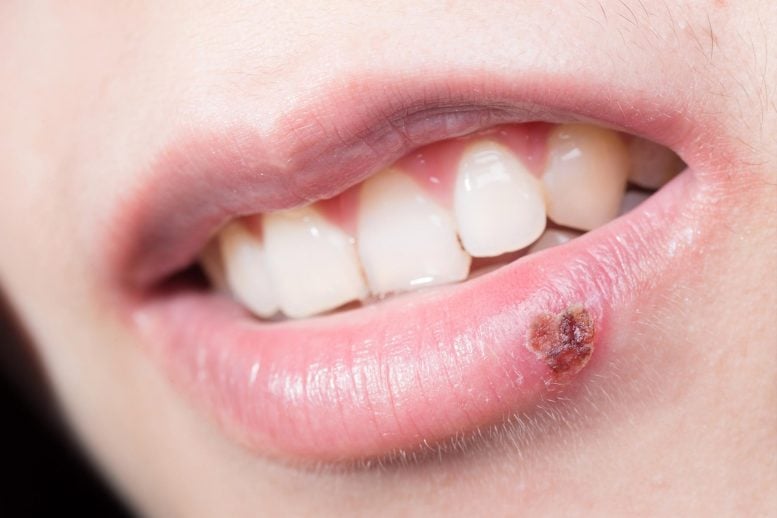
Oral herpes is usually caused by herpes simplex virus type 1 (HSV-1) and can result in cold sores or fever blisters on or around the mouth. However, most people do not have any symptoms.
A new study by researchers at University of Illinois Chicago suggests that when the protein optineurin, or OPTN, is present in cells it restricts the spread of HSV-1, the herpes simplex virus type 1.
In a “first of its kind” study, researchers also found a potential direct connection between neurodegenerative diseases, such as Alzheimer’s disease, amyotrophic lateral sclerosis (ALS), glaucoma, and the herpesvirus, said Dr. Deepak Shukla, the Marion H. Schenk Esq. Professor in Ophthalmology for Research of the Aging Eye, and vice chair for research at UIC.
The research paper, “OPTN is a host intrinsic restriction factor against neuroinvasive HSV-1 infection,” led by Shukla, was published recently in the journal Nature Communications.
Researchers sought to discover why HSV-1 can become fatal for individuals who are immunocompromised but not for healthy individuals. Herpesviruses naturally infect the central nervous system and can result in degenerative brain and eye disorders, as well as encephalitis. However, in most individuals, the virus is suppressed during a primary infection before it can significantly damage the central nervous system.
The new research suggests why HSV-1 is suppressed: OPTN, a conserved autophagy receptor, selectively targets HSV-1 proteins to degradation by autophagy, explained Tejabhiram Yadavalli, a co-author of the study and visiting scholar at UIC’s department of ophthalmology and visual science.
“OPTN stops the virus from growing and it stops it by autophagy — engulfing the virus particles inside tiny vesicles called autophagosomes. The autophagy that happens is very selective. That has meaning for other viruses as well,” Shukla said.
The researchers believe the results from this study will apply to all eight different human herpesviruses.
For the study, mice with removed OPTN genes were infected with ocular HSV-1. The virus growth was much higher in the brains of animals without OPTN, killing local neurons and eventually leading to animal death. This shows there is a faster degeneration of neurons when OPTN is not there. Additional studies are being planned to examine naturally occurring mutations in OPTN, such as the ones reported in glaucoma and ALS patients, and how they may affect neuronal health and HSV-1 infection, Shukla explained.
“Where you have mutated OPTN plus herpes, you have the recipe to create a disaster in terms of neurodegeneration,” Shukla said.
“The study also shows there is an impairment of immune response when there is a deficiency in OPTN. OPTN is needed to signal an influx of proper immune cells at the site of infection. When you don’t have it, you have issues,” said Chandrashekhar Patil, also a co-author of the study and a visiting scholar at UIC’s department of ophthalmology and visual science.
Some of those issues could include neurodegenerative disorders, which researchers believe further research may show.
“We think we will have data to show other viruses, such as Epstein-Barr, Kaposi’s sarcoma, varicella-zoster, are all going to share this mechanism because they share homologous proteins,” Shukla said.
Because the herpesvirus sits in neurons forever, there is speculation it is connected to neurodegenerative diseases. The immune system requires inflammation to constantly fight off the virus, and neurons have some degree of damage because of this continuous immune response, according to Dr. Tibor Valyi-Nagy, professor of pathology, director of neuropathology at UIC and research collaborator on the study.
