
Using C. elegans, Scripps Research neuroscientists Brock Grill, Kirill Martemyanov and Dandan Wang discovered a new genetic regulator of opioid signaling. Credit: Scott Wiseman for Scripps Research
A team at Scripps Research in Florida has discovered a biological system that manages cells’ response to opioid drug exposure. The unexpected discovery offers new ideas for improving the safety of one of the most effective, and most abused, groups of pain medications.
In a paper published as a “First Release,” in the journal Science, lead authors Kirill Martemyanov, PhD, and Brock Grill, PhD, describe how they designed and implemented a new, unbiased approach for decoding the genetic network that controls the actions of opioids in a nervous system.
They used a small soil dwelling animal, the nematode worm, to discover something surprising about one of the most-studied drug receptors.
“A study like this makes it clear that even though we may think we know everything there is to know about the opioid response, we’re actually just scratching the surface,” Martemyanov says.
Their system relies upon the nematode c. elegans, engineered to express the mammalian surface receptor for painkilling drugs, the μ (mu) opioid receptor (MOR). The receptor is not normally found in the worms’ DNA, and adding it made the transgenic animals respond to opioids like morphine and fentanyl. The researchers then exposed the worms to mutagens and selected the ones with abnormal responses to opioids. Whole-genome sequencing and CRISPR engineering were then used to pinpoint the genes responsible for those aberrant responses.
“Forward genetics—unbiased genetic discovery—has never been applied to probing an opioid receptor like this,” Grill says. “The opioid epidemic is a huge problem and we don’t have good solutions. This type of approach can bring a whole new array of targets and a new way of thinking about and going after an old problem.”
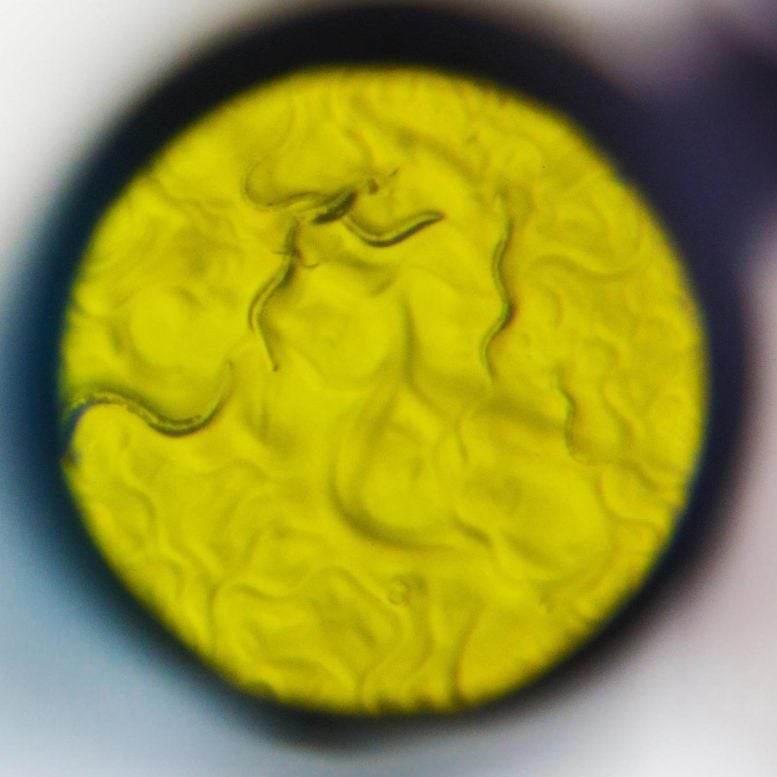
The nematode worm C. elegans is commonly used by neuroscientists to understand the nervous system. Scientists at Scripps Research in Florida used it in a new way to discover fresh insights about how cells regulate opioid signaling. Credit: Scott Wiseman for Scripps Research
The work ultimately led the researchers to the worms’ FRPR-13 receptor, conserved in all animals, and known as GPR139 in mammals. It is considered an “orphan” G-protein coupled receptor (GPCR) with poorly understood biology and unknown role in physiology. Further studies in mice showed that GPR139 was expressed on the same neurons as MOR and counteracted the effects of opioids on neuronal firing.
