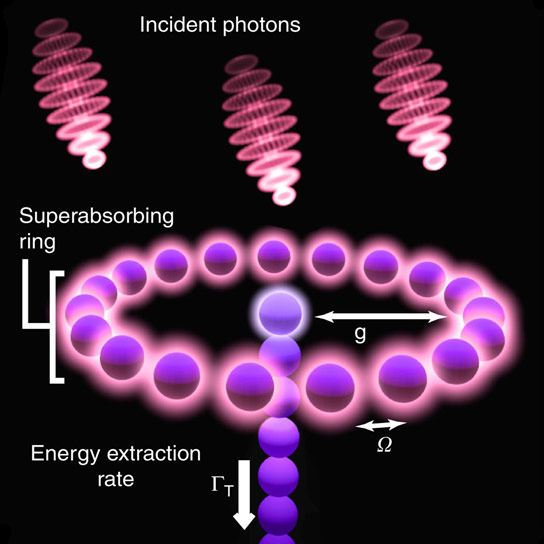
Photons absorbed by the ring give rise to delocalized excitons; ideally the ring maintains a specific exciton population to achieve enhanced absorption. Combined with a suitable charge sensor (for example, a quantum point contact) this enables photon sensing. We also model an application for photon harvesting, where newly created excitons are transferred from the ring to a central core absorber, followed by an irreversible process (for example, one-way transfer down a strongly coupled chain) to a center converting the exciton into stored energy. Credit: K. D. B. Higgins, et al. Nature Communications
Research from the University of Oxford establishes that superabsorption can be achieved and sustained in certain simple nanostructures, by trapping the system in a highly excited state while extracting energy into a non-radiative channel. The effect offers the prospect of a new class of quantum nanotechnology, capable of absorbing light many times faster than is currently possible.
‘Superradiance’, a phenomenon where a group of atoms charged up with energy act collectively to release a far more intense pulse of light than they would individually, is well-known to physicists. In theory, the effect can be reversed to create a device that draws in light ultra-efficiently. This could be revolutionary for devices ranging from digital cameras to solar cells. But there’s a problem: the advantage of this quantum effect is strongest when the atoms are already 50% charged – and then the system would rather release its energy back as light than absorb more.
Now a team led by Oxford University theorists believes it has found the solution to this seemingly fundamental problem. Part of the answer came from biology. ‘I was inspired to study ring molecules, because they are what plants use in photosynthesis to extract energy from the Sun,’ said Kieran Higgins of Oxford University’s Department of Materials, who led the work. ‘What we then discovered is that we should be able to go beyond nature’s achievement and create a ‘quantum superabsorber’.’
A report of the research is published in Nature Communications.
At the core of the new design is a molecular ring, which is charged to 50% by a laser pulse in order to reach the ideal superabsorbing state. ‘Now we need to keep it in that condition,’ notes Kieran. For this the team propose exploiting a key property of the ring structure: each time it absorbs a photon, it becomes receptive to photons of a slightly higher energy. Charging the device is like climbing a ladder whose rungs are increasingly widely spaced.
‘Let’s say it starts by absorbing red light from the laser,’ said Kieran, ‘once it is charged to 50% it now has an appetite for yellow photons, which are higher energy. And we’d like it to absorb new yellow photons, but NOT to emit the stored red photons.’ This can be achieved by embedding the device into a special crystal that suppresses red light: it makes it harder for the ring to release its existing energy, so trapping it in the 50% charged state.
The final ingredient of the design is a molecular ‘wire’ that draws off the energy of newly absorbed photons. ‘If you built a system with a capacity of 100 energy units the idea would be to ‘half-charge’ it to 50 units, and the wire would then ‘harvest’ every unit over 50,’ said Kieran. ‘It’s like an overflow pipe in plumbing – it is engineered to take the energy level down to 50, but no lower.’ This means that the device can handle the absorption of many photons in quick succession when it is exposed to a bright source, but in the dark it will simply sit in the superabsorbing state and efficiently grab any rare passing photon.
‘Eventually, harvesting sunlight in a highly-efficient way might one day be possible using superabsorbing systems based on our design, but a more immediate application would be building an extremely sensitive light sensor that could form the basis of new camera technology,’ Professor Simon Benjamin, a co-author of the report, explains. ‘A camera sensor harnessing the power of our superbsorbing rings would have very high time and spatial resolution. And it could pave the way for camera technology that would exceed the human eye’s ability to see clearly both in dark conditions and in bright sunlight.’