The study also showed that animals without OPTN and infected with HSV-1 after 30 days lost the ability to recognize objects. Shukla said this could be an indication that having HSV-1 along with a mutation of OPTN could accelerate neuronal damage, which would translate into cognitive impairment.
“Part of our translational research can be how can we correct the problems with OPTN so that we don’t have issues with neurodegeneration,” Shukla said.
Reference: “OPTN is a host intrinsic restriction factor against neuroinvasive HSV-1 infection” by Joshua Ames, Tejabhiram Yadavalli, Rahul Suryawanshi, James Hopkins, Alexander Agelidis, Chandrashekhar Patil, Brian Fredericks, Henry Tseng, Tibor Valyi-Nagy and Deepak Shukla, 13 September 2021, Nature Communications.
DOI: 10.1038/s41467-021-25642-z
Additional authors are Joshua Ames, Rahul Suryawanshi, James Hopkins, Alexander Agelidis, Chandrashekhar Patil and Brian Fredericks, all of UIC, and Henry Tseng of Duke University Medical Center.
This research was supported by the National Institutes of Health and National Eye Institute grants (K08-EY021520-02, RO1 EY029426, P30 EY001792 and RO1 EY024710) as well as the Butner Pioneer Award, Duke Health Scholars and Research to Prevent Blindness unrestricted funds.







I am a 73 year Lady who has had very bad cold sores ever since she was a little girl. One time it was so bad that it went the whole upper lip and up by her nose. I can not remember a summer with not having a cold sore. I use peroxide on the sore and the doctor has given me pills that I take immediately but it still takes 5 to 7 days for that to start to dry up. I can feel them coming on like right away with the thumping feeling in the lip. It would be nice if they could come up with some thing that would dry it up as soon as you can feel that you. Mine would come right after having a high fever or a fever and during the summer if I got sunburn if my lips got sunburn.
Use Carmex ointment,it will clear up faster.
A doctor told me that people that get cold sores are lacking an Amino Acid called
L-Lysine. I take it every day and now it is rare when I get cold sores and when I get them they’re not as bad. You can find it at any drugstore for under $10… Your welcome…;)
I have used Herpecin L since all my adult years.
I have had herpes simplex since I was a young boy around 1962. I am 65 years old now. I got it from a vaccine that was cultured using Rhesus monkey tissue. Unfortunately the vaccine manufacturer did not realize that that Rhesus monkeys carry the herpes virus. I remember that almost everyone that got vaccinated that year in my class started getting cold sores on the lip. This is a lifetime infection. The only thing that works is taking a 1,000mg of l-lysine, an amino acid. You must take it on an empty stomach with water or juice. Do not eat food two hours before taking it, and wait one hour after you take it before eating, otherwise lysine will combine with the other amino acids in the food to combine into protein and won’t be as effective.
You can take it as soon as you feel it coming on, like an itch on the lip. Take at least 1,000mg of lysine 3X/day. The amino acid lysine is harmless and is in most foods we eat. It is a strong anti-viral. It works!
Actually the person who discovered Lysine for cold sores back in the 70s, Dr. Kagan and I have a paper out for Sars-Cov2/Covid and it works extremely well for that as well. What we discovered is the extreme importance of avoiding coffee-caffeine drinks is key along as avoiding arginine rich foods and drinks like nuts, seeds and orange juice.
if you use Duckduckgo or other browsers other than G00gle you can find the paper under L-Lysine therapy for Sars-Cov-2.
L-Lysine by Solgar helps me. It prevents the virus from replicating. I’ve had them since second grade. Suffered with them for years until a new friend told me to try it. It works and can be taken daily. I increase the amount if I feel the tingling.
Try abreva ointment when you feel it coming and apply throughout the day and dont touch it. It will disappear before it leaves a mark
Hello, I’ve had cold sores ever since I can remember. About 2 years ago, I ended up getting very sick. It’s affected my eyes and brain for about two years now. My brain is extremely foggy everyday and my eyes a blurry, I can’t focus on anything without feeling like I’m going to pass out. This has affected my day to day life and have considered suicide because doctors don’t know what’s wrong with me. I found this article extremely interesting. As I truly believe I have this. My throat has also been affected with pain. Sometimes it’s so bad I can’t sleep. I always thought I maybe had acid reflux but i never had this pain until I felt sick in the brain two years ago. If people could please keep coming out with research for this I’d really appreciate it. Maybe someone like me could get cured one day.
QUIT COFFEE, AVOID NUTS AND SEEDS, TAKE L-LYSINE 1000MG HOUR BEFORE A MEAL WITH TWO CUPS OF WATER. YOU WILL FIND THIS TO BE MIRACULOUS.
