sucrolose intake.
Eat fish daily.
Worry and thinking hard have been shown to be able to burn off a whole Whopper daily, too. The brain uses calories, too.
You’re a bully and you’re embarrassingly wrong and playing doctor all at the same time. Congratulations.
What a s****y comment,”Reality”. I don’t even think you read the article.
Okay, but what causes obesity? It’s simply an excess of fructose from ingesting an over abundance of addictive sugars and carbs. While food scientists have taken full advantage of this in engineering highly processed and hybridized foods. Though unfortunately, dieting and fitness is way more psychological than anything else.
This is the most uneducated comment on here. Your ignorance is astonishing!
>what is fiber
I get the gist of your remark and agree that rationalizing a problem away for someone to save face is common. That said, however, this is a really interesting article that I will be reading more into. I bet there’s a lot of truth to this and probably, hopefully, this research will lead to good, new strategies to help people with real obesity problems. Please don’t dismiss it as just another excuse people with weight problems use, to do nothing at all about their situation.
More like weak people enabling weak people. It’s the liberal way. Like what they do with drug addicts and how they raise weak dysfunctional children.
Gopher Cakes are for everyone, fren.
Open wide; stuff your face.
There’s always room for moar Gopher Cakes.
https://youtu.be/AHmVcYOqGe0
Rather than talk about the nutrition fed to Americans the previous comment or states an obvious fact. We need to spend more of medicines attention on factory, processed and fast foods as the source of much of obesity.
We put corn subsidies in our soda as a sweetener. This is a problem at the federal level.
The article just tells you the why that having too much fat tissue is bad. Adipose tissue loses its ideal function in the body. It is not an excuse to stay that way. As a matter of fact it gives encouragement to lose weight to be healthy. Thank You.
I had a ruptured brain aneurysm and subarachnoid hemorrhage stroke. I was in ICU and rehab for 6 weeks. While I was in ICU they determined I had sleep apnea and they had to add supplemental oxygen. Not only did my brain blow a gasket, my entire body went to a place of its own. I had a seizure, fell off the porch and crushed my wrist. I have developed cysts on my thyroid gland and while they were doing a CT to find the blockage for my PAD, they found another cyst on my kidney. My veins have collapsed. I can’t walk and breath at the same time. I can’t lay down without my BiPap and oxygen concentrator on. I also can’t talk and breath at the same time. I have a PCP, a neurologist, neurosurgeon, vascular surgeon, pulmonologist and eye, nose and throat specialist. I’ve had 3 rounds of rehab and will be having my 6th brain angioplasty. With all this medical assistance why and how did I get 100 pounds overweight and how do I exercise to lose it when I can’t breath? And how can I walk with a large blockage in my femoral artery? They don’t have an answer… diet pills cause fluctuation of the arteries and the last thing they want to mess with are my arteries. And so I eat eggs and I eat salad. I have an occasional piece of baked fish.. about once every 3 months. So far none of the “experts” have any answers.
My grandmother had similar. I suggest getting off any unnecessary medications, intake fiber (metafucil even), and to walk every day. It will not be easy but it will be worth it.
Wrong again “Fren”.
Look into the Gerson Clinic and Gerson therapy. God bless you. You can reclaim your health. Believe.
Fatness still has little/nothing to do with personal choice and please cite articles about “causal” relationships of obesity to disease as the science typically indicates correlation, not the same. This would be a percentage of populations just as many fat people may have risk so do thin people (hint: everyone based on many factors of health like genetics- not just 1 factor of weight.)
Well I see you don’t belong to the sensitive society…..so what are your suggestions for us FAT people?
Fiber, fiber, fiber
No AC and limited home heating
Daily fish intake
Limit calories and even sucralose
Brain exercise
Daily movement, even driving
And absolutely no antidepressants or birth control of any kind! These do all sorts of damage.
I am so wildly sick the reading medical advice from someone who probably barely graduated high school
Many people can NOT choose not to take birth control or antidepressants. Steroids both inhaled and oral (IM/IV etc as well) also cause weight gain as do antipsychotics. Shall they not take meds either.
Stop sticking your nose where it does not belong.
Reality, did you even read the article?
The bottom line is: lose the fat and gain your life back.
>insert fight club soap joke
No society on earth tracks nutrition and goes to the gym the way this nation does, yet we’re fatter than ever and dying of it. If the adipose tissue is lose its plasticity and function, isn’t it time we look at the chemicals, including vitamin “fortification” that we load into our food? Nobody has the obesity issue we have here.
Nobody else adds HFCS to bread, has free soda refills, waxy Hershey’s chocolate, AC running all day, and doctors getting kickbacks for prescribing all this medication with obesity as a side effect. America is too spoiled and Wall-E was a prophecy.
Gee, where are all the kickbacks? Where do I apply for these kickbacks of which you speak? Because, yeah, no.
But hey, you just keep telling everyone all your outlandish ideas about medicine. Did you get your degree from Trump University or Google U? How embarrassing for you.
What can we do about it?
Turn down the d*mn heater, for one. Activate some of our brown fat in our sleep.
OMG! What are the thin “normal” people gonna do when their perfect bodies give out??!!🤔🤷
Hopefully, by then, we’ll be dead; what will it matter. Until then, we’ll continue to be coveted and feeling great in our clothes. And, why not? We’ve worked hard for it (usually).
So basically the type of fats that are in the food you eat still have an effect on their ability to be useful ?
“Reality” you’re an @sshole.
I hope you continue to study this and figure something out.
The article is very interesting.
I have dieted much and do much work outs and I never lose much.
Thanks
Dump all meds and consume more fiber. It really is that simple.
Superficial article st bedt that totally ignores very complex metabolic processes other than adipose deposition and retention. Loss of function.
Replying to myself. Article ststes that plasticity decreases eith she and therefore does not respond to bidy changes. This totally totally ignores the function of the liver. If you cut caloric intake by 50% for 14 days your liver starts turning fat back into energy for use by your body. Done it.
If reading comprehension is a problem then you should just say so. The article is giving additional reasons why fat is bad 🙄🙄
This was in reply to Reality
I can’t seem to loss weight it won’t come off
To Dan, most people who are fat do not have those kinds of tumors, they are overweight, inactive, and eat terrible food that makes them feel terrible and have no energy. If you really want a narrative that will tell you it’s OK you’re fat, you can’t help it, then no one can convince you otherwise. Why are there not more obese lions and obese woves? Because they eat a species appropriate diet that gives them energy and they burn the calories being active. Obesity has so little to do with genetics it’s ridiculous.
I say try walking in my shoes for 54 years of battling obesity. I was put on a diet at 6 years of age and I will be 60 this year. I have weighed over 650 lbs. I weighed 185 lbs at 13. My thyroid and my metabolism are both messed up dying my whole life and I plateaued at 450 lbs. A year ago I started my current weight journey and I now weigh 176 lbs. I don’t remember weighing this little in my lifetime. I feel like there’s always a monkey on my back trying to get me to 650 again. I have a goal to be 150 and would do anything to get there and stay there. I would be willing to take part in medical trials and or experimental pills. I would try anything to get that monkey off my back. It has never been a lack of willpower but a lifetime of trying not to be fat. Any advice or help would be appreciated. Please help me!
Stop all unnecessary medication. Consume more fiber. Get outside daily.
Dietary fat and carbohydrate (glucose) turned into stored body fat by insulin are two different things.
The jews have been poisoning the goyim for centuries. We put poison in your Wells, threw plague infected bodies into your cities, and now we just feed you processed food filled with industrial waste and artificial estrogen.
We used your worship of “science” to convince you to stop eating meat and eggs and that hydrogenated soy oil was healthier for you than natural butter.
Our word for you means “livestock” for a reason.
Imagine using the comments of a site reporting on advances in science to spew anti-Semitic garbage (and that’s describing your comment EXTREMELY generously). Scientists, engineers, etc. are trying to improve the world, and you just want to regress it to the 40s.
At the end of your life, how will you feel knowing that your only influence in this world was making it worse and that people will remember you around as much as used gum on a sidewalk (and think of you just as negatively)?
How about you do everyone here a favor and preach your lunacy on a street corner instead? Maybe ask some of the gum how it feels while you’re at it.
Soy made me a very sexy femboy. I freaking love science. Between xenoestrogens, soy, and neoteny… the world is becoming a very beautiful place for us femboys.
A willfully ignorant society is judgemental and tends not to look at what science can tell us. Instead they look at correlation and confuse that with causation which allows for victim blaming rather than working on reasonable solutions which are effective at treating the issue.
Eat fiber alongside quick carbs.
Turn off the heater and AC.
Watch you calorie intake.
Stop all medications with obesity as a side effect.
Obesity solved.
That seems like a non sequitur considering the evidence from this study would imply you can do something by about it.
>sleeping in the cold doesnt burn calories and activate brown fat simultaneously
Exactly
who cares either way if people are fat? if they die from it, they’re the ones dying, not me. i can’t understand interfering with someone else’s life like that. maybe people who care this much need to feel a sense of control over others because they have no sense of control over themselves.
>t. based libertarian.
What does the overall well-being of the society I live in even matter bro?
Ho-lee, give your head a shake bud.
Nice! Now if we can figure out the best method to get adipose tissue to *regain* function we’ll be able to help people. So many people struggle with maintaining their weight dispute diet and exercise, while others don’t seem able to gain any at all.
Brown fat activates when you turn off your heater. It helps save energy, too. Win-win.
This is common sense, not a scientific discovery.
People did not want to be Fat.
Yeah because fat people feel SO good about themselves already 🙄
There’s always someone to turn a scientific finding into an excuse for a political speech, in the foregoing example to condemn a “sensitive society.” Yes, those with unhealthy overweight should know of consequences, but much of the concern with minor deviations from that “ideal” weight is itself unhealthy.
America has men chasing other men the women have become so obese. This is hardly a minor deviation.
Im deveely detaerted
I look forward to my body finally shutting down.
Told my Dr this 25 years ago ! He says you need to loose weight you might feel better if you do ! I have arthritis in everything, all my joints swell up ! and ive got spores sticking out of and into my spine my vertebrate are splintered ive got bad spondolosis that feels like somebodys stabbing me in the spine 24/7 and when i exercise if i move the wrong way it knocks the air out of me ! Ive got shadows on my lungs and it looks like i might have half to 3/4 of a good lung left ! You want me to exercise so i might feel better ! Now 25 years later i dieted and exercised down to the weight he wanted me at ! Im in even worse pain cause im older !im In pain 24/7 cause i might get addicted to pain meds ! News flash im gonna die no matter what ! Most of my friends are dead cause they bought fetanyl pain pills cause they were also in pain 24/7 you would rather they buy fake pain meds that ended up killing them so it makes more sense to force me to go get fetanyl on the streets cause thats what most fake pain pills are now days than to allow me to live a pain free managable life ! Somebody checked their brains at the desk when they got accepted into college and it wasn’t me ! You are all fueling the cartels illegal drug trade and shipping pallets of money to China the world’s 1 producer of fetanyl because your government masters would rather you kill off your patients with fetanyl pills than prescribe them pills of known value and known origins ! That is exactly what you are doing ! You are killing americans to make mexican cartels and chineese labs billions ! You doctors are killing americans by refusing to prescribe opioids and the fda and dea are killing even more by waging war they will never win ! Legalize it all or face 90% of the USA dying from fetanyl ! That is in fact what will happen because at some point in a person’s life if they work a job which entails any real work they will at one point get injured its inevitable you cannot stop it ! My wife worked in the medical field she got flesh eating virus during a surgery she was doing ! It destroyed her kidneys so now she needs a transplant nobody cares shes in pain 24/7 we both gave our lives to saving others now we are trash because we are no longer tax payers ! With obesity the problem isn’t excess fat ! It’s immobility because we move for more than 10 minutes we are in pain and cannot do anymore fools ! I take illegal pain meds wham I loose all that weight like a bodybuilder going from 18 to 22inch biceps in 8 months cause hes spot injecting steroids ! Drugs exist for a reason ! Because they work ! Stop killing people by forcing them to buy fetanyl off the street !