The research team included scientists from Oxford University, National University of Singapore, University of St Andrews, and the University of Queensland. The work was supported by the UK’s Engineering and Physical Sciences Research Council (EPSRC). A report of the research, entitled ‘Superabsorption of light via quantum engineering’, is published in in Nature Communications.
Reference: “Superabsorption of light via quantum engineering” by K. D. B. Higgins, S. C. Benjamin, T. M. Stace, G. J. Milburn, B. W. Lovett and E. M. Gauger, 22 August 2014, Nature Communications.
DOI: 10.1038/ncomms5705
arXiv: 1306.1483









In a properly-known quantum effect referred to as super radiance, atoms can emit light at an enhanced rate compared to what is possible in classical situations. This high emission price arises from the way that the atoms interact with the surrounding electromagnetic field. Logically, structures that super-radiate will have to also absorb light at a larger price than standard, but so far the super absorption of light has not been observed. This really happens in the end life of white dwarf ending in a dark dwarf.This postulate was discussed by Dr.Chandrasekhar and finally neglected.
Just like water affine emission of waves fire affine emission of waves from White dwarf is also possible towards its end before cooling based on magnetic-field selection.
Ref:Fire of London in 1666
Citation: Asymmetric Light Absorption and Radiation contributes to an interesting energy swing along ellipsoidal ring forming strange spatulate ellipsoidal curves with dark phase in between controlling the energy amplificationA vector polygon that oscillate between a unit circle and 1-A beta as an interfering Amplifier of Nyquist as generative and degenerative amplification produces lower and upper energies collected on ellipsoidal spatulate curves as curves of lower spatulate and upper spatulate collections
Mathematical theory on the physical significance the linear and nonlinear space showing an asymmetric absorption of energy under the original linear flat space by a spin of rotation as a diadonal matrix operator by a zero value potential becomes a singularity by the eigen value vector by a distortion involved by the the transformation scle factor that oscillate between +1 and _1 triagular upper and and lower Eigen wave element differtiate for a high and low energies that swing between the neutral dark energy that constrains an amplification of energy by the unit vector operative in between by the selective pi frequencies along generative and degenerative type Nyqusit oscillatins in understanding y tpe Net working systemsThe
Eigen wave vector diagonalizegsved as element of zeros contributes to a singularity with+1 -1 oscillations as upper and lower triangular matric elements contributes to to produce unit circle rotating vector by selective frequency polygons along the plane of scale factor forming a matrics by the difference of frequency difference along the pi shifted poles and zeros as generative and degenerative amplification at 1,j0 point at which the amplification oscillates
Ref:Citation:Diffrentiated supernova is born ID 8001375120
Strange possibility of collection of lower energy particles that may be collected in between oscillating galaxies that really produces a enlarges ellipsoidal projections along the upper and lower axis contributes to strange information with dark matter phases in between ejecting two energetic domains in between the zero potential that strange constrain the collections in the middle.
The star y type web structures contributes differential phasor vector forming a neutral core predicts the soliton of dark matter in the center surrounded by an extensive halo of dark matter in the form of large “spots”, which are the slowly fluctuating density waves becoming visible bright portions dark matter acting as a neutral plane. Forming a y type Net working systems of space
The colors represent the density of dark matter, with red indicating high-density and blue indicating low-density. On the left is a simulation of how non-interacting cold dark matter produces an abundance of smaller satellite galaxies. On the right the simulation shows the situation when the interaction of dark matter with other particles reduces the number of satellite galaxies we expect to observe around the Milky Way.
Photon forming a three phasor energies forming a dingular eigen xero energy eigen vector pulling the energy level between one upper levels that eventually swing for opposite palne also having two resonances. Newly created excitons are transferred from the ring to a central core absorber, followed by an irreversible process (for example, one-way transfer down a strongly coupled chain) to a center converting the exciton into stored energy possibly on two lower and upper energy regions as a swing with dark matter phase in between the first PHASOR Photons so that its photons, when absorbed by atoms, turned the atoms’ kinetic energy into magnetic energy in between a compression of electric vector and magneticfield vector.In between the the electric field that oscillate between north pole and south pole by the interactive manetic field by north pole and south pole in series aligned as two attractive and opposing phases ejects or absorbs in between forming an inductance and opposing increase in magneticfield potential energy by the directional flow and next moment forming capacitive type modules by opposing an increase electric field potential becomes an attractor but opposite direction becomes a repulsive force and hence in between the phasor converges towards a neutral plane of draker domain that really controls the energy flow.