When researchers administered drugs that activate GPR139, mice dependent on opioid intake stopped taking the drug. Conversely, the genetic elimination of GPR139 augmented the pain-killing effects of opioids. The genetically modified mice lacking GPR139 also showed something remarkable—they showed very minimal withdrawal symptoms following chronic exposure to opioids. Withdrawal syndrome, a set of extremely unpleasant symptoms, usually sets in upon the discontinuation of opioids following their prolonged use. This compels people to resume drug-taking, fueling the dependence, Martemyanov says. The discovery could point a way toward lessening the suffering associated with opioid withdrawal, Grill says.
“A lot of addicts know that if they stop using, they are going to deal with anxiety, nausea, tremor and they are going to be in a lot of pain. That probably has a very negative impact on people wanting to go into rehab,” Grill says.
Opioids, a group of drugs that include fentanyl, Vicodin, OxyContin, and morphine, are highly effective at blunting severe pain, however, prolonged use can create tolerance and dependence. Excessive use can cause an overdose.
The discovery of GPR139 offers a new target for drug development aimed at making opioid therapies safer, Martemyanov says. The need for safer opioids is clear. According to the National Institutes on Drug Abuse, an average of 130 people a day in the United States die after overdosing on opioids. About 5 percent of people prescribed opioids transition to heroin, research has found.
The U.S. economic burden of the opioid crisis is estimated at $78.5 billion annually.
Martemyanov, Grill, and first author Dandan Wang, PhD, say they are hopeful that the discovery will lead to a new generation of pain medications with less potential for abuse and overdose.
Reference:”Genetic behavioral screen identifies an orphan anti-opioid system” by Dandan Wang, Hannah M. Stoveken, Stefano Zucca, Maria Daocesare Orlandi, Chenghui Song, Ikuo Masuhocaitlin Johnston, Karla J. Opperman, Andrew C. Giles, Matthew S. Gill, Erik A. Lundquist, Brock Grill and Kirill A. Martemyanov, 15 August 2019, Science.
DOI: 10.1126/science.aau2078
Funding for the work was supported by an NIH Cutting Edge Basic Research Award (R21DA040406) to Drs. Grill and Martemyanov, DA036596 to Dr. Martemyanov, and an NIH Center of Biomedical Research Excellence Grant (P20GM103638) to University of Kansas Genome Sequencing Core.






The statement was made ” About 5 percent of people prescribed opioids transition to heroin, research has found.” My response: nonsense! Either provide a reference or withdraw the statement.
No less an authority than Dr Nora Volkow, Director of the National Institute on Drug Abuse, tells us that “Unlike tolerance and physical dependence, addiction is not a predictable result of opioid prescribing. Addiction occurs in only a small percentage of persons who are exposed to opioids — even among those with pre-existing vulnerabilities…Older medical texts and several versions of the Diagnostic and Statistical Manual of Mental Disorders (DSM) either overemphasized the role of tolerance and physical dependence in the definition of addiction or equated these processes (DSM-III and DSM-IV). However, more recent studies have shown that the molecular mechanisms underlying addiction are distinct from those responsible for tolerance and physical dependence, in that they evolve much more slowly, last much longer, and disrupt multiple brain processes…”
Nora D Volkow, MD, and Thomas A McLellan, Ph.D., “Opioid Abuse in Chronic Pain — Misconceptions and Mitigation Strategies” . NEMJ 2016; 374:1253-1263 March 31, 2016]. http://www.nejm.org/doi/full/10.1056/NEJMra1507771
Addiction vs Dependance. Everyone has an opinion about it and very few are correct. People with Addiction will vacillate between various substances on being out of another. Be it paint, gasoline, transmission fluid, Heroin, Whip-it’s, Cocaine, Pot and on and on. They take everything to excess and have no off button. The people in Pain were hung out to dry by the Government to do one thing, save money, the REAL people in pain that need to go to a Pain Specialist, if they even could have been forced to taper down of dangerous meds at a rate that should not have happened. Threatening doctors with Pulling Medicare patients from pain clinics makes me wonder how constitutional this harassment is. It was to do one thing, save the Insurance Lobby from having to pay for pain in it’s entirety, someone with money was trying to force everyone through “addiction clinics who took a pain pill”. They also want us to be forced like addicts to pay cash for our pain meds. It still continues on, States and Republican’s moves on Women’s rights shows me that my assessment is correct. On Andrew Kolondy’s (the NON CDC scientist who invaded the CDC just to write this POS) 90 Milligram Morphine equivalent chart, they limited the use of buprenorphine and Subs for pain and everything else points to the Government not caring one rat’s ass about Pain patients or pain patients being addicted, only money.