Definitely based.
As my belly gets bigger and bigger, I just tell people, “Like the mighty camel, I’m just storing up fat for that LONG walk across the desert.”
It’s fun to watch all the anorexics come out of the woodwork and flex.
I have fought weight gain for over 40 years. I have been on every diet trend, worked 60+ hours a week at a physical demanding job (not a desk job) I was an industrial machine mechanic and spent 10-12 hours a day walking and on my feet. During this time I was on a low fat, low carb, high protein and sugar free meal plan. I also had a home gym (bowflex) that I used every night 7 nights a week. I lost 15 pounds over a 5 year period. I have been in construction and manual labor 95% of my life and regardless of the activity I could never keep my weight down. I have been told I have thyroid issues but medication would be unsafe due to the fact my thyroid enzymes fluctuate to severe highs and lows. I hate people who think it is about over eating and have no idea. I was on dietician recommend meal plans and activity recommendations on top of working 2 jobs and still retained weight. Stop assuming all people are gluttonous pigs and understand some seriously just have to starve and literally put their lives at risk just to lose weight. I pray they can find a medical reason and reverse it for the sake of those who have issues losing weight (not talking about the creepy people who call themselves FEEDERS for perverted sexual pleasure which is disturbing and disgusting).
Brown fat is why children don’t gain weight. Fiber in soda and banning air conditioning would solve the obesity epidemic instantly.
A lot of people use food to feel good because eating is an activity after all, but what you choose to eat is critical. Either we eat simple sugars that may appeal to us for a minute or we eat something more healthy that will actually make us feel better for hours. As far as exercise goes we need to do things but it can be so many different things. Life is short so we should enjoy whatever it is. I don’t like grinding at the gym. A walk in the park seems much more appealing to me nowadays!
Overall reality is that people who are overweight – especially obese that by simply eating properly & exercise regularly will in the VAST MAJORITY OF CASES will loose weight – quickly – there are rare exceptions & should NOT be considered unless a person has been supervised in diet & activity for at least 30 days with no loss of weight – otherwise in over 98.99% of the cases weight loss always occured with proper diet & activity.
There’s some people on here that don’t like the hard truth. And that its not healthy and a major risk to be fat. Yes genetics play a role in it. Im much more easily addicted to substances than my paternal cousin. So in our lives I had to grapple with and finally fight and win against addiction. While he and I did the same amount of experimentation, nothing seemed to grab him. These people need to understand that the fattest person in the world 100 years ago weighed like 400 lbs. We have like five 400+ pounders at my work alone. Yes our modern society doesn’t play to genetic weakness like substance abuse and weight gain susceptibility. But yelling at people telling you the hard truth that “in this specific battle you are gonna have to try harder than some others or else you will fail” will not help you. I witnessed my uncle die from a life of lethargy, bad eating and major weight issues. And it was the longest most brutal 3 months of blood clots and comas that I ever have seen a family member go through. He was 63
Kris E, you are my hero!! Rock on! I am so proud of you. I have never been at a constant weight my entire life. I am a diet professional – which means, I can take it off, but can’t keep it off. The news flash for everyone here is that, we are all going to die. Tall, short, young, old, black and white. I have known healthy people that drink plenty of water and are physically active, yet die from cancer. Your body is in control – NOT you. You can not prevent or stop cancer, dementia, strokes, and a host of other disease. The Dr’s will tell you – just like with Covid 19 – “we do not have all the answers” Anyone can find themselves havibg to take steroids for a medical condition and start blowing up like a ballon. Take care that you show compassion, as weight, may have never beem your battle. No matter what state you are in enjoy life – you are going to die – that is a fact.
A bigot for being real? Dan, let me help you get your head out of your ass.. Bleeding hearts like you encourage an unnatural way of life and you justify your “logic” by scrutinizing those of us who point you out. You say it’s “fat shaming”, I say it’s reality. Fat people need to make healthier choices and stop making excuses.
I think this info. may help someone someday. Research is good. Keep up the good work!
Getting molested and eating/drinking one’s feelings away is also a good way to get fat. Find a good listener. And your own conception of a God.
Before ‘discussing’ this article, or any other, some of you may want to think about why you are so hostile. Articulating your opinion while demeaning others demonstrates a lack of emotional intelligence and maturity. Are you this unpleasant face to face, or are you simply feeling powerful in your anonymity? In other words, see if you can converse with other humans without being such an @sshole.
So Dan is a whiny fat bastard himself huh?
Don’t blame your genetics bro, get off your ass or put down the potato chips. Not that hard.
morosil.
Don’t diet change your diet
https://youtu.be/LWcsT1RDpSY
Obesity is a result of a lack of willpower.
Here is an easy road map:
1. 25-50% calorie deficit indefinitely
2. Enable thermogenesis (caffeine-no sugar, green tea, certain peppers, etc)
3. Walk (elevation is a plus) for 30-60 mins daily
4.Drink WATER, no more sugar, if you must drink juice, dilute with water
3. Eat lean meat (chicken, turkey, fish)
5. Boost your metabolism: eat fiber, coffee, flaxseeds, lentils, GINGER
No Larry your path is ridiculous you are asking people to suddenly be able to deprive themselves it never last.
Instead just encourage people to add avocados and spinach or Kale to their diet and increase their protein and fiber intake. Adding these things will eventually crowd out their urges.
There are now protein chips, protein donuts, and fiber based oatmeal raisin cookies. Eat as much as you want and eventually the fiber intake, protein, and healthy fat in take just shuts down your other urges
Reality is a arrogant prick. God complex.
Seems like people claim calories like they claim tips. They say they haven’t eaten all day. Didn’t count the 44oz soda twice, the candy bar. You have to burn more than you consume.
First and foremost aging is a process where our body is incapable of burning the amount of energy we use as we were younger. We have different metabolism rate as we grow older. Hormonal changes in our body have a lot to do with weight gain and weight loss. Don’t forget about the stress of life changing in health and disease. I went through chemo treatment for two years and gained 40 lb from the steroids they have put my body through. The destruction of chemotherapy destroyed my entire body, physically,mentally and emotionally.
I have known people who were overweight, I have seen them eat large quantities of food, two, maybe three times what”normal” people eat. I know why they are fat. I eat normally, same meal for breakfast everyone else eats, salads as a meal, avoid sodas for the most part, skip the fries most of the time. I eat fruit often, nothing to axcess, semi low carb. I don’t eat sweets and dessert often. I am still overweight and nothing seems to affect it. I have no mobility issues and am fully functional. I excercise some, need to do more, but nothing seems to work. In many People’s cases, there is more going on then we understand yet
Americans will come up with any excuse to avoid responsibility for their actions. Obesity is caused by over consumption of food that is why bariatric surgery works.
I don’t see why the cause matters; doing your best to management weight is key. We’re all born into some undesirable trait, but that doesn’t preclude us (or someone else, in instances where one is incompetent) from seeking help and USING it. The word “shame” should be banned.
…unless it’s only used to describe capable folks who fail to help themselves.
Actually all the people claiming this is body shaming or that if you are obese it’s just generic and there is nothing you can do about it is so ridiculous.
Anybody on the planet who decides to do the following:
#1 Everday eat more avocados, spinach or kale even if it’s a smoothie
#2 Increase protein intake
#3 5,000-10,000 steps a day
#4 Walk 5,000-10,000 steps
#5 Lower white carb intake
Will lose weight. The spinach and avocado combination causes your brain to fill fuller and it changes your urges over the long term.
Try this and tell me you don’t lose weight and this is even if you continue to eat cheeseburgers and sugar.
Because over time your sugar urges will reduce.
You don’t always have to try to starve yourself you can just add healthier foods. So when it actually is this simple and someone declines to do it then there shouldn’t be such a high level of acceptance.
If you haven’t tried these things then don’t lecture me. Instead try it for 30 days and then tell me it doesn’t work. Except that wont be possible
Lol Dan. What a clown you are. My guess is your wife and daughter are disgusting fat bodies.
Let’s face it, we allow companies to put every chemical known to man in our food. It’s dangerous and often poisonous to some degree yet we still consume it. Coke is a prime example it will eat the paint off of a car but we’re more than happy to drink it by the truck loads and you think that that’s not going to do something to you? Don’t get me wrong I’m as guilty as the next guy of eating all the things with all these chemicals but it does stand to reason that that is going to change how your body functions and destroy a good piece of the mechanisms that make it work properly. So logically anyone with half a brain can see this is the real cause for obesity. Everything is a chain reaction and your body is no different.
Body shaming is wrong. Obesity is usually a medical problem. No history in my family of obesity. My daughter is obese first gastroenterologist told me to starve her. I didn’t agree so brought her to Boston Children’s. She has an auto immune disease (celiac), took her off gluten she lost weight. She was malnourished cause her body couldn’t absorb nutrients. Wasn’t an over eating problem. Maybe educate yourself before judging. A little kindness goes a long way.
Hello… If you all are still taking the advice of a conventional doctor, AMA, and the FDA. You all are suffering because of greed.
I guarantee you ALL are suffering from LEAKY GUT. FOOD INTOLERANCES. MOLD, HEAVY METAL TOXICITY, YEAST are possibilities.
MTHFR…serious
EPIGENETICS… you can not change your DNA. YOU can change your genetic path.
I wish I could speak with all of you. I have experiences that would help.
Research.
Feel better🌻
Everyone needs a “Fren”! Fren’s delivery is brutal AF, but who’d not get off their arse and make changes with Fren as a Life Coach?
…and, nothing Fren said is wrong.
Who was born obese? I’ll wait.
Wow this comment section is a dumpster fire. All they did was report on an interesting study and y’all be making it so personal and political. We all have better things to do.
I have pcwest polycystic ovarian syndrome and it’s hard for person like me to lose weight but unfortunately I had thyroid surgery 10 years ago and I went from 235 lb woman down to under a hundred my diabetes is under control but I still have high cholesterol hypertension and all that other stuff even though I followed the books and rules and I can’t digest food anymore because missing my thyroid glands they proved that with many tests so it doesn’t matter if you’re fat you appreciate that person for who they are they might not can help being fat there is nothing wrong with that as long as they’re healthy and even you skinny b****** that are healthy can’t have underlying conditions so just don’t blame fat people for all we’re lazy where this word at because we’re not we’re wonderful people so stop judging us until you know what’s wrong with us stop putting labels on our asses thank you
It doesn’t matter if you’re skinny or overweight. What matters is if you’re healthy. There are skinny people that are unhealthy and there are obese people that are very healthy and vice versa. We need to quit having this perfect mold of what “normal” is.
And to the prior poster, FYI some babies are born obese. Babies that are more than 8 lb 13 Oz are considered large or obese. That is about 10% of the babies that are born today.
Obesity is a choice, most you people just need to stop being whiny p*ssies I went from 160 lb to 260, because I stopped exercising and increased what I was eating, now that I’m 40 years old I have to fast all day long and only eat once a day so that I can stay around 200 lb otherwise I will blow up to 300 obesity is a choice I do not starve myself billions of people have done day fasting for thousands of years and this is actually a proven practice to defeat obesity so if you don’t even want to try there’s a plenty ways out there to try so just shove it up your butt
PSR suggests pesticides and plastics and other poisons especially when in the womb and first few years life. I like their fact sheets that mention things like dryer sheets, “cleaning” products, popcorn bags, shampoos, HF corn syrup, and tobacco.
Fren,
Are you saying shame is a motivator?
Prisons are full of repeat offenders.
Presidents? Seriously?
Other countries? Again, seriously?
Shame just makes someone feel bad or get defensive. Doesn’t change behavior.
And I also have to say that as of 150 years ago there was no such thing as obesity obesity is a brand new thing in society so therefore is completely controllable you never heard of it in ancient times you don’t hear of it in the Bible going Jesus walk by a fat guy and made him skinny because there was no fat people you need to get this straight it is a new age problem it is a problem and it is completely controllable these are facts
In Chinese medicine, this s nothing new and was well documented thousands years ago.