Kieran Higgins of Oxford University’s Department of Materials, who led the work. ‘What we then discovered is that we should be able to go beyond nature’s achievement and create a ‘quantum superabsorber’.’ the absorption of many photons in quick succession when it is exposed to a bright source, but in the dark it will simply sit in the superabsorbing state and efficiently grab any rare passing photon. a key property of the ring structure: each time it absorbs a photon, it becomes receptive to photons of a slightly higher energy.
The important aspect of photon interaction by analogy may have a star connected phase into the dark region of phase that reallty shifts the phasor energy towards upper and lower regions forming an ellipsoidal axis elongations as applied to galaxy expansions also.
Two dual two kinds of resonances: resonances localized to the Ag half of the particle and resonances involving the entire particle properties of phase-separated Ag–Cu nanoparticles. The particles are fabricated using simple thin film evaporation followed by particle formation by vacuum annealing. The formed particles feature a two-faced Janus structure. Characterization is carried out at the single particle level utilizing transmission electron microscopy in combination with electron energy loss spectroscopy and cathodoluminescence, and modeled by finite element method simulations. We find that these particle sustain two kinds of resonances: resonances localized to the Ag half of the particle and resonances involving the entire particle. This is due to the difference in onset energy for interband transitions for the two metals. As the resonances are excited in Ag, large enhancements of energy absorption can be achieved in the Cu half of the particle. We also find a slight asymmetry to the emission of the particles in cathodoluminescence, with a preferential emission toward the silver side of the particle. Enhanced energy absorption into Cu means an increased number of generated hot electrons. This together with the ease of fabrication of the particles makes such structures interesting candidates for plasmon enhanced photocatalysis. Furthermore, because of the inherent phase separation of the materials, stability even at elevated temperatures is enhanced. Not being limited to the Ag–Cu system, a similar approach should work equally well for other phase-separated systems.
Ref:
1Publication: K. D. B. Higgins, et al., “Superabsorption of light via quantum engineering,” Nature Communications 5, Article number: 4705; doi:10.1038/ncomms5705
2C. Bœhm, et al., “Using the Milky Way satellites to study interactions between cold dark matter and radiation,” MNRAS (November 21, 2014) 445 (1): L31-L35; doi: 10.1093/mnrasl/slu115
Citation:Brillouin Scattering Induced Transparency can be manipulated b linguistic basic alphabets which interact with photon speed that can be varied along three planes of blue fast red slow and yellow block dynamics.
A light wave sent through empty space always oscillates in the same direction. certain materials can be used to rotate the direction in which the light is oscillating when placed in a magnetic field. This is known as a ‘magneto-optical’ effect which can be carried out by sound waves, the effect depends on how thick the material is: the larger the distance to be travelled by the light in the material larger the angle of rotation by the interactive acousto-optic waves interact with photons
Brillouin-Mandelstam scattering, originally discovered in the early 1920s, is the coupling of light waves and sound waves through electrostrictive optical forces and acousto-optic scattering. It is the fundamental physical process behind BSIT, and occurs in all solids, liquids, gases, and even plasmas,” the exact response of a resonance, especially for frequencies far from the resonant frequency, depends on the details of the physical system, and is usually not exactly symmetric about the resonant frequency, as illustrated for the simple harmonic oscillator above. For a lightly damped linear oscillator with a resonance frequency Ω, the intensity of oscillations I when the system is driven with a driving frequency ω is typically approximated by a formula that is symmetric about the resonance frequency:[1the susceptibility x(w) links the amplitude of the oscillator to the driving force in frequency space Systems for which damping is important (such as dampers keeping a door from slamming shut) have Q = 1/2. Clocks, lasers, and other systems that need either strong resonance or high frequency stability need high-quality factors. Tuning forks have quality factors around Q = 1000. The quality factor of atomic clocks and some high-Q lasers can reach as high as 1011[17] and higher.[18]
Photon interactions on three planes by the changing the directions :
So the condition for resonance in a resonator is that the roundtrip distance, 2d, be equal to an integer number of wavelengths λ of the wave: as 2d is equal N λ So the resonant frequencies of resonators, called normal modes, are equally spaced multiples of a lowest frequency called the fundamental frequency. The multiples are often called liinguistic overtones. There may be several such series of resonant frequencies, corresponding to different modes of oscillation.
