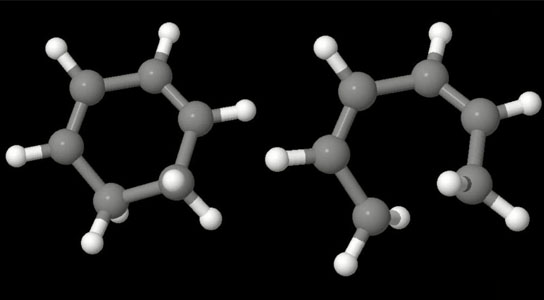
These molecular models illustrate a molecular ring in its intact state (left) and after it is “opened” using an ultraviolet laser pulse. Credit: Illustration adapted from animation by Jaehee Kim
After exciting small, light-sensitive hydrocarbon molecules with light from an ultraviolet laser, Scientists at SLAC’s Linac Coherent Light Source then made quadrillionths-of-a-second snapshots of the molecules at different stages of their transformation with the LCLS X-ray laser, establishing the first use of a “pump-probe” technique that employed X-ray fragmentation to study how molecules respond to light.
In pioneering experiments at SLAC National Accelerator Laboratory, an international team of researchers used light to break open simple ring-structured molecules and explored their transformations using a powerful X-ray laser.
The experiments prove the capabilities of a technique for studying certain types of fast-evolving molecular transformations at SLAC’s Linac Coherent Light Source, the world’s most powerful X-ray free-electron laser.
Those transformations relate to conditions, known as non-Born-Oppenheimer dynamics, in which the motions of atomic centers, or nuclei, are coupled with the motions of their associated electrons. While not fully understood, these dynamics are integral to a broad range of chemical processes.
“It is of fundamental importance to develop new experimental methods that provide further details of these processes, as it helps to make more accurate predictions about chemical transformations in general, from simple molecules to complex biological systems and advanced materials,” said Vladimir S. Petrovic, a postdoctoral researcher in Philip H. Bucksbaum’s lab at the Stanford University Department of Physics, and SLAC’s PULSE Institute for Ultrafast Energy Science.
Petrovic was lead author of a paper detailing the experiments in the June 21 edition of Physical Review Letters.
The group chose to study the transformation of cyclohexadiene, a molecule that plays a role in the creation of vitamin D, because of the way it shifts shape when exposed to ultraviolet light. It has served as a model structure for a range of previous experiments and theories.
Generally, molecular ring-opening experiments are interesting to scientists who seek to better understand and control light-induced processes, including biological processes, and to design molecular-scale switches and memory devices.
The experiments marked the first use of a “pump-probe” technique at the LCLS that employed X-ray fragmentation to study how molecules respond to light. The researchers “pumped” – or excited – small, light-sensitive hydrocarbon molecules with light from an ultraviolet laser, then made quadrillionths-of-a-second snapshots of the molecules at different stages of their transformation with the LCLS X-ray laser.
The X-ray laser pulses fragmented the molecules as they changed shape. By studying the energized fragments, researchers gathered information about molecular geometry and motion and were able to discern the various stages of the shape-changing process.
Importantly, this X-ray fragmentation technique can be used for analyzing a range of molecules, including carbon-containing species.
The synchronization of optical laser and X-ray laser pulses, which this pump-probe technique required, is now commonly used for a range of experiments at LCLS.
LCLS provided a far “cleaner probe” than other research techniques because the X-ray laser did not “induce unwanted, competing processes” that would have clouded the data, Petrovic said.
“The next steps are in the direction of extending the range of useful information that we can infer from these time-resolved X-ray fragmentation experiments,” he said.
Reference: “Transient X-Ray Fragmentation: Probing a Prototypical Photoinduced Ring Opening” by Vladimir S. Petrović et al., 21 June 2012, Physical Review Letters.
DOI: 10.1103/PhysRevLett.108.253006
Researchers from London’s Imperial College and University College, Western Michigan University, Daresbury Laboratory in the United Kingdom, and the University of Michigan also participated in this LCLS experiment.







Be the first to comment on "“Pump-Probe” Technique Explores How Molecules Respond to Light"