Ampicllin
Ampicillin
Unfortunately ampicillin will not work for cold sore since it is not an antiviral. Otc products can definitely help numbing the sore ease the symptoms and using things like alum spices can help drying the sore up for faster recovery, but ultimately the root is individual’s immune system strength and avoiding potential exacerbations, such as make sure body getting enough rest, no late night sleeps, proper hydration and nutrition, regular exercise, things like this can help
Crush up an aspirin and mix with water to make a paste and put it on the tingly spot immediately and the sore will not pop up.
Judy, if you put Blistex ointment on the area immediately at the moment you have the first feeling of it, it will stop it in it’s tracks. I used to get them so badly & after many times stopping it with the Blistex over years, they seemed to have stopped wanting to return. Give it a try!!
Really interesting. This could lead to big things for sure. Neurodegenerative disease is probably
among one of the hardest to treat, but with this finding we could potentially have a new mechanism to slow down progression of neurodegenerative disease, keep HSV-1 in remission more effectively, or even develop a cure eventually. really exciting findings they got here!
L-lysine works well.
I used to get cold sores since I was a little girl. In my 20’s I started to notice that they happened more frequently when I was stressed. Of course at the time there was nothing on the market that worked. My cold sores would laugh at OTC products like abreva and blistex. In my 30’s my doctor prescribed a pill (I forget the name though) but it was a life saver! As soon as I felt the tingling I would take one and it stopped it right away. The blister was not able to develop if I took it right away. Anyone reading this can ask their doctor about this pill as I believe it’s the only one on the market (not sure though) but a doctor would be able to know. Also, even though this pill worked on my blister I am always looking for a natural way to heal my body and so a friend recommended using hydrogen peroxide as soon as I feel the tingling. I tried it and it works. The trick is to use the hydrogen peroxide as soon as you feel the tingling. If you don’t use it right away the blister will develop, the hydrogen peroxide still works as it will dry out the blister faster than any OTC product out there, plus it’s way cheaper.
I have also suffered with cold sores my whole life. L-lysine is what I take daily, it’s an amino acid. There was a great article written in the 80’s re L-lysine and how these foods deplete your body of L-lysine…
chocolate, wine, nuts, chick peas and of course the sun. Fish
is an excellent source of L-lysine.
I have also had cold sores since I was a young girl in middle school, going into high school & I’m 30 now. I’ve tried a number of different remedies, including medication prescribed from doctors, in which none of that worked so over time I’ve developed my own remedy that helps me control them much better. Lately I’ve been able to control them to coming very minimal, maybe once a year, which I’m very grateful for. First, I’ve recognized when I usually get them, which is right after I’ve been sick or I’m really stressed out. So to try my best to prevent them, I would take a bunch of vitamin c, in multiple forms, I’d chew like 3 500mg tablets daily, drink liquids with at least 100% vitamin c & now I’ll drink at least 1 emergen-c packet daily. I also put vick’s vapor rub on my lips & nose on top of Vaseline, which is where I normally get them. Lately, the last couple have been on the inside of my nose, which I’m very grateful for because the appearance of them are so embarrassing and depressing. There are times when I’m not able to catch them in time or if they pop up in my sleep, so I’ll make my own remedy, which is using a little Vick’s vapor rub in a separate container that I’ll mix with about 15-20 drops of essential tea tree oil & about 10-15 of essential lemon oil. I’ve also tried using turmeric powder, but it stains really bad. But this definitely helps them to not get as big & bad if you’re able to catch it in time. I’ve spent a numerous years researching how help, & this works for me, a young lady who’s also tried some of everything. Ive also read that taking turmeric helps & if you can detox your body for a good 90 days or so, by completely juicing & use essential oregano oil mixed with a carrier oil & put that under your tongue for about 30 days or so, that this can or will get rid of that virus in your body. However, that has been something I haven’t been able to do as of yet, but I definitely hope to try that to get rid of it completely one day, especially seeing more of how this leads to what this article is explaining. I hope that this can help someone to at least control their outbreaks as well. God bless.
I have had them since I was little girl also…what I have been doing is take a aspirin and crush it up, then add a tiny amount of water to the aspirin..enough to make it like a thick paste. If the cold sore isn’t open ..pop it then apply the aspirin to your cold sore. Do this at night so you have privacy..its not pretty, but it works..keep applying because it will dry up and fall off. Before you know it ..it will be gone. I think the aspirin draws the fever out of it. Good luck, it can’t hurt to try it.😉
Campho- phenique. It is for cold sores. Its in a little tube like carmex. It is the only thing that helps me. I no longer have them in my eyes or nose since I apply this as soon as I feel the tingle. I rarely even get a blister anymore BC I am religiously applying it till the tingle is gone, usually that day.