Magnetic effects come about because particles have an internal magnetic direction called the ‘spin’, whereas electrical effects result from positive and negative charges within a material that can shift position in relation to one another as the electron spin varied for a multi eflection in between poles of electrons by pi phase selective variations in selective frequencies. It is so much easier to apply an electrical field with pinpoint precision, as all you need is a simple voltage pulse by avoiding a halls thrust by suppression of whistler wave dynamics making it inductive variations. Light at certain wavelengths can be absorbed out of a thin optical waveguide by a microresonator–which is essentially a tiny glass sphere–when they are brought very close
Certain basic alphabets such a cream hreem amm tahmm pamm lamm kamm gives an effect on human beings as well as on matter by sound interactive acoustic simulatio on photons. uses the extremely minute forces exerted by light to generate and control mechanical vibrations of microscale and nanoscale devices–a field called optomechanics. In resonant microcavities, these miniscule forces can be enhanced by many orders of magnitude. They are using these phenomena to unearth new physics behind how solids, liquids, and gases interact with light.
The number of times the linguistic alphabets interact on three planes of a phasor vector that is operative can be counted to brings out the results can be verified .It may be that the circular ring of solitons a can be oscillated to increase or decrease the energy levels in between in forming a spatulate ellipsoidal spatulate dynamics in between. they are nonlinear devices sometimes requiring the shuffling of alphabets
In future the interactive dynamics by certain acoustic effect on three plane phasor vector will be evaluated.This gives a clue on Thruva blocking the spinning and Athru dynamics initiated in those days by Saint Thirummooler in 8/16 plane phase oscillations.For example this may be used to increase the level of water as quantum replicative dynamics inside a well.
The pure sound of Om is also called as the “pranava” which means that it sustains life and travels through our breath or praana. Om is one of the myriad of mantras and Mantra means sound tool in Sanskrit. In fact various ancient languages like the Tibetan, prototypical Egyptian and even Sanskrit have all evolved as complex systems of onomatopoeia, where in the sound movements can produce energy and this is predominantly represented in the right hemisphere of the brain. On the contrary the narrative strand of sounds is governed mainly by the left hemisphere of the brain. As split up as aaa ooo mmm vibrations as three plane split-up as comparable with thre plane polarization of light as blue fast red slow and yellow block dynamics.
The use of sound as a healing therapy has existed for centuries. From the mantras of the Rig Veda, an ancient Vedic text, to modern gadgets, there are many therapies that utilize sound as the basis of healing. Some are passive, such as modern vibroacoustic therapy, while others require more participation on the patient’s part, such as in the repetition of mantras. Sound therapies include music, chanting, Tibetan singing bowls, and the modern BETAR table. This paper will explore some of these modalities.
Everything in nature vibrates at a particular frequency, including plants, rocks, stars, television sets, cellular phones and, of course, our bodies. When the body is continually exposed to vibrations similar, but not natural to its own frequencies, the new vibratory patterns may cause the natural frequencies to alter their vibrations to the new patterns. Dutch scientist Christian Huygens observed this principle, called entrainment, in 1655. He discovered that when he hung two clocks with different pendulum speeds close to each other on the same wall, the pendulums would ultimately synchronize and swing at the same speed.
When our natural frequencies become “out of tune,” the body may become out of balance and we may become ill. The same process that can cause our vibrations to be unhealthy can also be used to restore the vibrations to homeostasis. Sound healing seeks to return our vibratory patterns to their natural state. Sound not only heals physical, mental and emotional trauma, but it also is a conduit for progress on our individual spiritual journey, as some sounds lead us to higher levels of consciousness.
“Like ripples on a pond, the sound waves delivered by the focused speaker array resonate outward through every muscle, organ and joint in the body – a gentle music massage that melts away deeply-held patterns of tension, stress and pain. Riders float away from the cares and concerns of everyday life, a visit to a personal musical sanctuary where healing can take place.”
This paper for the review of Royal society of London
Sankaravelayudhan Nandakumar dt16-10-2018