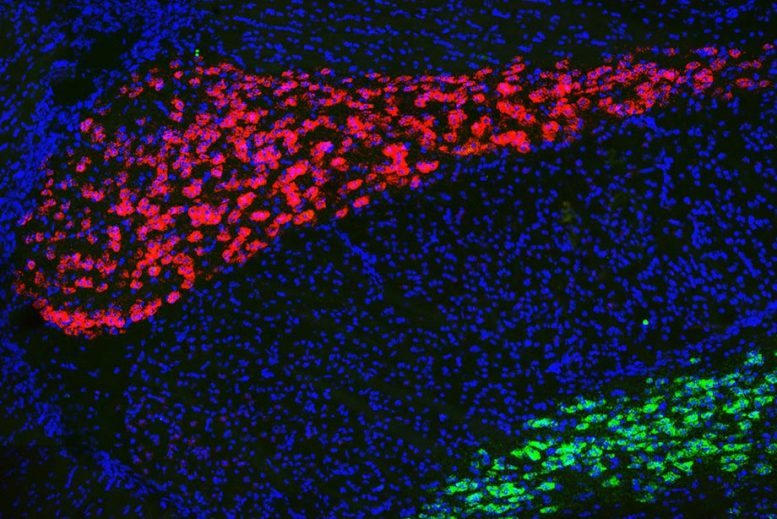
MIT neuroscientists have discovered a key role in memory formation for a region of the thalamus known as the anterodorsal thalamus, shown in red. Neurons of the neighboring thalamic reticular nucleus are shown in green. Credit: Dheeraj Roy and Ying Zhang
Study suggests a common mechanism underlies some behavioral traits seen in autism and schizophrenia.
Many neurodevelopmental disorders share similar symptoms, such as learning disabilities or attention deficits. A new study from MIT has uncovered a common neural mechanism for a type of cognitive impairment seen in some people with autism and schizophrenia, even though the genetic variations that produce the impairments are different for each condition.
In a study of mice, the researchers found that certain genes that are mutated or missing in some people with those disorders cause similar dysfunctions in a neural circuit in the thalamus. If scientists could develop drugs that target this circuit, they could be used to treat people who have different disorders with common behavioral symptoms, the researchers say.
“This study reveals a new circuit mechanism for cognitive impairment and points to a future direction for developing new therapeutics, by dividing patients into specific groups not by their behavioral profile, but by the underlying neurobiological mechanisms,” says Guoping Feng, the James W. and Patricia T. Poitras Professor in Brain and Cognitive Sciences at MIT, a member of the Broad Institute of Harvard and MIT, the associate director of the McGovern Institute for Brain Research at MIT, and the senior author of the new study.
Dheeraj Roy, a Warren Alpert Distinguished Scholar and a McGovern Fellow at the Broad Institute, and Ying Zhang, a postdoc at the McGovern Institute, are the lead authors of the paper, which was recently published in Neuron.
Thalamic connections
The thalamus plays a key role in cognitive tasks such as memory formation and learning. Previous studies have shown that many of the gene variants linked to brain disorders such as autism and schizophrenia are highly expressed in the thalamus, suggesting that it may play a role in those disorders.
One such gene is called Ptchd1, which Feng has studied extensively. In boys, loss of this gene, which is carried on the X chromosome, can lead to attention deficits, hyperactivity, aggression, intellectual disability, and autism spectrum disorders.
In a study published in 2016, Feng and his colleagues showed that Ptchd1 exerts many of its effects in a part of the thalamus called the thalamic reticular nucleus (TRN). When the gene is knocked out in the TRN of mice, the mice show attention deficits and hyperactivity. However, that study did not find any role for the TRN in the learning disabilities also seen in people with mutations in Ptchd1.
In the new study, the researchers decided to look elsewhere in the thalamus to try to figure out how Ptchd1 loss might affect learning and memory. Another area they identified that highly expresses Ptchd1 is called the anterodorsal (AD) thalamus, a tiny region that is involved in spatial learning and communicates closely with the hippocampus.
Using novel techniques that allowed them to trace the connections between the AD thalamus and another brain region called the retrosplenial cortex (RSC), the researchers determined a key function of this circuit. They found that in mice, the AD-to-RSC circuit is essential for encoding fearful memories of a chamber in which they received a mild foot shock. It is also necessary for working memory, such as creating mental maps of physical spaces to help in decision-making.
The researchers found that a nearby part of the thalamus called the anteroventral (AV) thalamus also plays a role in this memory formation process: AV-to-RSC communication regulates the specificity of the encoded memory, which helps us distinguish this memory from others of similar nature.
“These experiments showed that two neighboring subdivisions in the thalamus contribute differentially to memory formation, which is not what we expected,” Roy says.
Circuit malfunction
Once the researchers discovered the roles of the AV and AD thalamic regions in memory formation, they began to investigate how this circuit is affected by loss of Ptchd1. When they knocked down expression of Ptchd1 in neurons of the AD thalamus, they found a striking deficit in memory encoding, for both fearful memories and working memory.
The researchers then did the same experiments with a series of four other genes — one that is linked with autism and three linked with schizophrenia. In all of these mice, they found that knocking down gene expression produced the same memory impairments. They also found that each of these knockdowns produced hyperexcitability in neurons of the AD thalamus.
These results are consistent with existing theories that learning occurs through the strengthening of synapses that occurs as a memory is formed, the researchers say.
“The dominant theory in the field is that when an animal is learning, these neurons have to fire more, and that increase correlates with how well you learn,” Zhang says. “Our simple idea was if a neuron fires too high at baseline, you may lack a learning-induced increase.”
The researchers demonstrated that each of the genes they studied affects different ion channels that influence neurons’ firing rates. The overall effect of each mutation is an increase in neuron excitability, which leads to the same circuit-level dysfunction and behavioral symptoms.
The researchers also showed that they could restore normal cognitive function in mice with these genetic mutations by artificially turning down hyperactivity in neurons of the AD thalamus. The approach they used, chemogenetics, is not yet approved for use in humans. However, it may be possible to target this circuit in other ways, the researchers say.
The findings lend support to the idea that grouping diseases by the circuit malfunctions that underlie them may help to identify potential drug targets that could help many patients, Feng says.
“There are so many genetic factors and environmental factors that can contribute to a particular disease, but in the end, it has to cause some type of neuronal change that affects a circuit or a few circuits involved in this behavior,” he says. “From a therapeutic point of view, in such cases you may not want to go after individual molecules because they may be unique to a very small percentage of patients, but at a higher level, at the cellular or circuit level, patients may have more commonalities.”
Reference: “Anterior thalamic dysfunction underlies cognitive deficits in a subset of neuropsychiatric disease models” by Dheeraj S. Roy, Ying Zhang, Tomomi Aida, Soonwook Choi, Qian Chen, Yuanyuan Hou, Nicholas E. Lea, Keith M. Skaggs, Juliana C. Quay, Min Liew, Hannah Maisano, Vinh Le, Carter Jones, Jie Xu, Dong Kong, Heather A. Sullivan and Arpiar Saunde, 30 June 2021, Neuron.
DOI: 10.1016/j.neuron.2021.06.005
The research was funded by the Stanley Center at the Broad Institute, the Hock E. Tan and K. Lisa Yang Center for Autism Research at MIT, the James and Patricia Poitras Center for Psychiatric Disorders Research at MIT, and the National Institutes of Health BRAIN Initiative.






Be the first to comment on "Some Brain Disorders – Such As Autism and Schizophrenia – Exhibit Similar Circuit Malfunctions"