A decline in general health leads to improper weight. Especially digestive and respiratory health.
Modern diet and lifestyles just help exacerbate this phenomenon.
Living in a society where money is the only thing that matters is shameful. The reason for belly fat is processed foods and throwing sugar in our face. We are a busy society and the media knows it stuffing junk in all our foods causing us to have health problems later. Where’s the healthy food? Media doesn’t care. They just want your money honey. That’s it!
Dear fren
The the only advice you did not give is grow your own food and raise your own meat so you know how it was processed .
This article is very informative. Fat shaming to me is putting someone down that is obese, by calling the fatso, big, etc. Explaining what causes obesity is not. Explaining how bad it gets when we get older is not. We live in the most sensitive era of human beings that I’ve seen in my 46 years here on earth. Stop acting like yal don’t know that politicians get kickbacks for allowing these chemicals in our food. They do it to make food last longer on the shelves so they make more money. Then we get sick so big Pharma now gets paid. It’s all about money! The greed levels are at an all time high. If people stop buying foods with 40 ingredients their health will get better. In families with lower income they buy the crap that is full of junk because the price is right for their budget. Go to Walmart, then go to Jewel, Marianos, or Whole Foods. Totally different products. Walmart carries the crap at a low price while the other stores sell high quality food. I am overweight myself and have learned this info through experience. Now that I read labels and don’t eat at McDonald’s and other fast food restaurants my health is better. My fatty tissue in my liver isn’t increasing. I hope scientists continue to enlighten me and others on the dangers of obesity. As society will have you canceling everybody that speaks truth.
The article and many others is merely to share and show research and discoveries related to obesity. Body weight/size is not a measure of beauty or worth. It is a matter of health. As a person who was 320, now 150, I can attest to the damage it does to the body, both mechanically and physiologically ….until we approach people about health, eliminating all statements referring to any other aspects we will not help others to change. Learning food is fuel, bodies need certain nutrients and in certain balance, is the 1st step. Remember change is hard, will be and should be gradually or you burn out and say screw it
Recognizing and addressing health concerns is not shaming.
People who fixate on anti-shaming narratives tend to frequently disparage those with whom they disagree while failing to define the contours of the shame they allege they or someone else suffered.
People hold preferences to establish direction in life. Parents who prefer their children to enjoy the most physical potential from their young bodies avoid dietary approaches that contribute to obesity. That is not shaming, nor is it shaming to hold that aspiration for one’s progeny.
Thank you for this information. I want to learn the science of how my body works. If there is information that I don’t understand or question, I don’t look at it as shaming
Always a reason…
Calories in vs out
It’s that simple
Stop stuffing the pie hole
The facts: At least 2.8 million people each year die as a result of being overweight or obese.
Obesity is definitely a concern. It is not like most obese people want to be obese. What is needed is a solution not gimics that don’t work and fad diets that get little to no results. I see so many different diet plans out there and tons “miracle pills”. All they do is rip people off. So many have been tried by people who spent their hard earned money only to be scammed. Those need to be stopped. We all know moderation eating and exercise are important but for some that just isn’t enough. We all know that one person who can eat and eat and never gain. There needs to be a real solution with real results that is affordable for everyone. This country allows big pharma to control who can get the help they need and who can’t. I know people who have struggled with their weight all their lives and were never able to lose enough to be considered non obese. Only narrow minded people believe it is due to a lack of self control.
Being fat or obese is extremely unhealthy. Now please tell me how I shamed someone by stating obvious fact you prick.
It’s about common sense , eat a balanced healthy meals and exercise. If you get way overweight that’s on you. You are in control of your body !!!
Since when did shaming help anything? Do you think our criminal shaming has worked? How’s the churches doing with their attendance? Bringing people to the alters in masses because shaming is so succesful? Ludicrous!
Obesity has a myriad of causes and complications…not the least of which is a dopamine deficiency that is rampant. More science, less BS…
I like this article. Some of these comments I could do without.
I believe more people started becoming obese with the over processed fake foods in the ’80s.
High fructose corn syrup. It’s in everything. You put the wrong kind of fuel in a car and it will break down. The lack of exercise is another and lastly the lack of sleep.
I’m not well versed but I think it is a simple fix but would be a costly one. Just think how empty a grocery store would be if you took all the non nutrient foods off the shelves.
Thank you. To all the nasty posts…I am someone who eats right, exercises everyday and can’t lost weight no matter what I do. I also now have severe inflammatory diseases that have impacted me even more so I can’t exercise as much as I would like..so this makes sense. Science will show the truth. Our society is the way it is because of closed minded bitter people.
Based on most of the comments, a lot of people must think the laws of thermodynamics are nonsense
What you all neglected to mention: American people have been poisoned by Elites who GMO our food(not allowed in other Countries) and our water has poison in it,waste items like floride😳and we’re given vaccines practically from birth which contain poison (flu shots alone contain horrific heavy metals)Every other commercial is on drug brought to U.S. by big PHARMA.. They’re killing us..pay better ATTENTION 🎉
I’m sure some will call this fat shaming. I mean an extra couple hundred pounds can’t really be unhealthy can it??
Hey “The Truth”, you’re wearing a MAGA aren’t you? I can tell by the judging you do to people you know nothing about. typical redneck, ignorant republican bulls****. If you and Trump are so smart, why has it taken more than a year to realize you lost the election in 2020. Go back to your hole in the woods and reflect on why you’ll believe that Fox News is an actual news outlet.
Reality, You need to look up fecal transplants. When a slender persons stool transplanted into an obese person’s colon, they lose weight. And when a slender person has the same transplant from an obese person they gain weight. This is about the microbiome.
News flash- Skinny people die too. May wanna eat a few donuts. The sugar may make you happy and less of a nosey, it’s none of your business what strangers do- whole. Enjoy your inevitable death. I’m so glad I’m fat so I can annoy you with my disgusting ways!
Fat is nothing new. Fat people, again, nothing new. If the scientific community would spend as much time and energy on solutions for ending cancer as they do on obesity and erectile dysfunction, maybe – just MAYBE – cancer could be eradicated. Seems reasonable to me.
I saw no shaming in this article only facts telling you too much fat on your body amplifies and causes a cascade of other unwanted ailments. I have been there and it took effort and will to change it but I feel so much better physically now.
Thank you Dan
To all the people that say treating obese people critically is fat-shaming because some people “can’t help it”, I say total bullsh@$. It is a math problem, consume more calories than you burn and you gain weight. Every body uses fuel in the same order… glucose then fat then protein and finally itself. It’s like a campfire. I can put nice dry logs in the fire ring, douse them with gasoline and light it. I’ll get a huge blaze and when the gasoline is consumed the fire goes out with the logs almost untouched. The gasoline is glucose and the logs are protein in that example. If I put dry logs, kindling, paper and gasoline, the gas will catch the kindling and paper which will then catch the logs. Kindling and paper joining this example as fats. So if you have sufficient glucose from eating the wrong foods to meet the fuel needs of your body, you’ll never burn fat and the proteins will also be untouched… and you get fat
Hey Fren
What is brown fat?
Shop in produce and meat department at grocery store and get off of medication that only mask the problem.Also excercise and get off of social media
Thanks reality. I Lol’ed at the fat guy who called you a “body shaming bigot.”
Simple.
Stop eating so much. It’s disgusting
Damn, anything about health really brings out the nutty people. Why do they let people comment under these things? Nothing of value is ever said.
None of these comments have much to do with the article. If what it said is true, carrying excess weight in middle age and beyond wouldn’t necessarily mean a death sentence if the fat cells can, be means of medicine, be returned to the way they were. I hope I’m understanding this correctly. It could be a game changer. Weightloss is great too but no one deserves to die from obesity.
Hey Fren, there are 3 basic body types. All of which are predicted by genetics. GENETICS!!! Can shame change someone genetics make up? I can eat 500 calories a day and stay the exact same weight. My grandmother’s brothers were 6’5 and up. Close to 300 lbs of solid muscle. My genetics come from their mother mostly. She was 5’10 200 lb. Not a fat woman and not a slim woman. I am 5’9 and have always struggled with my weight. My mother’s side of the family have very very plump short women. I got the best of both I guess. I have two sons and my oldest has always been 99th percentile for height and weight his whole life. He was over 10 lb as a baby. Straight out the hospital and a size 2. Not even 14 yet and 6 ft 1 and 215.lb.
His father is 175 and 5’9. Can you tell who’s genetics have played a major role in his body type? Both he and I have 22 inch calves. All muscle. We both also have horrible knee problems and vericose veins along with circulatory problems. Neither one of us can run to save our lives. All from dad’s mom’s mom side of the family. Since childhood. It’s predetermined from the day you’re born and if you think any different you’re just clueless to human geneology. I of course got bullied and so does my kid for something neither one of us can control. The worst thing is watching my son have to go through the same thing that I did with family and friends who don’t understand scientific facts either. Please do yourself and society a favor and educate yourself. It’s ignorant bullies like you who never bother to look at the facts. Ignoring facts causes 99% of the problems in this world. Change. Be a better person.
When in doubt blame genetics. Check.
Know 2 fat people who blame it on insulin resistant but been around these folks THEY EAT too much and too often. And do not exercise.
Ok,so with all that explained. How does a person that has these issues fix them with fixing the gut first? We are in 2022 I think we need to figure what foods can help fixing these diseases instead of putting a blanket over the real issue by pumping one with pills after pills after pills, many many years ago what one could plant that’s what they ate and the animals were not eating food with chemicals and people and animals lived much longer, why? Because the ate the foods God intended for us too eat. Our bodies are not designed for us to only fix our bodies with pills alone the biggest factor is nutrition. What we are eating is killing us little by little …Our bodies are a machine that needs to keep feeding our stem cells,our vessels, and our organs then all things along with meds if need be, our bodies work properly……Nutrition needs to be taught way more in medical school then 4 months. You know thr old saying you are what you eat.
Have you ever met a fat person that runs marathons? Don’t think so. Genetics. Lol.
For 99 percent of people who follow a healthy lifestyle and exercise this isn’t a problem. Our American lifestyle is. We eat to many processed foods and spend to much time behind the computer or TV.
Fern is a bit. Eat more fiber indeed machine, god speed.
Here’s my unbiased opinion…kiss my entire fat ass! I am 50 you skinny hungry bastards need to eat real food and stop smoking that CBD OIL. At no point in my FAT LIFE did you ever consider my weight to be a necessary evil? Hell no…you compare pencil thin people to fat people and then have the audacity to say we’re the ones with a problem? You’re a jealous dumbass! Now wake up and go eat.
I would like to see regulations on some of the garbage that is added to foods that we buy. Why to we need tons of sugars added to foods. Why, so we become addicted and buy more of the same. There is just not one answer to this problem, it is multi level. To much screen time on phones, computers,TV, fast foods etc. Internet as great as it is has been our downfall. Greed has been our downfall. This has all contributed to weight gain. And so much more. And yes there are some genetics involved and diseases. Unless people are willing to cook meals with little processed food this issue is not going away. Remember decades ago it was homecooked meals of meat, vegetables and potatoes or what ever filler was served. Soda was a treat once in awhile. I could go on and on.
I was always fit & active, till I settled down and got married. My wife is an excellent cook, and I got lazy… 250 lbs lazy. I had never dieted before, so I read lots about it. I’m not smart, but it occurred to me that if our ancestors from the distant past stayed trim, I should only eat what they ate.
So, I stopped eating white foods (potatoes, pasta, rice, sugar, breads) and just ate fatty meats, cheese, butter, nuts, seeds, salad greens, eggs, coffee, coconut oil & fat, MCT oil, avocados, Brussels sprouts, and all berries (straw’, blue’, ras’.) I stopped shopping in “the aisles” and only shopped “the outside edges” of the grocery store.