I use cortisone cream from the dollar tree as soon as I feel one come on as campho phenique used to work, but often they don’t even form with the cortisone cream and I’m 65, had them all my life, even got them genital, so they say even though I’ve never had an outbreak genitally
I use prescribed valacyclovir 1 tablet in the morning and 1 at night for one day only at the first sign of a cold sore. This stops them in their tracks!
Take a L lysine everyday. Avoid citrus, coffee and tomatoes. Foods with high L arginine triggers them. Valtrex and generic equivalent stop em cold. Carmex didn’t do anything for me.
Please get a prescription for Acyclovir from your doctor. You can take this every day or just double the dosage when you can feel an episode coming on. This medicine is also crucial for treating shingles which is the same virus but much more serious. L-lysine has been a tried-and-true amino acid to take for treating herpes simplex but please do research on what you should not be eating and taking and that is Arginine. You would be surprised how many everyday foods have Arginine in them. Arginine can actually cause an outbreak. That’s all folks from my experience!
Mamm google a product called nano soma. Read the testimonies. Easy to use spray. Not much of a taste a little pricey . around 60 for a bottle lasting a month. I believe the results you experience will be something u cant put a price on
I too have had cold sores since I was about 7 and I’m 50 now. I take Valtrex now when I feel them coming on which is very rare now. 10 years ago a nurse told me once that triggers can come from your nose along with your immune system down stress and even the sun. So I started rinsing my nose especially after being exposed to dust pollen pets even after clothes shopping. I use salamis spray by arm and hammer I rarely get them now and I believe has helped tremendously!
I finally found something that works after spending most of my life fighting these sores and usually multiple sores at one time. It is boron. Very simple mineral that has a negative ion charge and it neutralizes the positive ion charge found in viruses. It stops it early and it makes it heal faster if you didn’t stop it early. You can buy it in pill form or drops. I usually get mine on amazon. It has been holding back the Epstein-Barr as well as the herpes for much longer periods Of time.
Uncontrolled viruses such as Herpes is indicative of a pantothenic acid deficiency. Boost your white blood cell count which fights viruses by taking extra pantothenic acid. (Vitamin B5).
Candimom is 100% right Acyclovir my prescription says Valacyclovir. Either way !! Will solve the problem. I’m 58 ever since I was in Elementary, I would get them. Very depressing. I tried everything that everyone has mentioned above! You must get this prescription. As soon as you feel the tingling. Take 1 or even 2 immediately and BOOM. Stopped in it’s tracks! Haven’t had 1 in YEARS since taking this.
Take lysine for cold sores. Clears in a few days.
I too have suffered humiliation from having a lip the size of a melon in high school. Pain and scrutiny. 30 years ago I ran into an article while researching, it talked about raw brocolli stopping the virus in its tracks and suggested at least two handfuls in the morning and evening at the first sign of itching/ tingling. I tried it and it works. Now I try to spread the word because big pharma managed to erase the article. The scientist knew it worked but could not explain it. I even eat plenty before a big event. It has to be raw though, health and peace All.
I’ve gotten cold sores since I was little, my doctor prescribed me Acyclovir which helps. I also put bleach on a Qtip and put it on the spot as soon as I feel a tingle, for about 30 seconds and it prevents the sore from appearing. I know bleach is harmful and isnt good for skin, but it helps me! Hope this helps!
@Alyssa Cordero. Have you been tested for Chiara Malformation. My wife has suffered from many of the symptoms you’ve had since a teenager and finally described her symptoms to a different doctor and was diagnosed and confirmed right away. Something to consider if you haven’t.