As someone who was on prescribed pain meds for 17 yrs and then suddenly had them pulled, with no notice and nowhere to turn for help – just what did anyone expect to happen? Life my crappy life in agony 24/7? Be morally condemned for committing suicide to escape the horrendous pain from trigeminal neuralgia that defied all forms of treatment but opiods? Or sit quietly and suffer needlessly? My insurance wouldnt cover removal of my teeth (which ended up stopping the pain). I ended up moving to another state, qualified for medicaid, had teeth removed and am now on suboxone for physical dependence. If only my insurance had removed my teeth 17 yrs ago. Personally, if there was less finger pointing and more action to actually HELP people, the situation would never have gotten to its current state. Kudos to you for exploring avenues no one elsr has bothered to venture into!
“About 5 percent of people prescribed opioids transition to heroin, research has found.”
That’s not true. A toss-off statement made without any support. The rest of the article reads like a sci-fi piece that is interesting but so what.
Plus it has nothing to do with pain care. The article admits “Opioids, a group of drugs that include fentanyl, Vicodin, OxyContin, and morphine, are highly effective at blunting severe pain, however, prolonged use can create tolerance and dependence.” No mention on the millions of people who take this medicine for control of chronic pain. “Excessive use can cause an overdose” fails to mention that Rx opioid medicine when taken as prescribed is safe, effective, and virtually risk free.
“About 5% of people prescribed opioids transition to heroin” is absolutely 100% untrue! These are numbers made up & passed off as fact by PROP, CDC, FDA, DEA & Andrew Kolodny. 130 people a day (if that number is even correct) overdose on heroin or illicit, Fentanyl laced heroin which is smuggled into the country from China and/or Mexico! Very few are overdosing on legally prescribed pain medications! However, people ARE dying from chronic medical conditions being exacerbated by untreated pain! Chronic pain patients are are being cut off pain medication for no other reason than Physicians being afraid of the DEA busting down their doors and taking everything they’ve worked their entire lives for! For doing their job. For following the oath they took to ‘do no harm.’ So many people have gone from hardworking members of society to totally bedridden, unable to do even basic tasks! Many others have chosen to end their own lives because they were unable to take the daily, never ending pain. Why can’t we treat chronic pain AND help people who are addicted to heroin, methamphetamine, or whatever their drug of choice is? Why does it have to be one or the other? I pray someone puts s stop to the torture of innocent people before it’s too late.
As a husband and caretaker of a wife with chronic pain ( CRPS ) I have been reduced to what basically is a widower. Watching my wife either sleep constantly, if she is lucky , to watching and listening to her cry and pray to God to help with her pain. Her doctor is terrified of prescribing more than 20 mg. of methadone daily for fear of the DEA. My wife doesn’t want to take any narcotic medicine but has come to terms that it is all that will help
She has taken herself off of oxycodone, vicodin and currently morphine. My beliefs keep me from wishing ill will for anyone but if our lawmakers had to watch a loved one go through what my wife must they would convene special commitees and any other options needed to solve this problem
Take away all the political contributions made by big phar and things would change quickly. Shame on all those in positions to help those in need who are doing nothing but taking contributions to continue their terms in office without thinking of the people suffering who put them there. May God have mercy on your souls!
And what about if we were speaking about all the other mammals on this planet earth….what happens when say a whale is in intolerable pain or a dog or cat every veterinarian doctor on the face of the earth with the folks at Peta would put all their efforts into finding a way to help these poor animals live with their pain, right? But when we talking about actual human beings that are having life long intolerable pain with most times showing none of their conditions were caused by anything they did or did not do to themselves. Like car accidents or work injuries which occured from mechanical mishaps or from lack maintenance of machinery or not fixing broken vehical’s properly leaving these items to run down instead of upkeeping them properly then causing the worker using them to be injured at no fault of their own. And now are living in chronic debilitating pain which is consuming their every actions and moves. But we are not the helpless mammals of the earth so we do not seem to matter now do we?