Turns out, without knowing it I was on a peculiar diet they call, “low carb,” or “Atkinson,” or “keto,” or something else (that I can’t remember.)
Anyway, I lost 75 lbs in a year. My friend says it’s because I’ve been eating like a big ape, lol. Maybe he’s right, I don’t know. When asked casually about looking trim again, I just answer, “I cut out white foods, mainly sugars in anything, pasta & bread.)
YER FAT !!!🤣
Time restrictive eating and intermittent fasting every other day helps restore the functionality because people who do this have increased insensitivity and better functioning fat cells
Supertoilet,
Have your daughters been checked for Polycystic Ovary Syndrome? You may want to read about it, or ask your daughters doctor This may give you some answers.
I see where your daughter is taking Metformin, and that’s what is often prescribed for PCOS.
I hope you will you will find this helpful.
Would you goofs stop complaining in the comments about things entirely irrelevant to the article? None of this shame stuff (pro or con) is pertinent to the studies mentioned or the article about them.
I have been up and down the scale due to medical issues and this article pin points the fact that fat can make your body weak ,it is very True for myself when your muscles or lack of muscles are weak you can not move as well or as safe it allows for movement in joints that shouldn’t happen ,so ending up on high end of the scale im weaker and tired more
Have more scrapes , when im lower on the scale it is easier to move about but I still have to exercise to build back muscle that helps tendons to keep things in place and moving well this adding the flexibility we need to move well. To those who have trouble due to medical conditions and want to improve ,my approach is one step at a time each day one more step
Then add on and on and soon who knows where you’ll be .set your own goals wish all the best
Dan, doth protest too much. Eat better and get some exercise. This is the new progressive ideaology: fat is good, fit is bad, cops are bad, and people are born victims or aggressors purely based off biology. Pretty weak and ignorant.
This is a great conversation with many good points. Is it possible to carry on while loving the person who opposes your opinion?
As a fat person, let me say that every doctor i see tell me im fat first thing when talking to me. I ask every one of them to help me with my weight BUT NONE of them will.
But society shames smokers, drinkers, etc in the name of health. Being overweight is very unhealthy, so eat less move more. Maybe try intermittent fasting or keto, worked for me.
There’s a difference between having no solution and just not liking/choosing to implement the solution. Noone is meant to be overweight, it’s a direct result of our diet/ eating patterns
Reality…no one hates my fat as much as I do. It has made my life miserable. I assume you have no excess pounds & I’m happy for you. In today’s judgmental society you fit in! But don’t forget you may drop dead at any moment or you may have a stroke or heart attack & be damaged for the rest of your life.
Can’t we all love one another and have a friendly debate? I can tell some of you are on social media 22 hours a day.
My dad use to say, “All you have to do is watch the History Channel.” to learn how to get skinny.
Whenever a POW camp was liberated by Allied Forces in WWll, every prisoner was super thin. That was because they worked them hard, and feed them little.
A lot of fat people will argue but it’s true.
Obesity is a societal problem. Fast food on every corner. TONS OF COMMERCIALS advertising unhealthy food,limited exercise opportunities, poor health care. NO emphasis on healthy food and drink.
Can we all agree that any food sold on any shelve is ran through the FDA system? Yeah , most of it is. Y’all need to snap out of it, IT’S CALLED SUGAR! SUGAR IS THE PARASITE THAT THRIVES ON OUR IMMUNE SYSTEM. IT MULTIPLIES THE MOMENT IT REACHES THE BACTERIA.,
So why don’t you all mind your own business. Leave people be! Who even cares what your opinions are, I don’t
Cheers
To those with a transgenerational obesity problem.. fats kids are 100% your fault no matter which way you look at it. Knowing you have a higher genetic component to put on and store fat but never decided to eat real food, shame. Diets don’t work, healthy lifestyles do. Learn the difference.
It’s an allergic reaction to whatever additives they put in food nowadays. I’m a decent weight female but I have to work nonstop at it. I’m literally eating nothing but triscuits that taste like hay and drinking nothing but water through the week. I don’t lose weight but don’t gain any either. Maintaining a decent weight is expensive,time consuming and just plain hard. Some times after a bad day I just want a twinkie but that’s an extra ten minutes to my workout routine. I ate the damn frosting off a cupcake once and gained three pounds overnight from it!! If it not food allergy that caused it then I sure want to know what it was!!
🙋♀️ I have been overweight since I was born I lost weight was on yo-yo diets took diet pills anything to try to keep my weight down. 21 I had a set of twins I got back on the diet pills I lost the weight I stayed thin until 10 years later when I had my last child. I had C-sections and I have had other surgeries but I have noticed but then the last 10 years of my life I do not move as much, I do not go as much, this fat does not move at all it stays right in the front and Lord I pray for all of us because it’s hard give us the courage Amen ❤️🙏🙋♀️💯
Stop ! Obese people do not want to be obese from my experience. There are underlying legitimate reasons why people gain weight – medical, emotional, mental, environmental etc. What I do know is that gaining weight has had detrimental effects on my physical, emotional, and mental wellbeing. I did not cause all that led to my weight gain but Im responsible for my weight loss and how I treat people in my life. Stop the hate , people all have to deal with life problems. I plan to get strong to help myself and others.
Cant we all just agree that its the gov and SUGAR manufacturers that is to blame for all of this everybody is so caught up in my side is right and your side is wrong nobody is looking at who put us as people on the mutha f×^kin board wake up
So instead of telling facts as that obesity kills in many ways such as diabetes, heart problems, ect.ect.
We say or do nothing because it’s shaming ?
I have watched a dear friend for 30 yes
Kill herself by eating and drinking tons of soda, oh but it’s diet soda. Ya ok.
I’ve watched her be sick her entire life and reject help from all of us whom love her . When you care about your friends, family, society, you tell the truth .
Your heart cannot take that kind of abuse , fluid forms , always. And carrying that weight is devastating. It kills many people everyday.
So we just sit silent
Because we are accused of FAT SHAMING ?
Think of the children and how it’s a fact
The younger you are
The easier it is to lose weight and be in shape . Get these kids off the computers and get outside and run and play as we use to do .sports helps kids and exercising is the best for anyone .
So no one is shaming
They are concerned about people’s health
And this growing concern we see .
Be there and help people or at least try .Instead of enabling them due to Hurt feelings ..
What happens when the Dr tells you the truth ? My friend changed drs as she was mad ..
How sad …
Not everyone who opens this is a brainiac…Speak English
People who make fun of some one for their appearance are scandalous but it is importtto keep your weight good health is important always inform or be truthful with friends or family members about their weight. And people who lie about that are evil.
Judge not lest you be judged….You don’t have to agree,you have a right to your own opinion,just don’t expect others to agree,but be respectful.I know first hand what it’s like to feel like your body has been highjacked, and no matter what you do to get that termer body back you hit a brick wall. In 2007 I came down with Lyme disease and did not catch it in time and through the years it took control of my whole body and has moved from one organ to another and has effected my whole being, I’ve tried everything I can search to get me back,My metabolism is shot,but I’m working to get it built back up, I’ve watched my fat in take and cooked on the healthy side, yet my insulin just increases its at the point now that I have insulin resistant, and with that push my cholesterol up in to the 300s putting me 80 lbs. Over weight, So it’s not coming from over eating, it’s a condition that I need help with because all I’ve done to help myself just it’s working, If someone can, I’m so in please contact me at
[email protected]
1 pound of fat = about 3500 calories ingested and not burned off. Count calories, make adjustments, and monitor results. It is much easier to watch what you eat than try to burn off the calories or make excuses.
Ha, fat!
Well i went from 290 to 210 by walking an i stop sugar hit my a1c frist year out i dont drink pop only water an a little milk in cearal you give up the sh*t food you can loss it its in you to do it just do it you will see everything eles is bull sh*t do what it take just will power stop eating crap
Those who believe in fat shaming, are the same ones that personally don’t like only skinny people.
They are the ones that say “fat people have no right to be seen at the beach”. And similar insults.
They don’t really care about the health of those they enjoy shaming.
It’s just an excuse.
They used to shame women for having sex outside of wedlock and for having abortions.
Now they promote it, saying “shout your abortion”.
They used to shame people for not getting a job.
Now “the great resignation” is promoted as virtuous.
Whatever is politically correct is what is promoted.
Whatever isn’t is what is shamed.
It’s about politics, not health
If you don’t want to be shamed don’t be grossly overweight. And you know what I mean as there are plenty of fat people not getting shamed. I’m talking wearing big athletic shorts in 20 degree temps because you’re too big for pants or your behind hadn’t seen denim or a no stretch material since your youth.
If you don’t want to be shamed don’t be grossly overweight. And you know what I mean as there are plenty of fat people not getting shamed. I’m talking wearing big athletic shorts in 20 degree temps because you’re too big for pants or your behind hadn’t seen denim or a no stretch material since your youth.
Wow your religion teaches you better you should be ashamed!Judgment will come to you remember one day! Body shaming is no different than racism. You should know that being your culture. I feel sorry for you!
Seems to me comments in reference to this article should be encouraging and helpful. Not a battle ground for opinions on fat people skinny people etc. Adult discussion about the article and helpful ideas would be great.
So I have eaten actually the same thing my BF ate for a whole 2 months and he lost weight and I did not. So you can’t say to eat fiber and cut out sugar. People are made differently. Female and Man are not equal women are scientific proven to carry more fat (aka to carry a baby ect). So don’t you think that Genes also play a part. Those people that say eat less and body shame I bet are white 20-30 year old men/women that Daddy paid for everything for them because they can’t take a sh*t by himself….if they had to survive by by themselves they would just die because Daddy did everything for them got them a job and everything…karma is a b*tch remember that
Obesity is caused by eating more calories than you burn for a prolonged period of time. Yes we can cherry pick infrequent cases where someone has awful genetics but the majority of overweight and obese people just eat too much among other bad habits. It’s not body shaming to call a fat person fat.
Are people really arguing over fat people? Some people can’t help their mass x gravity = bulldozer. Science has proven that chicken + flour + grease = bulldozerass. There is nothing genetic about poor diet decisions or lack of exercise which is the main cause of fat bodies in the US.
If you notice, the same overweight person is in the comments on every thread justifying the over-intake of calories on a daily basis. Don’t blame corporations or anyone else, the fact is we choose what we eat.
If you choose better you’ll feel better, making excuses will keep you in your current belt size.
Just quit being a fat f**k!!!….try a salad for God sakes!!….its not that hard!!
lol so in other words being fat is still bad. All this article is doing is dancing around this established fact while drawing the same conclusion. Fatties are unhealthy.
Maybe if you stopped eating like a pig and exercised (not walked for 30 mins once every 2 days) you would actually lose some weight? The only “genetic” factor in an obese family is the passed down stupidity from parent to child and lack of self respect to allow one’s self to live past 50 and be able to tie their own shoelaces.
And surprise its a loss of function due to fat
I think your write up here left out one very important role that fat cells also play in our physiology. That being their role in cell division which is something I think that people should have some consideration of when they think about what it is to provide yourself with a well balanced diet. And is likely to have a significant part in why you will sometimes hear the phrase “healthy fats”??
When your born into a family who are already overweight, have Thyroid,diabetes, and heart disease, the battles and genetics are real! Now I’m not saying this is an excuse for being fat but when you’ve tried everything under the sun, including several dieticians that say eat this, not that, you get so many mixed emotions when it’s all different information. Everything from pills to powders, exercise and gyms feed off this! It drives a person stir crazy. I wish instead of logistics, genetical info, and money hungry lions taking advantage, someone would say they specialize in caring about making “you” healthy and loosing weight along the way would be a better approach. So do you see counselors, trainers, dr”s, dieticians and Endocrinologists? Yup, done it all! Guess what, I’m a overweight, happy. Fat cat!! And still engage and be and engage into society. I have 2 degrees, substitute teach and a caregiver to many! Good luck to all on your journey.