You guys, I’m 31 and I’ve gotten these since childhood as well. As an adult, I don’t even have to worry about it anymore because it’s rare that I get one, but if I do, rubbing alcohol saves the day every single time. As soon as you feel a tingle, put some on a cotton swab and press it into the affected area. It’s gonna sting like hell, but just try your best to ONLY put it on the sore spot. Qtip would be good too. Anyway, put the rubbing alcohol on it for the whole day, and it should be gone, or almost completely gone by tomorrow. 70% alcohol is fine. Use Shea butter or something natural (No chapstick– it has drying agents in the ingredients) to moisturize after, and try to minimize the spread of infection by changing the swab often (soon as it dries up), and keeping your fingers clean with the alcohol. For sores in the mouth, this won’t work unless you have it somewhere on the lip. In the mouth, I’d probably opt for an antiseptic wash that has alcohol, like listerine. Just attack it mercilessly with the alcohol, it will really help your body fight and kill the infection. No healing period just back to completely normal in 1-2 days MAX vs 1 week+ with those dumb creams. Also, a daily probiotic has worked wonders for me for the past year, maybe that also helps (strengthens immune system, gut health, and ability to fight infection)
I am 72 now .had horrible cold sores since early childhood .in my thirties I was told by a friend to take L lysine amino acid. I take 2000 to4000 mg. Daily with food and no cold sores. When I got lazy and stopped taking,bamb! Cold sores started up. It’s great. Once get capsules from vitacost .easier to swallow than caplets. Do it!
Infra red lights held a certain distance from your skin I have read can help damage the virus and improve your bodies immune response. Think like the heat lamps at a spa, depending on the wavelength and the distance from your skin it can help, I forgot what I’ve read but there were solid findings with it and the fda approved some kind of silly expensive infra red wand that has very mixed reviews.
Lysine never worked for me, but I was unaware of its bonding with other aminos and foods to produce whole proteins so perhaps that’s why. I’ve used alcohol and peroxide before to dry it up and I’ve used abbreva and acyclovir. The antiviral has some shady side effects I’ve read about before and is the reason I won’t take it unless i have some brutal outbreak that lasts over a week. I usually just deal with it now. But like someone said in the above I’ve had some crazy brain fog plus eye problems the past few years that no one can figure out as well. Perhaps related?
As for the neurodegenative links they have been studied a long time ago. The links aren’t particularly new I’ve read some scientific study about 10 years ago about finding something in Alzheimer’s plaques that was linked to the herpes virus. My own doctor said that its not really worried about in the medical community because it’s considered a “nuisance” disease. It causes discomfort and unsightly sores but because those are the known side effects it’s not really fought against or researched like other infections and std’s. Essentially the money isn’t there to figure it out due to what is widely accepted about it. Most other findings like this study here are coincidental.
Herp it up y’all!
I had a bump seed on my lip for over a year. I often squeeze pimples with little fear of scars. I chalked this bump off as a H1 thus let it be. Feeling how hard it got I successfully pushed it out 100%. I should have donated the seed to science.
72 yrs old. Tried everything mentioned in these posts. CURE: EAT REAL FOOD!!! DON’T TAKE DRUGS/PHARMACEUTICALS.
I have had the herpes complex since I was 28 years I have been taking acyclovir 400 mg every day since no out breaks but maybe two or three times in the past 34 years. It works
Yes, you can stop it all together. If you take high doss L Lysin with distilled water. Along with Oregano oil . Detox your body and eat fresh only organic vegetables and plant based proteins. Roasted nuts can set off an attack. Do your own research. I am not a doctor. This is my experience…
Alissa Cordero . I believe everything have a cure . Learn and read about u síntoms and search for information. First u need to kill parasites in u body . Do a detox ( read search for information) don’t eat meat change u diet , low inflammation…take tumeric, chamomile tea ginger tea, pregnancy vitamins they said are better the regular ones . Loss weight . And pray. Unbalanced of vitamins can be one of the reason. Or a bacteria or parasite behind u eye . Check about probiótics ( they keep gut healthy to prevent toxins going to blood . Try to be lactose free . Read about parasites bacteria’s how are human body works and why probiótics are the solution to prevent any type of illness . And prevent any type of inflammation…..some doctors said is the solution to prevent to be sick and vit c. U can help u self learning. Find a good doctor u must change doctor and tell them u want a complete blood test . Don’t believe in everything they said I was sick one day with a lot of pain per few months . I change the doctor and in few hours I was ok ( the Medicine help me right away) good luck
I’ve had cold sores all my life and I’ll be 60 next month.. But I also have neurodegenerative disease, my eyes are worse every year. My back is so bad, surgery scares me. I pray everyday that the wheel chair keeps it distance and don’t roll any closer. My mother had the same thing. And mom passed at 77, but other health issues also contributed to her passing. Be nice if they had a cure to stop this from getting worse without surgery and surgery may not work any way…
Per Anthony William, Medical Medium” the herpes virus (there are 60 varieties of those suckers) that causes hundreds of diseases (ANY kind of arthritis, migraines, MS, Eppstein-Bar, cancer, nerve issues, on and on. He teaches what anti-viral foods to eat AND what not to eat while you starve the virus. The longer you have this virus, the worse it gets…stage 4 is rheumatoid arthritis, stage 5 is diabetes, stage 6 is alz/mental issues and eventually death. If you have had it for decades, it will take you 3 or 4 years to starve it, have to be patient and it includes fresh celery juice (you have to make because these mineral salts in the fresh celery only live for 24 hours so no commercially made kinds). Healso shares what supplements you need to have as well. Western medicine had always said they do not know where it comes from, there is no such thing as autoimmune folks, it was some darn bug that bit you that had the virus and then gave it to you!
