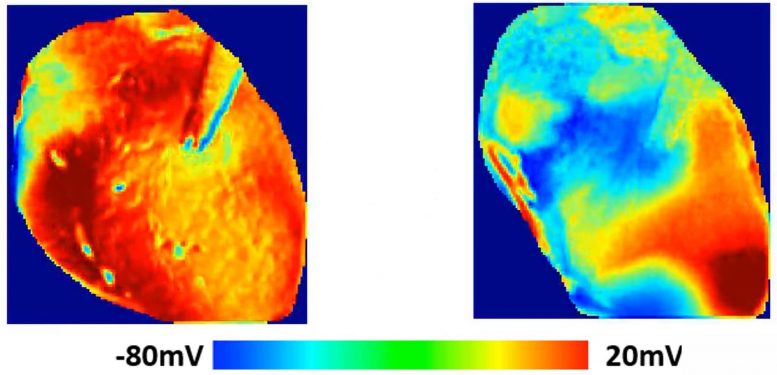
Images show the voltage surface on a rabbit heart with and without HCQ. Without the drug (normal) the electrical activation spreads homogeneously, while with HCQ, waves propagate unevenly, generating complex spatiotemporal patterns and arrhythmias. Credit: Georgia Tech School of Physics
The malaria drug hydroxychloroquine, which has been promoted as a potential treatment for COVID-19, is known to have potentially serious effects on heart rhythms. Now, a team of researchers has used an optical mapping system to observe exactly how the drug creates serious disturbances in the electrical signals that govern heartbeat.
The research, reported on May 29, 2020, in the journal Heart Rhythm, found that the drug made it “surprisingly easy” to trigger worrisome arrhythmias in two types of animal hearts by altering the timing of the electrical waves that control heartbeat. While the findings of animal studies can’t necessarily be generalized to humans, the videos created by the research team clearly show how the drug can cause cardiac electrical signals to become dysfunctional.
“We have illustrated experimentally how the drug actually changes the waves in the heart, and how that can initiate an arrhythmia,” said Flavio Fenton, a professor in the School of Physics at the Georgia Institute of Technology and the paper’s corresponding author. “We have demonstrated that with optical mapping, which allows us to see exactly how the waveform is changing. This gives us a visual demonstration of how the drug can alter the wave propagation in the heart.”
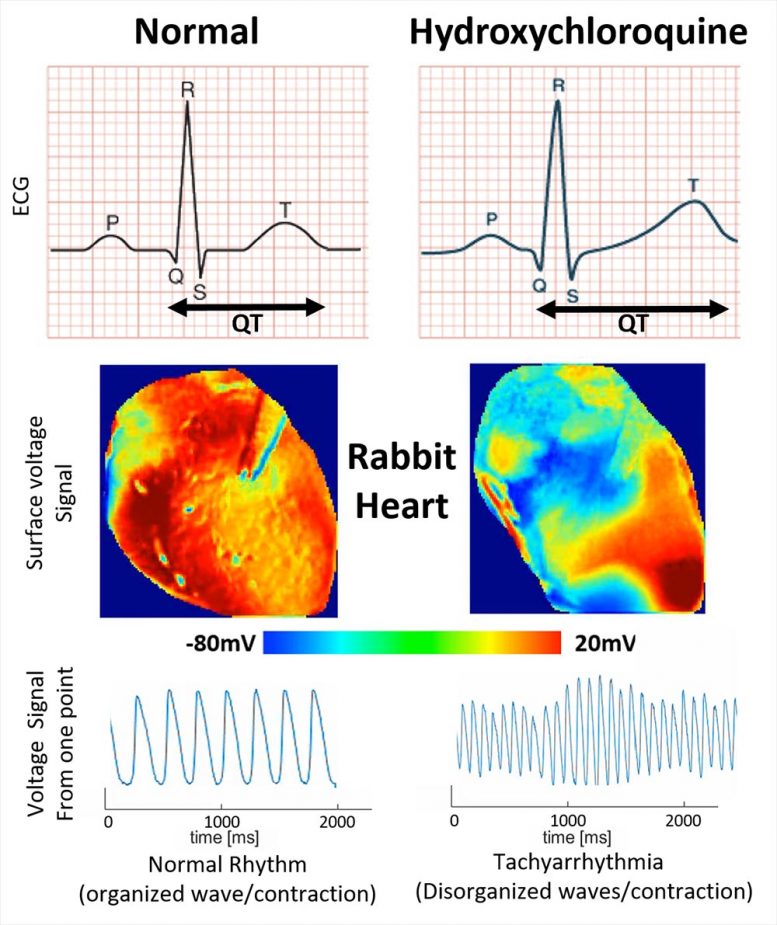
Images show the voltage surface on a rabbit heart with and without HCQ. Without the drug (normal) the electrical activation spreads homogeneously, while with HCQ, waves propagate unevenly, generating complex spatiotemporal patterns and arrhythmias. Credit: Georgia Tech School of Physics
What the team saw was an elongation of the T wave, a portion of the heart cycle during which voltages normally dissipate in preparation for the next beat. By extending the QT portion of one wave cycle, the drug sets the stage for disturbances in the next wave, potentially creating an arrhythmia. Such disturbances can transition to fibrillation that interferes with the heart’s ability to pump.