After reading through these comments , one thing occurred to me there is alot we need to learn about this subject yet from further scientific research which is not the same necessarily as my opinion. One thing that comes from the statistical world and demography. Correlations are not cause but they begin to help direct science research.
Great article. The people leaving comments need to see a shrink.
People MUST be honest about caloric intake. The three meal day plus snacks & dessert is a Western antiquated paradigm. Genetics by themselves will NOT make you FAT. The term “Fat Shaming” is ploy by the Food & Pharma industries b/c over indulging is very profitable
It’s the food and what’s being put into our foods we need to go back to the natural way
If the answer is old fat please explain the phenomenon of the 450 pound 12 year old.
I don’t have time to read through ALL the nasty comments, but what studies have been done about other contributing factors to obesity, such as plastics, chemicals or environmental triggers? It’s true that humans were very rarely fat in previous generations, making genetics the unlikely universal cause. I also know plenty of people (including myself) who follow strict diet and exercise guidelines and keep gaining anyway, so it’s not just about willpower. Something is definitely makes us sick, and fighting with each other over cheeseburgers will not get us any closer to answers!
At the end of the day eating healthy food matters the most whether you are obese or not.
Very interested in this article. Where can I find this study. Everything I read makes perfect good sense. Please disregard the turkeys and ignorants leaving absurd and sad comments.
Expert find that asphyxiation death isn’t cause by lack of oxygen but by the heart not beating…
Really Is stupid
On my, that’s the problem in our world, all the judgement on one another, the separation, the lack of support, lack of love and empathy. Any addiction is accepted more than being over weight, there is sex, drug, shopping, porn, video, internet, tv, addiction and the list goes on and on. Most of them are to cope or avoid feelings.
I have met a lot of so called healthy people who feed their children fast food, sit at Applebees order a salad, down three beers, put their children in the car and drive home. Yes, being overweight has complications that CAN come with it, but unless you are GOD no matter how you have lived your life it’s all in his hands when and how that life ends.
We all have a right to our opinions, but we don’t have a right to think we are better than others.
Oh and yes I am overweight with health issues, but I am also a human being just like YOU.
Articles that are about the medical and overall well being of being overweight is not fat shaming. Fat shaming is making a fun of those by how they look. Society has gotten to the point that they’re offended by anything.
I think people have been interchangeably using the terms “overweight”, “fat”, and obese, which all have different meanings. those who are concerned about being “overweight”…. are you happy? if not then keep trying. those whom are arguing about “fat” …. this is subjective, I can call a 105lb 5ft woman fat because it is my opinion. Obese has medical implications. so most of you sham fools full of shaming shamers should consider much more than attacking opinionated statements as the ones that know won’t help you to know which often perpetuates your idiosyncratic self serving ego boost for the day. obvious unfortunate stories, are just that…unfortunate, genetics should be used in moderation as what evernody considers to be normal within one family may be completely normal in another, thus causing “hereditary” features, this can easily be as simple as not washing your bed sheets weekly . all baby’s are born fat…that’s just how they look lol
What are you people talking about?
Did you even read the article?
Do you see the pictures and you think you understand the article,
Geez..please read before you write a pointless opinion and start a controversy that is isn’t there.
What are you people talking about?
Did you even read the article?
Do you see the pictures and you think you understand the article,
Geez..please read before you write a pointless opinion and start a controversy that isn’t there.
The one thing that I’ve taken from this article for sure is that Dan, is an @sshole…
Still; which one of us isn’t adversly affected by the weekly loss of the art Linkletter and petticoat junction show’s.
Great answer Fran, eat less, drink a lot of water, fast, walk or do Tai chi, stop making excuses, do a little everyday and you will loose weight
If body shaming worked everyone would be skinny. Overweight people know they are overweight. If dieting worked we would not have an obesity issue. Obesity is a complex issue which has lots of causes physical, economic, ethnic, genetic, emotional and social. Bodies developed to hold on to and store fat. Cutting calories and increasing exercise are no guarantee for weight loss. It is a chronic problem which needs to be addressed with gradual changes of life style. And the American “ideal” of the skinny body is not healthy, realistic or attainable. Bodies can be healthy by making gradual changes. And kind support from others.
Don’t eat and lose weight. Simple. Takes discipline which is what fat people lack.
My cousin was living with his girlfriend who weighed 615 lbs. One day, he came home and found her dead on the living room sofa. She was 59. They had to take the front door off the hinges to remove her body from the home. She wouldn’t fit into the hearse, so the funeral home had to send out for a box truck. Ask yourself if this is how you want to go out
@Stormy Look into the Gerson clinic.
The leading cause of most diseases are grains and sugars.
Vegetables are extremely important and they actually cure most diseases because of their vitamin content and the fact that they contain living cells which replace dying cells, and and their alkaline forming and hydrating cleansing effects.
Yes fiber is important, but its even more important what kind of fiber we consume. If its grain fiber, it causes lots of issues where as veggy fibers heal.
And yes anything that is chemical based is a bad idea.
All natural diets provide tons of energy, and keep us more naturally active and bustling around.
Natural fats and lipids like avocados and olives lubricate our systems and are good for us just like oil for the tin man, while refined sugars and simple carbs are what cause weight gain.
Most people over exhert them selves in exercise, which makes them actually keep their weight cuz they insight their stress response.
Mixing bursts of muscle building cardio 15 min a day is better then running for an hour.
For those of you who say that shaming people get results obviously have no idea what kind of damage that can do to a person. It isn’t always over eating. There are health problems that can prevent people from losing weight especially if it is a hormone imbalance or even a tumor. There is a long list of reasons as to why a person is obese. Unless if you have walked in their shoes and lived in their body then you should probably keep your negative opinions to yourself. Maybe the best way to handle it is to be supportive and not negative. Shaming someone for their weight is cruel.
A hormone imbalance!? Orrrr a tumor???! Haaaah that is so funny. Like they all have one of those. A hormone
Imbalance or tumor! Lol. Ummmm, NO
Plain English please?
For the lady on here saying that her daughter has been fat since she was a toddler because her father’s family members are fat you decided to have kids with this man knowing the risks but now you want to blame his family.
You can’t talk about the problem of obesity without being accused of fat shaming. My state has one of the highest obesity rates in the country which makes it also one of the fattest places in the world. The US in general has extremely high obesity rates making us one of the heaviest countries in the world. It was not always like that but people are less active and eat too much junk, consume too much sugar and other empty calories. I have a gut myself, not as big as it was before I was diagnosed with diabetes but I’m still a little overweight. I’m not tryouts g to far shame people but we must acknowledge that we have a problem and try to correct it even if it hurts some peoples tender feelings. Covid has torn my state up in large part because we are so overweight and because conditions like diabetes and heart problems an so on that go hand in hand with obesity are so common here. But you can’t bring it up or you’re fat shaming, now pour me another glass of sweet tea and it better be thick like syrup with all the sugar in it. Then we’ll go get McDonalds and maybe stop at Starbucks for a 900 calorie coffee drink.
Despite how other people are interpreting this particular article, the information I received from it is important to me.
I understand that body fat is not the best thing for anybody. I understand that body fat does play an important role in the human body – when the body is healthy. Comprehending how body fat functions is essential to understanding its role.
When obesity occurs, it definitely does contribute to certain types of disease, which in turn make obesity even more of a problem. The flip side of this coin is that when fats are consumed in the diet, these lipids are NOT the same thing as body fat. This is where people get the wrong idea.
While a small amount of dietary lipids are essential to healthy bodily function – an unhealthy body requires even fewer dietary lipids. So, when a person is obese, they require fewer dietary lipids along with muscle use in the form of exercise, which could be as simple as going for a daily walk.
This article provides some basic information that has garnered interest in my brain and this has resulted in my desire to research this subject even further. This is the primary reason articles of this type are written.
Also, from the comments, I have developed a strong repulsion to anyone who believes that shame is a good idea in any situation. Why do something that has proven itself to be counterproductive? It amazes me that people are comfortable spewing shame. Just. Why?
Wow. Has anyone studied the effects of reading the comments sections of ANY ARTICLE on the prevalence of stress and its pals, raised blood pressure and stress eating? It would be so nice to see a thoughtful conversation. I will now go thank my book club, and try to resist eating a handful of Wilbur Buds (the best chocolate) by going for a walk. Sign me “Not skinny, not obese, a little fat, over 65, out of estrogen and low on thyroid hormone”. We all have our problems. Be happy nobody in this thread is starving to death. Geez.
Reality, only posted a premise which could be true in many cases. As far genes go, yes that might be true but the mind should be more powerful; keeping a good diet will drop the weight, feeling happy about eating the right things is something that takes practice and patience…again Molding the Brain.
Okay, I read some of these. And please guys! We have always known what causes people to be fat. Overweight. Obese. I do appreciate the conversation. It does expose the underline narratives the Marxists are trying to put forth in every aspect of our lives. There are no differences, we are all the same. No, we are all only all the same in the sense that we are all human beings. And there’s a large variety of us. And we all know that. Some are fat. Not because of a hormone imbalance, or because they have a tumor, those CAN happen but are rarities. People are fat because they eat too much for their activity level. And if someone gets so heavy they look like a ball, It is pretty odd. And yes we will stare. Because it’s such an extreme neglect and abuse of the human body. We truly can’t imagine how they can let themselves go like that. Just eat nonstop? Maybe they do have a real illness. But come on people, that is rare indeed. And NOT the reason for most of the obesity we see. Most of those extremely, disgustingly fat people are just over-indulging. And it is as simple as that. Gluttony used to be a sin. One of the 7 deadly sins. For a good reason. Gluttony destroys people. Kills them. It’s about time the time we take it seriously again.
When I was growing up in the 60s I rarely saw people as fat as the ones I see in profusion today. Moreover,what was consudered “fat” at that time would now be considered average. What has changed over this oeriod other than diet, lack of discipline and acceptance? Why is it that I see normal- (yes normal) size children with hugely obese parents? I have to assume their parents started out normal and and year after year of first 10, then 20, then 30 poubd weight gains led them to be immensely fat. Why is it that I go to any European or Asian city and find that the people I pass on the street are not fat the way their American counterparts are?
If obesity was only due to eating unhealthy or eating to much then this wouldn’t be possible. I am very skinny. I always have been very skinny. However my son has been big since the day he was born. We basically ate the same foods for 16 years until he started working so he didnt eat at home all the time.
But im still skinny and he is overweight with us both eating the same foods. His brother has some extra weight now that he doesn’t live at home but he didnt have any extra weight when he lived at home. So thats 2 skinny people and 1 overweight person all eating the same food and amounts of food. Infact I probably ate more than they did. But im still skinny and he still struggles with his weight constantly.
This proves it isn’t what we eat or even how much. Uts something else in our genetics or something that causes it. Thats the ONLY WAY to explain this situation.
Oh just to let yall know my sons thyroid has been check many times and its fine. Also my thyroid has also been checked and its fine.
Is there anything that can help one lose some degree of weight, even if it’s a slow process? The problem I have with psoriatic arthritis is the incredible pain that never stops. Tylenol does nothing for it and I really need to walk. I love to walk, but my body dorsn’t allow for mich of it without paying me back. My doctor doesn’t have any answers…just do the best you can. Unfortunayely, any weight-bearing exercize is excruciating.
Not everyone can afford a gym membership or getting exercise equipment.
Survival of the fittest . Only the strong survive . Call it what you want . If your obese, get some medical help, push the chair back from the buffet. Take a walk . Proven fact , slim people live longer . If you have no self esteem, just keep pounding the donuts.
My comment is that why is that people tend to attack the obesity person because of high blood pressure or diabetes why it can’t be because they are kidney failure or any other symptoms because it is hard when you are in these people shoes trying to lose weight and maintain a positive impact and weight loss
Change is required rather than shame. Obesity is a health issue and can be prevented with better food choices and moderate exercise. We should never accept or encourage what we know to be bad for us. We need better choices when it comes to food and encouragement to be healther. We don’t need name calling.