Canabis stops it in its tracks,your welcome
I have been able for some 30 years to have acyclovir 400 mg tablets (generic for Zovirax) prescribed for me to treat (and prevent) cold sores. They work wonders, and I am grateful. I can up the dosage when I have an attack — but that hardly happens anymore. I was advised to always hydrate when taking this drug.
L-Lysine is a good vitamin for Cold Sore to not come around
I have been taking the valtrex or valacycloavir which is the generic form for years now. I don’t have to take one a day, I take a half of one a day and it has kept everything at Bay. If I run out, I will have a terrible, terrible outbreak, but as long as I’m taking a half a pill a day I generally never have an outbreak. I might have a very small one once a year but it only last a day maybe two and it’s very very small. My sister uses the L lysine and she takes the capsule form because the others don’t dissolve as well. She starts taking it at the first sign of an outbreak, the first sign of tingling, and she takes probably five to six thousand milligrams the first day and the second day and then she begins to decrease a little bit each day. she does this until it disappears and she can sometimes stop it in its track without it ever appearing. She has to begin that immediately at the first sign or hers will go ahead and develop into a full-blown outbreak. She is very diligent with her eating and her vitamin intake and everything and she has been diagnosed with hashimoto’s which is a form of an immune deficiency. I think autoimmune disease, so it could be totally related.
Zovarix (spelling?) Cream is a prescription that works fast
Take lysine…it works better than abreva and all other methods. Thank you and you’re welcome! 😄
Valacyclovir
Shelly M. Is right , boron works, but the easiest and cheapest is to buy a 4 lb. Box or Borax at wal mart @ approx 4.35 . Heat up some distilled water in a pan and add some borax till its disolved. Let cool and put it in a bottle. Put some liberally on a paper towel to make it really wet then put it on the herpes,if you refrigerate the borax water after dissolved, it works faster (for all types)the herpes virus makes the area hot, cold borax water pulls out the heat quicker, The boron in the borax cools and stops the progression. Sometimes it needs held there for hours but stops the itch and progression. Popping herpes sores is not advised, but if it do breakes, just keep putting more borax water on it, (DO NOT RUB BUT ONLY LIGHTLY DAB, BUT mostly just hold it on the spot ) . It will clear up in a day or two ,sometimes quicker if caught right at first itch.. There is a wipe you can buy made by cottonelle that works great and fast for genital type herpes too called: Gentle Plus with / avec Aloe & E.it has a green lable.For some stramge reason it says tested with plumbers, (not sure why?) Look up the benefits of borax as it has many more uses and is harmless if used in moderation.
I have had cold sores since I was an infant. Some outbreaks covered my entire
lips and even chin. I have used everything mentioned for treatment & prevention. How do these findings relate to a history of cold sores? Is there a test to know if we are affected?
Lysine is the cure.
I second the comments of others who say to get a prescription for acyclovir. I have suffered from myalgic encephalomyelitis for 38 years, likely triggered by the mass malathion spraying in several Northern California counties in the early 80s,and officially diagnosed (I knew long before) at UCSF Medical Center in 2015. There is a definite connection between all eight HS viruses that affect humans, and ME, but whether that link is causative or associative is a matter of considerable debate.
Early on in the research about ME–then called CFS, and often said back then to be the same as fibromyalgia*–numerous researchers believed that Epstein Barr (HSV 4) caused the virus, owing to the history of mononucleosis common to many ME patients (the discovery of the link between EBV and mono was then recent–when I had a serious bout with it at 13 in 1964, my doctor, Gerald Besson, a medical advisor to the White House and certainly no slouch, assured me the cause was unknown, but that some doctors believed it to be a permanent infection–today, we know that both HSV 4 and HSV 5 can cause mononucleosis, still called infectious fever in some countries).
By about 2012,leading researchers were leaning towards HSV 3–herpes zoster (which causes chickenpox)–as the root cause, but within several years the suspicion was falling on HSV 6, a newly discovered virus, some researchers also thinking that both HSV 6 and 7 might lead to ME. There currently is a controversy among researchers about the link to HSV 6,but many are still convinced it’s the culprit.