I use codeine, Tylenol #4 or #3 for neck pain which is not very addictive with me. I’ve stopped using it for days without any withdrawal symptoms and sometimes forget to take it for a day when I’m not feeling any pain. I had withdrawal symptoms after stopping fentanyl in the form of Duragesic patches, and had withdrawal symptoms when I stopped taking methadone, and the codeine seems to work just as well. I can’t take OTC medications, like ibuprofen, because they interact with my anti-rejection medication for a liver transplant after getting cancer.
Now the hospital is trying to stop all its doctors from prescribing opioids and my doctors has to reduce my dosages each time he writes a prescription. These kind of laws are forcing people to go to their local bars to find a heroin dealer for their pain.
I have lived with chronic pain for 17 years, and some of these addiction and tolerance statistics are so untrue. While my pain has increased from additional pain causing diagnoses, my level of pain medication has barely risen in that time. Am I tolerant to my medications? Yes. Am I dose dependent? Yes. That comes along with many medications, not just opioids. I have several non scheduled medications that I could not go off of without tapering without suffering from severe side effects, but none of those carry the same stigmas, so of course they don’t get mentioned in literature. Studies show that people in chronic pain who take their medication correctly and without issue for the first year of treatment have a less than 1% risk of abuse, addiction, or subversion of their medication. We don’t take these medications because we like to or want to…we take them to be able to have some semblance of function. I can guarantee that pretty much every chronic pain patient would gladly hand over all of their medications if it meant having a normal, functional, pain free life. And I would more than gladly go through withdrawals to get there!
Thanks Jeanette for sharing your experience. My story is pretty much the same as yours. The generalizations that are often tossed out are so inaccurate and unfair to those who have legitimate need for long term opiate pain meds.
I am in my late years of life and think something is wrong about prescribing pain medication. I simply want to have a choice about my health care. Women have a choice to end their pain of an unwanted child by having an abortion. They, in a sense,have a choice to end their pain by selecting death. I want the same right to make a choice to choose a pain free life by using pain medication for long term chronic pain. I choose to have life just as they choose to have death.
Dont listen to this CRAP, ive been dealing with chronic pain due to rotator cuff problems and degenerative disc disease, im 52yrs old and I have been taking 15 mg oxycodone for 5 years at 6 pills per day,180 per month, and several times over those five years I have went at least 1 week without taking my pain medication deliberately just to see what type of side effects the withdrawals would cause in order to find out if it was something I could bare, besides a little insomnia I have had no withdrawal symptoms whatsoever, I credit that to taking the medication exactly as prescribed, taking pain medication away from those who truly need it because a bunch of jackasses want to abuse the medication should not be accepted, punish those using this medication inappropriately and illegally not the patients who truly need it, and stop trying to scare the public about these medications to further your false narratives !!!
Yet another story with a click bait headline that makes reckless unsubstantiated statements wrapped in a medical journal article of some merit whence shows some promise for treating the grandchildren of current chronic pain patients. So much politik, so little science. To hell with those suffering chronic pain NOW. We’ve been sent out on an ice flow so politicians and doctors who have drank the “opioid crisis” Kool Aid. This country has an illicit drug problem born in a generation of hopelessness. The government can no more solve the real problem than they can payoff this country’s debt. So they divert to the more visible sideshow of smacking around the most vulnerable in our society.
I couldn’t believe all these comments. It is people who are living the same nightmare. When is it going to STOP!? I have been dropped fr my pain med since Feb of this yea, 2019. I keep hoping & praying, but nothing is changing. When do we as people, that struggle w/getting everything! Come together to ask Why? When? How? If any one person was suffering or the spouse of one in Govt or in the Health Network… as soon as there were opioids needed…they would be provided! With or w/o the law. I ask myself almost Every Day… do I live in the USA? Is this Really Happening!? We KNOW every new drug bust or Pharmacist that goes on trial is on for quantities so outrageous that this has NOTHING to do w/regular pain sufferers!
It has to Stop! Really, like one said above, you cant take Ibuprofen, if you can…it is eating a hole in your stomach not to mention the damage it can cause for heart disease & worse.
Statistics are sketchy at best without exposing the data and #’s used to reach those. “Stats” are an easy way to present information. Information is a perceived representation of information from one person to another. Generally speaking, the host of information is presenting a persuasive message of “analytical ” data to support their case.
I call bulls#!t. We are not worms. Humans and you are describing a genetic rare DNA chromosome???