The ability to easily trigger disturbances known as “long QT” reinforces cautions about using hydroxychloroquine (HCQ) in humans — particularly in those who may have heart damage from COVID-19, cautioned Dr. Shahriar Iravanian, a co-author of the paper and a cardiologist in the Division of Cardiology, Section of Electrophysiology, at Emory University Hospital.
“The hearts used in the study are small and very resistant to this form of arrhythmia,” Iravanian said. “If we had not seen any HCQ-induced arrhythmias in this model, the results would not have been reassuring. However, in reality, we observed that HCQ readily induced arrhythmia in those hearts. This finding is very concerning and, in combination with the clinical reports of sudden death and arrhythmia in COVID-19 patients taking HCQ, suggests that the drug should be considered a potentially harmful medication and its use in COVID-19 patients be restricted to clinical trial settings.”
Georgia Tech postdoctoral fellow Ilija Uzelac administered HCQ to the animal hearts — one from a guinea pig and one from a rabbit — while quantifying wave patterns changing across the hearts using a high-powered, LED-based optical mapping system. Voltage-sensitive fluorescent dyes made the electrical waves visible as they moved across the surface of the hearts.
“The effect of the arrhythmia and the long QT was quite obvious,” said Uzelac. “HCQ shifts the wavelengths to larger values, and when we quantified the dispersion of the electrical current in portions of the heart, we saw the extension of the voltage across the tissue. The change was very dramatic comparing the waveforms in the heart with and without the HCQ.”
The drug concentration used in the study was at the high end of what’s being recommended for humans. HCQ normally takes a few days to accumulate in the body, so the researchers used a higher initial dose to simulate the drug’s effect over time.
In a normal heartbeat, an electrical wave is generated in specialized cells of a heart’s right atrium. The wave propagates through the entire atria and then to the ventricles. As the wave moves through the heart, the electrical potential created causes calcium ions to be released, which stimulates contraction of the heart muscle in a coordinated pattern.
Drugs such as HCQ modify the properties of these ion channels and inhibit the flow of potassium currents, which prolongs the length of the electrical waves and creates spatial variations in their properties. Ultimately, that can lead to the development of dangerously rapid and dysfunctional heart rhythms.
“The wavelength becomes less homogeneous and that affects the propagation of additional waves, producing sections of the heart where the waves do not propagate well,” Fenton said. “In the worst case, there are multiple waves going in different directions. Every section of the heart is contracting at a different time, so the heart is just quivering. At that point, it can no longer pump blood throughout the body.”
Patients taking HCQ for diseases such as lupus and rheumatoid arthritis rarely suffer from arrhythmia because the doses they take are smaller than those being recommended for COVID-19 patients, Iravanian said.
“COVID-19 patients are different and are at a much higher risk of HCQ-induced arrhythmia,” he said. “Not only is the proposed dose of HCQ for COVID-19 patients two to three times the usual dose, but COVID-19 has effects on the heart and lowers potassium levels, further increasing the risk of arrhythmia.”