Early on in my illness,the primary symptoms were the hideous relapses (involving enhanced pain, extreme weakness, and sensitivity to all forms of stress), to which ME sufferers are universally subject, now cited by the CDC as the chief defining symptom of those with this illness. When these relapses occurred, I often developed lesions on the back of my thigh which matched the description of HSV2 lesions, except not in the usual location. I never expected hsv2 at the time, as I was unaware that one can contract lumbar herpes from hsv2, a totally nonsexual manifestation, which can be contracted by, for instance, sitting on a wet bench in a locker room. But the symptoms pertinent to lumbar herpes, including severe lumbar radiculopathy, matched up with what I experienced. In 1990,I caught hsv 1 from my lovely wife, which I expected would happen eventually; I was given a prescription for acyclovir, which I maintain to this day. The cold sores tended to coincide with my relapses, and I soon discovered that when I felt a relapse coming (they are called PEM Episodes today), taking acyclovir could sometimes prevent its onset. I found that it was possible to do effective suppression therapy by taking one pill a day of either brand name Zovirax or a high quality acyclovir generic; this prevents most relapses. I have now been doing this for 25 years, and only very rarely do I get even the prodromes of a cold sore, let alone the actual lesion (I am something of an expert now on the relative efficacy of acyclovir generics-there are many today, and some do not work at all for me, most notably Teva, the first acyclovir generic, no damn good in my experience–the best generic I have found is by Carlsbad, almost as good as the original acyclovir, brand name Zovirax, which is far more effective than most imitators, and can even prevent cold sores during the prodromal phase
–unlike generics–but costs almost 1k per bottle).
Anyhow, my research has uncovered the fact that the common medical boilerplate about HSV1 and HSV2 is in many ways inadequate and misleading
–I strongly recommend reading up on the potential complications of those viruses all over the body, including nerves, brain and internal organs, that can result from HSV 1 and 2. You’ll find out some very interesting things that contradict much of which you may have heard. These viruses are damned insidious, and we can carry them for YEARS without a noticeable infection or skin eruption. I have had some very bizarre symptoms in recent years…
It is very important to have a doctor who will listen to you, and who admits that the penetralia of HSV phenomena is still not well understood by the medical profession. It is frequently stated that ME patients have to be “their own doctors”. We have suffered a great deal collectively from this deficit of understanding–many ME patients have experienced medical and judicial malpractice and derisive disbelief from employers and partners (remember the “Yuppie Flu”? Big joke,Jeff Foxworthy, you ignorant ^§¥&$¥¶!!!).
Today it is believed that whatever is the ultimate etiology with respect to viruses, ME is usually triggered by either toxic exposure, as likely happened to me, or by illness. And the reason I have written at such length, is that among the comments on this post, are at least three by people who, judging by their listed symptoms, I suspect have ME. I encourage everyone to read my words carefully, and if you suspect you may have ME, get on it NOW. Don’t wait. FIND A DOCTOR WHO SPECIALIZES IN ME and get a diagnosis. This is a vicious illness and often missed by doctors, as most still know little or nothing about it. Most people with diagnosed ME use either acyclovir, or valcyclovir, which is metabolised as acyclovir, for suppression therapy.
And good luck!!!
*(total BS; everyone with ME has some degree of fibromyalgia, but the reverse is not true–FM is a catch-all term for neuromuscular pain and dysfunction that can result from a variety of causes).
Ciao, vi scrivo dall’Italia e saluto tutti. Io soffro di hsv con macchie molto dolorose. In marzo 2021 sono stata invitata da una piccola azienda italiana a provare un prodotto per uso topico. Da quel momento, applicato solo 2 volte a settimana non ho più avuto sfoghi. Ho parlato con altre persone del test e ci sono stessi risultati. L’azienda si chiama Xathä Bio Lab e il prodotto non è ancora in commercio.
There is new medicine for Herpes FemiClear, you can find at CVS, Walmart. They say it’s kills the virus 99.9%, bought it for my self, it’s 50 or 60 dollars, there are two choices. Good luck getting well.
As soon as you start to feel a cold sore, put ice on it for 15 minutes. Try to do this every hour for a couple hours. If you catch it early enough it will reduce the blisters and heal faster.
I know it sounds crazy but I was told by an old dentist I used to have to spray Lysol disinfectant spray on a q-tip and put it on the cold sore as soon as it starts coming up. I’ve been doing this for 20 years and it not only makes them go away faster but it also made me not have any for about 15 of those 20 years! No not telling anyone to do this because I’m sure it says not to somewhere. I’m just saying what experiences I’ve had with it. I also put an ice cube in a washcloth and get it a little wet and hold it on my lip wherever the sore is until the ice cube melts. Of course I take it off and put it back on several times so not to freeze my lip. 🙏 It does State on the can that Lysol kills the herpes virus.