How about spending $ on opiod replacement with cannabis. How about studying the endocannabinoid system and the over 188 available cannabinoids…….
This is years away! How can you not see that there are so many of us suffering today but we’ll never make it to see anything nrw! between the pain and the l9nely solitary lives they have now given to us … if we are not already dead we are dying!
It is disgraceful what the politicians have done to some of the most susceptible people in our country. I wish someone with the resources and time would open a political foundation to represent millions of Americans who live in pain and have lost their effectiveness because of federal intervention of fair and humane medical practises.
The thing is, the OVERDOSES from HEROIN & those from OXYCONTIN that happen daily are linked with PAIN MGMT PRESCRIBED MEDS. Ppl do OD on Pharma OXYCONTIN. BUT, NOT IF THEY TOOK IT AS PRESCRIBED, OR DIDNT TRY TO BREAK IT DOWN FOR IV INJECTION. THE PPL THEY ARE FINDING DEAD DAILY ALL JUST INJECTED STREET BOUGHT OXYCONTIN OR HEROIN. IV DRUG USERS.
I HAVE SUFFERED HORRIBLY SINCE BEING WEANED OFF PAIN MGMT. TO THE POINT OF BEGGING GOD TO LET ME DIE. IVE WATCHED 80+ YR OLD POL TAKEN OFF THEIR 7 OR 8 YEAR DARVOCET & 5MG HYDROS GO FROM SOME QUALITY OF LIFE (BECAUSE THE ELEVATED SENSE OF WELL BEING Got these STILL PRODUCTIVE, active ppl out of bed & on their way) ; to being unable to walk 4 miles a day, work in a garden, or stand for hrs volunteering. IVE ATTENED 3 FUNERALS SINCE SEPTEMBER 2018 WHERE 80+ YEAR OLDS STUCK A GUN IN THEIR MOUTH. COULD THEY HAVE CHOSEN HEROIN OR OXY? YES. BUT, RHEY ARENT JUNKIES.
Just once I would like to read about a research project that discovered an alternative(s) that helps people with severe chronic pain who really depend on opioid treatment. I would be suicidal without medication. I have Trigeminal neuralgia and dysethesia from the surgeries to fix it. It’s called the suicide disease. I am treated like a criminal for taking opioids but there is no alternative. What about terminal patients and their pain? Veterans with permanent battlefield injuries? Researchers should be working on trials that test methods for repairing damaged nerves, repairing neural pathways, ameliorating neuralgia, stem cell therapy, alternatives to current meds.
Thank YOU actually, Dr Richard Lawhern PhD, the first commentator on this asinine article! You are the FIRST, I BELIEVE, to post ANY referenced rebuttal to the constant villianization of opioid meds!! All I ever read, like daily, are supposed factoids condemning the prescription of these life-changing meds, even though they can single-handedly bring any semblance of quality of life to the sick and suffering! It’s a witch hunt spawned by the fact that a minority of idiot patients get themselves hopelessly addicted due to their own ignorant choices in life. .kudos to you sir!! We need more people like you to speak out before the talking heads succeed in trashing these meds for good!!
I have chronic pain that has debilitated me for years. I’ve been on opioids continually since 2012. Without them I would have no quality of life. I am so sick and tired of being blanketed under the umbrella of addicts. The amount of oxycodone I’m prescribed has lessened while my pain has gotten worse. I don’t understand the logic. Why are the responsible ones being condemned. I comply with all the rules like taking urine tests monthly and at random along with med checks. I’m not the one that they should be looking at, I’ve done nothing wrong. I’m allergic to NSAIDS and I’ve tried so many other options for pain that DO NOT work. I’m so frustrated beyond belief.
I’ve no clue if these numbers are correct or not what I do know is I have a high tolerance for opiod meds. My doctor also got scared prescribing me morphine for my chronic pain when she cut me off of 260mg a day & told me I was an addict & to sign up for methadone who h later into methadone. My teeth have rotted out from it, I’ve now switched to suboxin which apparently doesn’t work for pain I’m on 24mgs the highest dose, I barely get any relief & my life has turned into something I’d rather not even live! I have a 1 year old granbaby I can’t even care for because my pain has put limitation on my life, this isn’t fare I didn’t ask to be an addict, my doctor did that all for me! I’ve become someone I no longer know & act out in ways I never would if I weren’t in constant pain, I hate who I’ve become & I see no other alternative! A write-up about worms barely scratching the surface of the actual issue doesn’t sound too promising but there’s always hope I guess!