Why be so quick on you hateful comments on this report? Keep your negativity to yourself.
How can you be so critical of as study that was done to prevent and restore health for people that have a problem? Would you have told Dr. Salk and Dr. Sabinthe same when he was finding his vaccine?
I was looking at a group photo of my 110 classmates from 1965. Maybe one or two students were overweight. Not a lot. Different story now.
In the August 2019 of Good Housekeeping, it states 1) fat cells serve as hormone command centers. 2) They can suffocate if they get too big and oxygen can’t get in and causes them that something is very wrong and starts releasing cytokines. and 3) Once you’re an adult, you almost always have a fixed number of fat cells. Childhood obesity tees you up for weight struggles as an adult.
Mmm…I love seeing all the comments that led to my mother putting an extremely active and happy 13 year old me on a strict 700 calorie a day starvation diet for over a year desperately trying to turn 250lb me into a 130lb me so that my bullies would stop bullying (news flash: they didn’t) and my mom’s side of the family would stop calling her a bad mother and saying “she would be so pretty if she just lost the weight..” despite the fact that I hit all the same landmarks every single woman in my dad’s side hit. Like a sudden intense weight gain at the onset of puberty with no changes to diet or activity level.
Did so much damage to my mind and body that I am now nearly 400, severely depressed, suffering over whelming anxiety, and unable to work. I love having had my whole life obliterated by shamers.
I love having doctors actively harming me by attributing everything to my weight and pushing severe body deformity onto me every time I seek medical attention…because obviously I should allow them to permanantly mutilate me for a CHANCE to lose weight.
I love having a broken tailbone and a lifetime of pain and misery because when I complained of severe pain in my ass they x-ray’d my lower back and said “It is because you are fat and sitting too long.” No doc it is because my tail bone snapped and got shoved the wrong way and healed that way because when I said “I don’t remember falling.” You took it as “I didn’t fall” and x-ray’d the wrong damn area.
I also love being tuned out and told my sudden leg weakness and worsening condition was not being used to exercise after my physical therapist STOPPED TREATMENT because my legs were doing things they should not be doing and she wanted it looked into before she continued so I was not permanently injured. I loved demanding he have blood work done so I could prove that my back is potentially Psoriatic Arthritis. I loved watching him roll his eyes and order it, only to come back next appointment like EVERY DOCTOR I have ever been to going “You’re right, I am sending you to a Rhumatologist because it is most likely Psoraisis related.
Screw shamers. If I wasn’t treated worse then a dog my entire life I would probably be fine now doing the job I wanted to do since childhood instead of trying desperately to think of things I can do to support myself without it leading to panic, terror, and suicidal mental breaks.
I don’t care that this is ‘just anecdotal evidence’ either. I really don’t because it is MY reality and that should be good enough to make people think about their own actions and how it affects others!
Oh and yes, every single doctor I have ever had has PRAISED my mother’s actions. “Good! Anything to get the weight off!” And every mental health professional and Dietician has been horrified and emotional distraught because what my mom did was beyond cruel and I defend my mother because she was doing what she thought was best at the time and was desperate to protect me from all the garbage spewing from the mouth of shamers.
I came from a fat mother. I became a fat mother. I have a fat daughter. Until someone lives in this cycle I am sorry but you really don’t get it. Our society loves to help you and has plenty to say about it. Example I went to a doctor for an ear infection and before leaving I had to endure the help of how I was wrong and not handling my tragic weight problem. My ear?
My grandfather was fat(RIP), my dad was fat and lost over 80 pounds(all in his 40s) when the doctor told him he was gonna get diabetes. I was fat and lost 40 pounds when I wasn’t as fit as I wanted to. Calories in, calories out: exercise more and eat less calorie dense food. Notice I didn’t say less food, me and my dad still eat a ton of food but we eat healthy and workout to burn extra calories. I went from a 10:30 mile 2 years ago to a 6:05 now, I plan to run for track and get a sub 5 by senior year. Its doable, you can lose weight, but not if u pretend it’s impossible.
Chubby people give the best hugs! I’ve dated those labeled “overweight”, and I think they wear their weight very well. Fat can be beautiful. Some people are just made bigger, and I’m happy for it!
I was made slim, and I’ve never put much effort into staying this way. I have watched loved ones put loads of effort into losing pounds, and when that amount of time & effort became unmanageable the pounds came right back. I didn’t mind…
The things that we hate in others are just reminding us of what we subconsciously hate about ourselves. I say don’t hate; Ask a chubby person on a date!🤗
Fren…you are an ignorant, pompous, supercilious, self absorbed, self righteous, arrogant ass. You sense of intellectual superiority is only exceeded by the size of the anal sphincter you have your cranium pushed pass and inserted into your fiber filled alimentary canal
“Being fat is beautiful, name a thing you can’t do? Jumping Jack’s, run a mile, Live past 42?”
Good to see new research showing up on this topic.
Calories in, calories out. I used to be pretty fat and could barely run a 10 minute mile. I lost around 40 pounds in 6 months because I knew it was better my health. For a lot of people the problem isn’t they have to big of an appetite, it’s that they chose the wrong foods to satisfy themselves or don’t exercise. for me all I needed was to be more active, I did a lot of running, but other activities can work too, you just have to find what is best for you. We are not presisposed to obesity, it’s just that nowadays foods are wayyy more calorie dense and people are wayyy less active.
Why is this picture Black??
It’s genetics y’all. Everyone in my family is tall and athletic. I’m 6’5″ 185lbs. And have been this size since high school. I’m 52 now and couldn’t gain weight even if I ate at Golden Corral three times a day. Americans waste billions on diets to lose weight and they rarely work. Control what you can to keep healthy. Exercise is beneficial even if you’re fat. Quit drinking, smoking and doing drugs. And most importantly focus on your spiritual and mental well-being.
NO NO NO! You idiots! How many times do we have to tell you that fat IS HEALTHY AND BEAUTIFUL!? 😑😑
Stop telling us bad things about being fat. WE. ARE. HEALTHY. AND. BEAUTIFUL.
My 3XL grandfather suffered from bad knees, constipation, diabetes, senility from obesity (lived to 98).
His wive had a heart attack which is how she found out she was diabetic. She would ask me the same question 4 times within an hour. She had her knees replaced. Their daughters and son all overweight as well affecting cognition.
Myself had afib, congestive heart failure, diabetes, chronic kidney disease, high blood pressure (149/90 mmHg) memory decline, and grumpiness. November 2020 I switched to Whole Food, Plant Based, no salt, no oil, no sugar (WFPB-SOS)lost 50 pounds, reversed my type diabetes, my blood pressure is now 110/70 mmHg. My memory, mood and cognition is as good as 20 years ago. I am 66 now and went back to work last year designing bumper, fascias and grilles.
2/3 of obese people suffer major medical issues from the condition. BMI of 35 and higher were 7 times more likely to be hospitalized from COVID-19. Uncontrolled diabetes with a1c of 8.0 or higher were 14 times more likely to end up in the hospital. My advice is to get your BMI in the 18 to 25 range and definitely under 30. Sumo wrestlers do fairly well with their weight until they retire with lower activity.
Question is are you fat on the inside (fatty liver, interstitial abdominal fat) which is the most dangerous. Table sugar, fruit juice, fructose and alcohol all raise triglyercides abd cause fatty liver (so stop soft drinks including diet, use of sugars and sugar subsitutes, alcohol and enjoy your senior years as well as the early years.
Six million people of every physical and genetic makeup occupied concentration camps in WWII, all on calorie-restricted diets. Not one case of obesity.
According to Cyrus Khambatta and Robbie Barbaro of Mastering Diabetes the insulin resistance is due to intramyocellular lipids blocking insulin from entering into cell. I followed advice from their book and removed meat and processed foods from my diet (including oils) and dropped 50 pounds in 10 months and got off metformin and blood pressure medicine. Many Whole Food, Plant Based doctors, dieticians and nutritionist follow this advice of getting rid of saturated fats from the diet.
Our country is in death spiral. We either change or our country could be just like Rome. All empires must end.
Fren you are an idiot.
Throw away meds and AC?
Hell, don’t stop there. Get rid of anything discovered since the 18th Century and return the actuarial tables back to about a 40 year life expectancy.Everyone will be skinny b/c they are either sick or starving. What a fool…
Wow..I seen more drug addicts the so called skinny cocaine,meth,pilly people drop dead nobody escapes it stop living your skinny lies ..😂🤣😂🌍
Obesity is as simple as poor habits. Which leads to poor health. If you don’t want to be a fat f*ck put down the fork. Absolutely that simple
Well,well,well!! I see after reading through a ton of these comments that 1)quite a few of you must be terribly miserable with your own lives. 2)others really are fat just because they spend all day watching “soaps” and gobbling down ho hos and bon bons.3)there are actually a few level headed comments here. with those 3 realizations in mind I will now add my two cents. Regardless of what you try to say to defend yourself period bottom line unavoidable, unequivocally truthful,and absolute fact is if your here defending fat shaming or any “shaming” whatsoever you are in fact a ignorant turd Regardless if it’s because your miserable in your own life or you just get your (sick/demented) kicks from hurting another human. If your excuse for being obese is “its impossible to lose weight” no matter your reasoning you are a liar because whether it’s lying to yourself therefore lying to others or lying as a defense mechanism it’s still a lie. Anyone can lose weight although it may be extremely difficult for some and to the point that it’s just not worth the trade off to do what it takes to lose the weight.Shaming someone for being illiterate is looked down upon by society as a whole and will get you the “pos” lable instantly although there isn’t any acceptable excuse for being illiterate just like being fat. You can always seek help with literacy. There is literally numbers to call and help on the web(not sure how effective since you can’t read smh) and family members etc. So bottom line the only way a person can remain illiterate through their lives is because they actively “choose” to remain that way. I’ll only use this one example for the sake of time and space. So while you are being scolded by society you are also told what? Your unsolicited negative commentary against this illiterate person is a big part of the reason the person is still illiterate because shaming them makes them scared to reach out because they don’t want to be ridiculed. So with this line of thought think about this, your fat/obese every where you go you get “looks and negative comments” you actually want to go to the gymn but just stepping out of your vehicle at the gymn and walking toward the entrance with your bag you get chuckles,laughs,sneers and looks of disgust before even entering the establishment and can only imagine how bad it would be inside so you get back in your car defeated,sad and guess what “hungry” for food or is it for feeling something other than rejection? You go to the nearest McDonald’s and drown your misery with a big Mac and fries. You go to the grocery store and get the same negative experience with people looking in your cart and you knowing they are searching for the pizza pockets and snack cakes but only finding healthy foods so mumble something about it being the only salad you eat all year.Are you starting to get it yet? When you are openly ridiculed both for being obese and trying to do something about it then you are way less likely to be successful at losing the weight or more often than not you actually get worse because it becomes a mental issue.For all you “tough guys/gals who would retaliate to me with the well they should suck it up let’s be honest you would fold like a chair in the same situation. This is the most ignorant assanine meat crob I’ve heard in my entire life.All you punks are looking for is an excuse to bully. Would any of you walk up to random people and tell them their hair cut looks like complete sh*t? Would any of you walk up to someone built like a MMA fighter just to tell them they are ugly as a horses ass? Let me answer hell no! Do you honestly think their haircut or looks require you to go out of your way to tell them how sh*t they look? Again let me answer hell no! So when you walk up to an obese individual and call them fat ass or sneer and/or laugh what is it that your trying to achieve? Once again let me answer but in parts 1) most often you think because they are fat that they are automatically weak and at the very least slower than you at running and a easy mark to try and bolster your self esteem by ridiculing them like your mother when she accurately screamed that you are worthless and will never amount to anything. 2)You suffer from a mental issue like low self esteem and somehow feel better after (again) insulting a person who a) you think is too weak to pose a threat b)too slow to catch a coward on the run. Then there is 3) You are trying to impress someone i.e. girlfriend/boyfriend or friend and or family. The bottom line is no matter the reason it all comes down to the same shameful cowardly reasons and has absolutely nothing to do with wanting the “best” for anyone and everything to do with your (sick/depraved) needs to feel dominant or better about yourself by bringing down another person
Just like any doctors office that wants ti weight you! It’s degrading and depressing! So this year I’m telling my doctors, if they can’t give me a valid reason for why they need to know my weight,then I’m not getting on the scales! And if they want to put me on BP meds, I’m asking for blood work to check my adrenal gland! People don’t realize some meds cause high BP.
