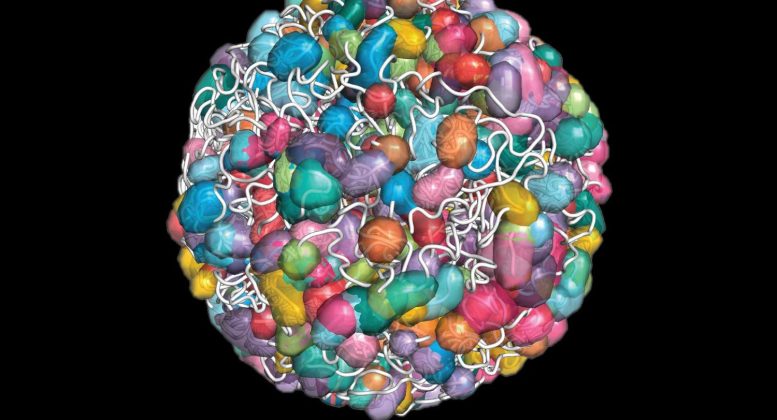
DNA and associated cellular machinery is gathered and organized by certain non-coding RNAs, each region shown in a different colored jellybean shape in this model. Credit: Courtesy of Inna-Marie Strazhnik
The human genome can be thought of as a massive library, containing over 20,000 different “instruction manuals”: your genes. For example, there are genes which contain information to build a brain cell, a skin cell, a white blood cell, and so on. There are even genes that contain information about regulating the genome itself—like books that explain how to organize a library. The ability to regulate gene expression—in other words, the cell’s ability to turn various constellations of genes on or off—is the basis of why different cells (such as muscle cells or brain cells) have different forms and functions.
For any library to be useful to a reader, it needs to be organized in an easily searchable way. For example, all the books pertaining to world history may be on one shelf, whereas the cookbooks may be in an entirely different section of the library. In a cellular nucleus, there is over six feet of genetic material packed into a space 50 times smaller than the width of a human hair. How is the “library” in the nucleus organized? When a cell needs to regulate certain genes, how does the cellular machinery find the right ones among 20,000 others?
A new paper from the laboratory of Mitchell Guttman, professor of biology, uses a powerful new tool that can peer into the world of the cell’s genetic material (DNA and RNA) in order to find answers to these questions.
Led by former Guttman lab graduate student Sofia Quinodoz (PhD ’20)—now a Hanna Gray postdoctoral fellow at Princeton University—the team found that molecules of non-coding RNA are responsible for establishing “compartments” within the nucleus and shepherding in key molecules to precise regions in the genome. Noncoding RNA are molecules that do not encode for proteins, and instead have an array of functions that are often still mysterious to biologists. In the library analogy, non-coding RNA molecules act as the “shelves” that organize different groups of genes and the machinery that interacts with them.
Understanding how genetic material is organized spatially is a crucial part of understanding the basic workings of life. Dysfunction within the nucleus is a hallmark of many diseases, including cancer, neurodegenerative disorders, and others.
The research was made possible by a powerful tool developed in the Guttman laboratory that enables detailed views of the RNA world, called RD-SPRITE (RNA and DNA Split-Pool Recognition of Interactions by Tag Extension). In essence, RD-SPRITE works by tagging molecules of RNA and DNA with minuscule unique barcodes based on their locations; analyzing the barcodes can then tell you which molecules were at which positions within the cell.
“This tool is something I’ve dreamed of since I was a grad student. It’s remarkable that Sofia was able to make this happen,” says Guttman. “It changes what we can look at in the RNA world. It’s like developing a new microscope; you can start looking at things you could never see before. This discovery about RNA and organization is the tip of the iceberg in terms of what we are able to start finding in these data.”
The team plans to use RD-SPRITE to compare the spatial organization of the nucleus between healthy cells and disease cell types, to understand how gene expression and the physical structure of the nucleus may be affected in disease states.
Reference: “RNA promotes the formation of spatial compartments in the nucleus” by Sofia A. Quinodoz, Joanna W. Jachowicz, Prashant Bhat, Noah Ollikainen, Abhik K. Banerjee, Isabel N. Goronzy, Mario R. Blanco, Peter Chovanec, Amy Chow, Yolanda Markaki, Jasmine Thai, Kathrin Plath and Mitchell Guttman, 4 November 2021, Cell.
DOI: 10.1016/j.cell.2021.10.014
The paper is titled “RNA promotes the formation of spatial compartments in the nucleus.” Sofia Quinodoz is the paper’s first author along with co-second authors Caltech postdoctoral scholar Joanna Jachowicz, graduate student Prashant Bhat, and former postdoctoral scholar Noah Ollikainen. In addition to Guttman, other coauthors include former graduate student Abhik Banerjee, graduate student Isabel Goronzy, research scientist Mario Blanco, former postdoctoral scholar Peter Chovanec, senior research scientist Amy Chow, Yolanda Markaki of UCLA, former research technician assistant Jasmine Thai, and Kathrin Plath of UCLA. Funding was provided by the Howard Hughes Medical Institute, the National Science Foundation, the National Institutes of Health, the UCLA-Caltech Medical Scientist Training Program, the American Cancer Society, the Division of Biology and Biological Engineering at Caltech, the National Heart Lung and Blood Institute, and the USC MD/PhD program.






Be the first to comment on "Powerful New Tool Can Peer Into the Vast Genetic Library Inside of Your Cells"