Carmex will burn the area worse. Use L-Lysine or Abreva.
Take 5000 mgs of lysine for cold sore up to shingles, all will be gone in a few days. Had shingles took 10,000 mgs for 3 days, all gone. Lysine is God sent.
J’ai contracté l’herpès génital quelques semaines après avoir accepté de coucher avec mon partenaire. Il était absent pour le travail quand j’ai été diagnostiqué et il m’a fallu tout mon courage pour le lui dire. Je ne savais pas ce qu’il penserait ou dirait, je pensais qu’il me larguerait. Sa réponse initiale a été “Eh bien, vous ne l’avez pas obtenu de moi” jusqu’à un jour plus tard, quand il m’a dit qu’il avait un bouton de fièvre. Mon docteur était un peu prude à ce sujet et a fait toute une histoire d’avoir à le signaler aux autorités sanitaires afin d’obtenir l’autorisation pour mon script. Heureusement, c’était le VPH de type 1 dont elle m’a dit qu’il était rare de se développer sur vos organes génitaux sans qu’il ne se propage sur tout le corps, mais les démangeaisons étaient intenses et douloureuses. Quelqu’un nous a demandé de contacter le Dr Kujo, ce que nous avons fait, il nous a envoyé le médicament à base de plantes après que nous l’ayons commandé et l’a utilisé en conséquence pendant 30 jours. herpès négatif. (6 mois jusqu’à maintenant, je n’ai même plus eu de bouton de fièvre ou de démangeaisons depuis). Mais c’était tellement effrayant, j’ai fait la même chose en cherchant sur Internet et j’ai traversé cette agonie toute seule ! Je ne l’ai toujours dit à personne, pas même à ma mère et j’espère juste que je n’aurai plus jamais à ressentir cette douleur ! Mon fiancé et moi en plaisantons souvent maintenant, surtout quand je veux à ma façon, je lui rappelle qu’il m’a de l’herpès ! Je dis à tout le monde maintenant que le Dr Baba Kujo est le seul à avoir le remède contre cette maladie mortelle, ne perdez pas de temps à le contacter comme nous l’avons fait. t
As a small child in Nicaragua I remember playing with friends running in the house screaming with pain in my mouth and my mother would look into my mouth and put a rock, alum, in a glass of water and made me Rinse with it,, saying ” don’t swallow any of it” and ever since I’ve been plagued with cold sores, when they’re outside I use blistex, or Carmel and I keep an alum rock in case they’re inside, but I think I’m going to try for inside and out hydrogen peroxide, thanks for the tip. Blessings to all.
Judy, valtrex will stop the cold sore when you feel it coming on. Ask your dr. for a prescription and keep it on hand.
LYSINE. LYSINE. LYSINE!!
EVERY DAY.
Don’t like talking about it but if it helps got diagnosed with simplex one almost 20 years ago in that period I had a total of 3 out breaks. Here are some factors. I was given the small pox vaccine during my time in the service the vaccine messed me up to the point the only meat I could eat is pork Wich is heavy on lyseane I know I butchered the spelling but I think one of those things has kept me from having repeat episodes like other people hope it helps some how I repeat no outbreaks in 20 years knock on wood
Maris Ferguson
I have had them for about 50 yrs also. I take valcyclovir 500mg every day and this prevents them.
Recently diagnosed with G-Hsv1 about 2 months ago. Dont really know how to figure out what really to do to treat my symptoms. My doctor advised not to take any medication.
Hello
When my HSV symptoms started, I thought I had a yeast infection. The following day, I had a swollen lymph node in my neck behind my ear. A couple days later, I felt stinging on my labia while showering, which I attributed to ingrown hairs. Later, I noticed bleeding and small ulcers. They began with dull pain and tingling, then my entire genital area was swollen and red. I’ve had an outbreak every month like clockwork dealing with the pain, itching and blisters i got tired of antiviral drugs and decided to try herbal medicine, i found Dr. Bulukona Website: bulukonaherbaltemple.wixsite.com/bulukona-temple and i discovered he was a professional in herbal medication and he has helped many people, i contacted him, we talked on phone and i confirmed he was genuine, i did all what he told me to do, i received the medicine, used it as he has prescribed and glory be to God almighty i am completely cured, i went back to my doctor to confirm it. They say there’s no cure but my case was different.
great
The best so far