My name is Michael and I am, unfortunately, in pain 24/7 from Cervical Spinal Stenosis. I had a surgery (cervical discectomy and fusion-a fancy way of saying they removed the disc from between C5 and C6, and then fusing together the 2 vertebrae and adding titanium plate and screws). Fusion didn’t take, and I was constantly going to ER (my primary physician was not willing to help with the pain, stating that I’d had surgery to fix the problem), asking for an Xray of my neck. I was constantly in pain that was worse than before the surgery. I’d never ask for pain meds, just an Xray, stating that something has gone very wrong with the fusion, I could feel it. After bugging them for 14 months, they finally asked me, if they gave me the Xray I was, in this doctor’s words, “supposedly always asking for,” would I please stop coming to the ER? Well, of course I said yes. Even the Xray Tech could tell-actually any fool could- after 14 months of 2 vertebrae loosely held together by the plate and screws at the front of my neck and now not even a broken disk to cushion the two from clacking together bone to bone, the tech showed me a nightmare on my Xray film: the titanium plate was bent into more than a parentheses mark: ‘(‘, but not quite as bent as a ‘C’. I can only figure that my body slowly bent this harder-than-steel plate. Oh, and the vertebrae were a hot mess that, using punctuation marks (I apologize for the crudeness but I can’t describe it), the parentheses mark is the plate and the less-than symbol is the angle at which the two vertebrae had gotten positioned over 14 months: (< so that the plate had the front of the 2 vertebrae crushed together and then it only opened from there, to about 1.2cm/a half of an inch opening in the back of my neck. Keep in mind the spinal cord is running down through this.
Fast forward to another surgery where they made a 6 inch incision in the back of my neck to get around to fix the front plate. And then they tried to make it all fuse again, wrapping titanium wire to jury-rig their screw up and now the two are really fused. I came out of anesthesia already leaking tears and going back and forth between, "Oww, owww, it hurts even more now!" and, "Why am I still in so much pain?"
Fast-forward again to 13 years later. The pain had been since early 1998. As the pain got to the point where I could no longer do Home Healthcare (my own business- I was an Air Force Medical Technician which is an EMT and Nurse combination), in late 1999, I was taking care of Stanley B. Sr. and because of pain, I gave a few months notice and went for surgery #1 in early 2000, #2 in 2003 (yeah, it took over a year and a half to set up the 2nd surgery from the time of the Xray discovery, "You got a broken neck". Okay, 13 years later, I get my pain meds torn away from me almost over night. 7 months later, December 8th, 2016… I am living with a friend who has what I have, plus 2 rotator-cuff injuries, and I was trying my best to help out, but I slumped into a depression that was so bad I stopped taking my Depression, ADD, Fibromyalgia, and headache medication, then I had finally had enough. I had no gun. But after 3 days of no sleep, meds, and barely eating, I got up, grabbed a serrated knife that I knew was sharp enough and began sawing at my neck.
Of all the days to get up early, my friend came out to the kitchen to find me crying and cutting my neck, she called 911. I tried even harder to cut deeper, now pressing the blade using my free hand and pressing on the back of the blade.
Now, I am in a Nursing home-type 3 (Assisted living with staff on around the clock, but it's not like a locked psychiatric unit. And now I have pain still, of course. Plenty of it. And sometimes I still feel like stopping my pain permanently. But I know to tell someone now before trying that. Oh and I am on 16 mg Buprenorphine/Suboxone. Why? Because I got screwed from the Pain Clinic when they shut down.
I agree with everyone going through chronic pain. Our society always goes black to white and no gray. For those of us having pain from real physical issues should not be punished for those that abuse…it’s ridiculous and now as they said..the Dr’s don’t up things due to DEA…I have never abused any drug ever…and I am angry that our society always does this…next will be benzo’
as I already heard of horrifying report. I wish there was balance..those of us in pain are not abusers for the most part..I don’t’ have accurate scientific studies…I would guarantee we in pain mostly do not abuse and do need increases since that is how any medicine works..including cannabis…should we be monitored every month…yes…I am upset and frustrated..and instead of having groups pop up with other solutions..nothing because it will take years to find substitutes which would be wonderful…I bet 99% of pain sufferers would be very happy to go to non opiod choices