We have to weigh you because that is an important vital sign. Jesus folks. Stop playing doctor. You aren’t good at it.
I’ve made major discoveries checking people’s weight, like heart failure, when I’ve found massive weight gain with no reason for it. If you have high blood pressure or diabetes or just about any medication, we need to know your weight. We may also need to know for dosing medications (not always, but some medications).
If you don’t want to know the number, ask your doctor’s office to weigh you backwards, as we do with our patients with disordered eating. We CAN’T just not weigh you because it’s depressing anymore than we can stop taking someone’s blood pressure because that’s depressing for some.
Regarding adrenal testing…we can’t just do that because you want it. It has to be medically indicated for it to be covered and for the results to be medically significant (due to the potential for any test t have false negatives and false positives).
I know it’s depressing. Most of us are overweight or obese too.
I see a lot of comments, none which point to the more obvious explanations. Obesity is the result of food loaded with sugar and animal protein. Americans eat way more protein than needed, and if not used will be stored as fat. About 45 grams of protein a day is more than enough for a body builder and 1 grand slam from Dennys tops that.. Sugar is unatural, and I’m not talking about fruit, I’m talking about processed sugars. To your liver, alcohol and processed sugar are no different. So you might as well drink instead of eating a sleeve of orios because either way we are doing liver damage, gaining weight, and becoming addicted.. One soda a day will add 17 unuseable very hard to get rid of pounds per year. Don’t expect your main stream health professionals and poloticians to tell you America’s largest cash generators such as sugar, beef, and dairy, are making you sick and now need Surgeon General warning labels too.. Ag laws and anti terrorism laws prevent them legally from doing so. Also don’t wait for your doctor to tell you because it’s practically illegal and not beneficial to the industry to do so until you are sick or diabetic, it’s just not something Doctors are trained to teach you in preventing. Everything legally on this topic is on the consumer as of now until everyone is aware and sick of being sick. It benefits the peddlers of these poisons to keep us dumb and addicted, and yes, animal protein and sugar are more addictive than cocaine. Yet harder to quit because we know about cocaine, its expensive, it’s illegal,, and it’s not in our face every place we go.. Anyone working in health care would be putting themselves out of business if they spoke the truth about how dangerous the American diet of meat and sugar really is because that is what is fueling a very large majority of their revenue and almost all our illness. Our diseases are caused by food that we pay for. Then that disease is treated by the health care we pay for. America makes twice the revenue off of unhealthy food vs healthy food. Every one wins except for the purposely uneducated victims like us who then unwittingly pass this cycle of life to our children. That’s why holidays like Halloween and Easter make me sick anymore because to the kids it’s all about the candy and I don’t want to tell them no when the whole world around them is doing it.. Knowing isn’t easy and many will read this and get angry, deny the facts that are out there and published. That’s how I felt at one time too, however, I should have known then that the only one I could count on to be honest with me is my instincts and they were trying to tell me all along. Now I’m skinny again. Also if you want to loose weight a little quicker, stop eating meat as much as possible or never. Yes, it’s delicious, yes it takes a lot of effort to avoid and work around, but if your overweight you don’t need all that protein. Plants are all you need, they won’t give you diabetes and heart disease, only animal protein will do that. Also anyone who eats daily will not die of a protein deficiency. Have you ever heard of such a thing? It’s actually quite opposite in America.
Fat is an organ just like skin or a kidney. This article addresses its function and disfunction. I see no “fat shaming” here.
You guys have some serious emotional problems. Just want to hear yourself cry. I’m over overweight its good not to just bandaid everything stop being so sensitive 🙃 little pu##ies thats whats wrong with this country. Take responsibility fix your health balance your hormones stop being little snowflakes….. the end
Oh yeah….you sound REALLY healthy. 🙄
If cutting weight is impossible for someone through diet they can always try amputation. I dropped 25lbs instantly and it’s stayed off.
And I no longer eat processed garbage food and lie to myself about everything being not my false. Personal responsibility concerning your health is so last century
Fat placicity, well that says it all there. People need to question what isn’t being said, such as how have the processed foods affected our cells, fat and otherwise. Not just the foods but the packaging. Pretty sure it has much to do with our illnesses and the stubbornness of losing fat. Corporations pay good money to squash studies that show any rhetoric involving lawsuit possibilities.
You dip-sh*t know-nothings. This is a SCIENCE article. It’s not written to make anyone feel better or to negate personal responsibility. It’s meant to inform and educate. There has been growing research over the years indicating that there are a number of reasons why an individual is overweight and has problems losing it. Hormonal imbalances, insulin resistance, genetics. Even someone eating all of the “right” foods might still be missing a key component.
I hope anyone who needs this information is able to ignore the self-righteous d**kheads and use it to their benefit.
Some people on here need to stop playing doctor.
Do NOT stop taking meds without medical approval. If you have a serious medical condition (like myself and many of my own patients), you cannot go off meds.
Science has proven no statistically significant improvement in metabolism with “turning off the heat and AC”. 😂 Wow. Just wow. Likewise, there no consistent data to breaking up meals and eating more frequently. In fact, many overeat doing so and do better with regular meal times.
Many people gain considerable weight on medications.edications which can’t be stopped without serious medical consequences. Many people DO overeat and many Americans in particular overeat due to stress and UNDER sleeping due to being overextended with family and work.
Obesity is multifactorial. There is no one cause and even one obese individual typically has more than one reason they are overweight. Shaming doesn’t help and studies show it is counterproductive.
In medicine, we look for peer reviewed randomized control study data to drive medical decision making. Which is in fact why we have not solved the issue of obesity and overweight in the US. It is a combination of eating too many calories, the wrong kind of calories, public science and health policies that favor subsidy of all the wrong foods and corn sugar, lack of public education in health and diet, decreasing physical activity, increasing rates of disability and poor health both related and unrelated to obesity and overweight, pollution and obesogens… And not even taking into consideration that countries with better public safety nets, paid vacation for workers, public transportation and universal health care have lower rates of obesity.
A certain segment of the population likes to cry about everyone else’s feelings while exhibiting the most embarrassing displays of emotions and feelfeels. If we addressed obesity as the public health crisis it is instead of using it as an opportunity to bully others, we’d all be better off for it.
Go back and read the first comment. I don’t think this is shaming. Regardless of the reasons, overweight people are more prone to serious physical ailments. Not all overweight people are unhealthy, but the propensity is there.If you can’t agree with that, then you very likely believe the Earth is flat, vaccines are for population control, and Trump won the election. Why do so many statements need to attacked as an attack on others. Full disclaimer, I’m 6 ft 3 and 230 lb. I don’t like my weight that high. My knees, my lungs, and my digestion were much happier when I weighed less. One thing worsens the other and it becomes a negative spiral. I believe I can get myself back into shape, but this is an ongoing battle for me as it is for many other people.
Everyone wants to tell everybody else what to do & think. It’s been like that since the origin of Homo sapiens. In fact, it may predate that to pre-humans since chimpanzees can also be pretty nasty animals. We can’t seem to mind our own business. It’s all about who has power & influence since most of us are basically so insecure that we must lord it over others to feel secure & superior. We just can’t “let it be.” Our downfall as a species. Unfortunately,other more noble animals will be dragged down with us when we annihilate each other.
Reality hasn’t ever heard of thyroid disorders or weight gain caused from medicines. Reality has never stayed on a diet for one day. I’ll not be taking weight loss advice from reality. Reality won’t be reading the comments because it’s his choice to remain incredibly ignorant.
Obesity itself is a symptom of underlying disease at times.
To all those folks who struggle with obesity – I feel your pain. To those who have never been obese, count your blessings. The bottom line: If you don’t have anything helpful or nice to say about a very serious health issue, consider saying nothing – especially if you lack perspective and experience regarding the same.
Shame and Blame. Truly American. Speaking of getting fat, anyone remember the Crazy 20’s when they sent us to our room for 2 years? A we get older it’s more difficult to get around, certain injuries from the past crop up and it’s harder to keep weight off. Let alone lose weight.in my 20-50’s I said “I’ll never let myself go!!”. I never anticipated stenosis or spondylitis. Nor bein sent to my room, out of work, for 2 years either. Nor RA. You fat shamers can take a breather. There’s nothing you can say or write that’s half as cruel as what we say to ourselves about ourselves. But bc you’re head is up yourself-righteous arse you won’t hear a word of this. Get over yourself and do something nice for someone today, how about a lift to my PT ? My car died after my Dad died after my 18 y/o best friend, my dog SnoopDogg died. Depression is fattening,too. Judge that. 1 finger pointing out leaves your other 3 pointing back.
By the by, I’m not critiquing the Article, which is 💯 on target. The Arrival did not blame shame or judge. It spoke about additional, scientific information meant to help and guide. I’m critiquing the idiot critics deciding what’s fact and what’s not. If your friend wants to eat herself to death, you can’t change her,foo. Move on, find new friends to mend that broken(seriously?) heart. If it’s a good addiction, go to an OA Meeting and listen to reality. If you are so good at looking good, get away from your toy, hit a club and bore someone else who can look at you with you. Foo don’t know what they don’t know but in this forum,finally an audience! Let out your outrage. Yawn.
Shame and Blame. Truly American. Speaking of getting fat, anyone remember the Crazy 20’s when they sent us to our room for 2 years? A we get older it’s more difficult to get around, certain injuries from the past crop up and it’s harder to keep weight off. Let alone lose weight.in my 20-50’s I said “I’ll never let myself go!!”. I never anticipated stenosis or spondylitis. Nor bein sent to my room, out of work, for 2 years either. Nor RA. You fat shamers can take a breather. There’s nothing you can say or write that’s half as cruel as what we say to ourselves about ourselves. But bc you’re head is up yourself-righteous arse you won’t hear a word of this. Get over yourself and do something nice for someone today, how about a lift to my PT ? My car died after my Dad died after my 18 y/o best friend, my dog SnoopDogg died. Depression is fattening,too. Judge that. 1 finger pointing out leaves your other 3 pointing back.
It all boils down to the many use the true medical problems of the few as cover for a problem that has no medical bearing laziness and indulgence
“Reality” he says lmao. braindead. the only way a fat persons body is shutting down is if they have a bmi of 50+. maybe stop pretending like you’re some genius and read the article and realize its not anything about giving unhealthy people an exscuse. its about how the fat itself in overweight individuals is not the killer. its what the excess fat causes. uneducated skinny guy pretending hes superman bc he was born with a fast metabolism. how about you get off your ass and get some muscle on your body and thn talk. ive been fat, skinny, and now fit. you’ve probably been an unfit lazy mf ur whole life acting like you’re greater thn those that arent as fortunate to be able to get good food to eat that isnt hotpockets and ramen. open your eyes.
As well as some intentions may be on this comment trace instead of fixing everyone else how about we fix ourselves. You do you I’ll do mean and when our time on this rock is done whether we were thin or fat, tall or short, race creed or color what will matter how those we interacted with that are left behind remember us. Will we kind compassionate loving forgiving or we self-centered self-loathing projecting ourselves on other people to make ourselves feel better. there’s all kinds of science behind weight loss and genetics The biggest picture of all is how we treat each other. every culture and faith has its own basis in you reap what you sow, yin and yang, do you want to others as you would want them to do unto you. basically you do you I’ll do me and when I’m gone I hope my children in my friends remember me kindly in loving. I don’t believe if there is a next life it’s not judged on our waistline or bank accounts rather how we treated each other.