Fenton and his colleagues have already begun a new study to evaluate the effects of HCQ with the antibiotic azithromycin, which has been suggested as a companion treatment. Azithromycin can also cause the long QT effect, potentially increasing the impact on COVID-19 patients.
Reference: “Fatal arrhythmias: Another reason why doctors remain cautious about chloroquine/hydroxychloroquine for treating COVID-19” by Ilija Uzelac, Ph.D., Shahriar Iravanian, MD, Ph.D., Hiroshi Ashikaga, MD, Ph.D., NealK. Bhatia, MD, Conner Herndon, MS, Abouzar Kaboudian, Ph.D., James C. Gumbart, Ph.D., Elizabeth M. Cherry, Ph.D., Flavio H. Fenton, Ph.D., 29 May 2020, Heart Rhythm.
DOI: 10.1016/j.hrthm.2020.05.030
The study, which was supported by grants from the National Institutes of Health and National Science Foundation, was also coauthored by Dr. Hiroshi Ashikaga from Johns Hopkins University School of Medicine; Dr. Neal Bathia from the Division of Cardiology, Section of Electrophysiology at Emory University Hospital; Conner Herndon, Abouzar Kaboudian, and James C. Gumbart from the Georgia Tech School of Physics, and Elizabeth Cherry from the Georgia Tech School of Computational Science and Engineering.
This research is supported by the National Institutes of Health under award number 1R01HL143450-01 and the National Science Foundation under grant 1446675. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the National Science Foundation.




Great, its problematic in higher dosages, as is Azithromyicin… yet both were approved by the FDA, and remain in use. I have to wonder, a a layperson, how many OTHER drugs, if any, are tested this way? I wouldnt know the answer, nor does the public.
But if this type of test is only being conducted on this particular drug, I sure as heck want to know why that is the case.
Off label use of drugs has been going on far longer than Covid-19 has been around. All this proves is that just because the FDA hasnt approved a drug for a certain disorder, doesn’t mean its ineffective, only potentially harmful in larger, untested dosage.
I’m not slamming or dismissing the science, only the motives and reasoning behind which drugs are tested,and to what level, before they’re approved.
Great comment about a drug used for 70 YEARS by MILLIONS of patients EVERY DAY.
“Gosh, I had just called all my lupus and rheumatoid arthritis patients to tell them that they should immediately stop that horrifically dangerous drug, hydroxychloroquine…! I realized that if it were too dangerous to use for patients with a life-threatening illness, it surely was ridiculous to use in much higher doses for people who merely have diseases causing inconvenience or pain.”
As someone who did NOT vote for Donald Trump, even I can see that SOMEONE is going through great lengths to discredit a 70-year-old drug used by millions of people worldwide either for disease or where Malaria is present around the world. The only thing giving small animal hearts HCQ at HIGH human dosages proves is that there’s a danger point for ANY drug…next time let’s try aspirin, at high dosage on a small animal heart to really scare the public.
Clearly this “study” was designed for the purpose of creating this headline…
“Study Shows Hydroxychloroquine’s Harmful Effects on Heart Rhythm – Urges for COVID-19 Use Restrictions”
Many Lupus patients take HCQ every day…has anyone heard about ANY Lupus patients coming down with COVID-19 same for RA patients and those who take HCQ for Malaria each day. If there were any I’m sure the media would be all over it as proof HCQ had no protective effect for this patient population….but they haven’t. Truth is this all began when Lupus doctors in Wuhan China noticed none of their Lupus patients were coming down with COVID-19. That’s really what started this whole investigation into HCQ/COVID-19. This just came out today in India.
“4 or more hydroxychloroquine doses reduced risk of coronavirus in healthcare workers: ICMR study” – The case-control investigation of the ICMR reveals that consumption of four or more maintenance doses of hydroxychloroquine led to a significant decline in the odds of healthcare workers getting infected with the coronavirus infection.
So is HCQ a cure of course not…but as we continue to wait for the randomized studies to complete…it sure looks like it could significantly help.