It’s hilarious how many lazy fat people here are using every psychologically warped angle that they can muster, to stave off those commenting truth.
If they spent half the energy they are using to do this, on exercise, they’d lose weight and feel free, and better about themselves. Groupthink and Marxism keeps them fat and in denial.
Your diet can turn on or off genetic markers resulting in your health outcome. Exercise also has a profound effect on turning these markers off for obesity through epigenetics. I come from both obese parents and grandparents. My siblings are all obese. I am Vegan and I workout 5 days a week with both weightlifting and cardio. I have 20 percent body fat (considered athlete for woman). I do believe genetically we are predisposed to many things but we also have control over what we can do about it.
I have done some research and it is true that obesity from one or both parents is passed to the born child. Something in the genetics becomes deformed and mutated and it causes a child to have an obesity problem from the day they are born to the rest of their life. The other driver of course is lack of exercise and our poor eating diets. In 1998 my car broke down and so the only thing I can do because I didn’t have the funds to buy now the car was to ride my bike for 6 months in Seattle here to Seattle has several Hills and I had the bike up those Hills I tell you what that was really hard. But within a few months all my body fat had literally disappeared and turned into lean muscle. I felt really really healthy that felt like a spring word it was amazing. Now I’m over 50 and I’m alway obese cuz I don’t exercise. Part of the reason why I’m obese has cuz my age every year the metabolic burn rate drops a little bit. There’s a true story that I heard. A sixteen-year-old kid that is sitting in a chair burns more calories per hour did a four year old that is jogging down a track
Sounds to me like a wana be know it all.
Listen, much of the food we eat are meant to make us fat. Rather, made for shelf life or look pretty & the result is harmful to some more than others. Sadly, no one wants to lobby for a Better quality of food ie; gmo, pesticides hormones you name it. Quit screwing w/the food & the food fed to the cattle & animal meats. If food was not so full of toxins that harm fat cells & metabolism & glucose etc etc those with weight issues would be able to live a healthier life, in turn a thinner less body fat, life.
Enough Already.
Everyone is different, situations are different. I wonder why people are concerned about people who have drug and alcohol addiction. Smoking sessation programs are available that are covered by insurance, and there is no help for the overweight or obese people. Other than very expensive programs that insurance companies don’t cover. Financial situations need to be looked at, families have difficulties buying inexpensive healthy food. Unless you have been there in the poverty level with several children don’t even comment about it. If you think eating canned food from a food bank is healthy. There are so many legitament reasons. I guess why nearly everyone on here uses a fake name so they can hide behind hurtful, shameful or ignorant comments.
@Reality you should be ashamed of yourself for breathing the air other people can use to be healthier. Go take your participation trophy for not being able to spell empathy and hold it over your face till you stop moving. It’s a fun way for you to stay in shape.
Nothing ever justifies shaming or bullying. Science has proven that people are so much more likely to meet their goals in a supportive environment. e.g. a child will just grow to hate reading if laughed at every time they make a mistake.
So just stop being a jerky jerk hater who wants others to fail.
Having said that, do not let our blame focused society give you an excuse not to get better. Whether you truly have special circumstances or not let me ask you this: who holds the power to make a change?
1. Interesting article. I would love to see what more research and practical applications of the research can do.
2. A lot of people commenting here are full of themselves. Wouldnt have commented, but damn…There are a number of reasons people are fat that have nothing to do with inactivity r over eating. Nor does it have anything to do with fiber, ac or heaters. If people turn off ac or heat they can die or become quite ill in many places Fren. Oh, and Fren, some medications cause weight gain but keep people from dying. So stop offering that little nugget too.
Shaming heavy people doesn’t help either. But it tells a lot about fat headed egos and who to avoid. Rather a heavy good person than a fit egotistical idiot.
When Dr’s can figure out hormone imbalances, genetic tendencies, and healthy food becomes more affordable and more available to all, then maybe we will see some more progress.
In the meantime, until you walk in others shoes, don’t be mean.
And maybe all you smarty pants should go back to preschool and learn some more social skills.
People are unhealthy. The fat is a byproduct. It’s not shame. People are literally wearing there health on their body
Their*
Since I had an early hysterectomy, at fifty two i.had a great healthy figure I’m sixty one and am fifty pounds over weight I eat healthy I excersize even more and I still can’t lose weight oh I also took lithium for bipolar so some times the body just doesn’t metabolize food fast enough I also walk my dog every day ride my bike everywhere don’t own a car live in a town house so I walk up and down stairs several times a day I don’t eat junk food never have don’t eat sweets don’t drink pop koolaid I drink one cup of coffee water and two percent milk oj I squeeze but I’m still fifty pounds overweight of course I’m swiss and nezperce five eight I was ran over last April and broke my femur had to have surgery was unable to get around for about three months gained twenty three pounds because I couldn’t do my daily excersize the fat went to my boobs belly and butt it won’t come off I even eat very small meals with low calories it’s my age the meds I took but shame me all you want I’m happy and can kick a guy twenty years younger ass I’m strong healthy have great immune system. Don’t take pills but I smoke pot to help me sleep nights i.havent been to a resteraunt or fast food resteraunt in years I grow my veggies in my garden every year and home can them don’t eat prossessed food so why am I still fifty pounds over weight it doesn’t bother me to much but why does it bother you. I’m still a good person I’m still healthy and funny and happy to bad you all are not by the comments I read you can try to shame me but I have no shame to my game. SINCERLY Laughing Dove.
The real question is…If its not YOUR body, YOUR health, why do you care so much? Ah, not care..As Americans we are incapable of minding our own damn business..
I remember participating in after work activities, not understanding why my body was aching so bad, and HEART kept racing B4 I even got started and remained on fast all night, so much that I couldn’t sleep, yet everyone wanted my life…jus show-up at a physical activity, hardly do anything and continue to loose weight, despite having both height & weight, within portion at the time of my 30yrs of age.
In the meantime, I felt so fatigued & alwayz hungry. I was also feeling Anxious, Confused and out of Sorts. My new normal appearance went from well kept to unkept & lack lusted, behind extremely dry skin & hair that both shedded on command, like a snake. Pretty soon the almentioned and thinner frame, became the source of drug rumors, yet I barely ever took a pill to relieve my monthly cramps.
Fast forward, I began going the other way, gaining 25lbs in one month, while all the other symptoms remained unchanged. Nobody was envying me now, so new rumors of over eating began, jus as my doctor was telling me to halt exercising, because test results showed that my heart could only function for three months at it’s current rythmn.
Topped off with Iodine treatment, I easily put on 100lbs with this thing call Hashimotos… although I got hospitalized, behind the whole ordeal, fat shaming is what it came down to, no matter how much I tried to explain.
We live in a sedentary lifestyle. Processed food is cheaper and faster to make and does have a short term filling sensation. People in general don’t want to be bothered in preparing healthy options. Healthy options that are pre-prepared are more costly. It’s the excuses I’ve heard of. Convenience=unhealthy. Just don’t have the drive to actually get up and move anymore.
So since I became handicapped 3 years ago and as a result, I gained weight since I’m not a active as I once was, now I’m supposed to be shamed for it? Well since this has become a country that’s ‘o so sensitive , do I get to cry and whine too with the rest of the candy asses? Can I cry that I’m being discrimated against? ? Sounds great. Where do I start since I finally have a legitimate bitch? Can I punch out the idiots that hurt my feelings? Will Rev. Jesse Jackson or Rev. AL Sharpton come to my rescue? I think that if people have that much time in their hands that they have to find a new way to make a person or persons miserable then they are neglecting their own imperfections. And why am I handicapped now- because an illegal citizen with no drivers license or insurance was driving drunk and I got hit by their illegal vehicle. They made bond and hauled ass to parts unknown. I not only ended up like Iam now, but the best part of an the money I lost from drs, having to replace my car, and loss of employment. Priorities are definitely screwed up in this county now. Let’s kick a person down and destroy anything positive they had to offer.
I totally agree with a couple of you guys.WE NEED TO REINSTATE SHAME!! IT WAS LOVINGLY GIVEN TO US BY OUR CREATOR.it helps us to get back on track. America has tried to cancel SHAME, and we are just now seeing the obvious results. When I was a child there were very few FatFkrs. Why. SHAME.
Go to YouTube and look up a few who can bring you out of the calorie in/out dark ages. Dr. pradip Jamnadas “Amazing Fasting Benefits” & “Insane Fasting Benefits”, Dr Jason Fung, Sten Elbert, David Sinclair and others intermittent fasting and insulin reality, a hormone that once triggered often turns body into a fat saving haven and then our bodies just don’t get the chance to use the fat. They’ve know for over a 100 years calorie in/out is BS in the long run. Look around the 70’s and at the food pyramid and the statistics show the truth. Stop your insulin spikes then you’ll see change for those of us Insulin Sensitive folks and finally we can see light at the end of the tunnel. Everybody is unique and it’s insanity to think most of the world wants to be fat We evolved through feast and famine and now there’s no famine and those who tell us to follow a flawed system such as the food pyramid that was constructed under false theories of the heart not body composition realities.
I am a 48 year old female with PCOS/stein leventhal disease. I was diagnosed at age 17. I suffered from lack of periods which we thought was from my extreme workout routine. I was a semi professional athlete moving towards being a professional athlete. 5’5” and very muscular. As it turns out my lack of periods was caused by PCOS. String of pearls cysts on both ovaries, high androgen and other markers. As a professional athlete over the years I have had access to Olympic team nutritionists and trainers and each decade despite eating healthy and training up to 7 hours a day I have GAINED WEIGHT. BMI is a terrible marker for muscular people but now at age 48 I am considered clinically obese…and yet I still exercise and lift weights for hours each day and make extremely educated food choices both in glycemic index, fodmap, and portions. I cannot begin to tell you the resentment and I’ll say it, utter hatred, I have for people who make comments like those in this thread about lack of self control, sedentary lifestyle and gluttony or poor diet choices.
Peer pressure, societal pressure and norms a proven methods of behavior modification that governments use successfully all the time without people even noticing it.it is not shaming and it is effective across the board, including in most obese people. The answer is not to simply celebrate obesity and shame those people who care about others enough to not ignore it.
Lose fucion
People keep making poor choices and they end up being fat. I am still the same weight that I was when I left high school and that was over 30 years ago. Stop eating crappy food and start getting active.
I will share my experience. My mom had 6 children. By the time I was 2 she had 5 of us. She started to ask the doctor why I was so fat. All my siblings were normal but I was getting fatter and fatter
The doctor poo-pooed and said it’s babyfat. A 5 year old is not feeding themselves. I was eating same food, same portions but my body was processing it differently.
I grew up in the 70s when Twiggy was the thing. Believe me I know shame intimately. It does NOTHING other than reinforce a horrible self-loathing.
I don’t blame others anymore for being jerks. We are so ignorant about science and biology but pretend like we know. And jerks have been told fat people are just lazy and have no discipline. They believe that.
I hope one day they can figure out to help those like me where it isn’t psychological but until then it will continue to cause psychological harm because of jerks and well intentioned shaming.
so I remember my parents both of them weighed over 300 pounds.They decided to do keto, and yes at first it was hard but it was worth it.my mom I’m so proud of her she dropped over 150 pounds and is in the best shape of her life.my dad is at 230 right now and is still dropping.they do not exercise they just ate healthier nutrient dense foods.now what works for my mom and dad may not work for others that’s the thing,I know countless people that are obese that eat healthy,but don’t work out.some people need to diet some need to exercise,it’s what your body needs.God created this glorious body it is formidable it will tell you what you need that’s how it was designed.now it’s up to you to decide to listen to your body or neglect it.
Now on the other hand there are people that are either born or diagnosed with obesity or other weight disorders that is what’s not there fault.If I get skin cancer because I don’t put sunscreen on that’s my fault.If I get skin cancer genetically,well there’s nothing I can really do about it other than hope that someone figures out how to help