
Recent research finds that the insulin receptor protein (INSR) is pivotal for brain stem cell longevity and growth.
According to a Rutgers University study, the cell receptor protein also stimulates the growth of brain cancer stem cells
Stem cells are the body’s raw materials — they are the cells that give rise to all other cells with specialized functions. In the right circumstances, stem cells in the body divide to produce new cells known as daughter cells.
Humans contain neural stem cells in their brains. These brain stem cells may develop into neurons, astrocytes, or oligodendrocytes. Because neural stem cells generate all of the brain’s cell types, there is a multitude of stem cells in an embryo’s brain. In fact, the majority of brain cells are born in the embryo stage. These cells persist till adulthood and can be found in particular regions of the brain. Neural stem cells are essential for your brain to properly function.
According to research from Rutgers University, a receptor that was first identified as necessary for insulin action and is also found on neural stem cells found deep in the brains of mice is crucial for brain stem cell longevity, a finding that has important implications for brain health and future therapies for brain disorders.
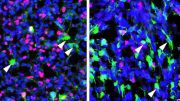
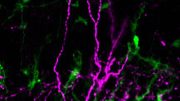
The research, published in the journal Stem Cell Reports, focuses on a particular protein known as the insulin receptor (INSR), which is prevalent in neural stem cells in the brain’s subventricular zone. Neural stem cells give rise to the entire nervous system throughout development and persist into adulthood. Over the course of a person’s life, these neural stem cells generate new neurons and non-neuronal cells that help the brain’s infrastructure and function.
Alternative Uses
Separately, while studying brain tumors, the researchers discovered that INSR plays an important role in the survival and maintenance of a population of specialized brain cancer cells known as glioblastoma (GBM) stem cells. They were able to reduce the growth of those primitive tumor-forming cells by inactivating the INSR in GBM stem cells.
“It’s important to understand the molecular mechanisms that are critical for the growth and sustenance of the brain’s stem cells under normal and abnormal growth states,” said study author Steven Levison, a professor of neuroscience in the Department of Pharmacology, Physiology, and Neuroscience and director of the Laboratory for Regenerative Neurobiology at Rutgers New Jersey Medical School. “Comprehending the signals that regulate these primitive cells could one day lead to new therapeutics for brain disorders.”
Many neurodegenerative disorders, such as multiple sclerosis, Parkinson’s disease, and Alzheimer’s disease, are connected with the destruction of brain cells, said co-author Teresa Wood, a Distinguished Professor and Rena Warshow Endowed Chair in Multiple Sclerosis in the Department of Pharmacology, Physiology, and Neuroscience at Rutgers New Jersey Medical School.
“If we could influence how brain stem cells function then we can use this knowledge to replace diseased or dead brain cells with living ones, which would advance the treatment of neurological diseases and brain injuries,” said Wood, who also teaches and conducts research at the Cancer Institute of New Jersey.
Cell receptors such as INSR are protein molecules that reside on the surfaces of cells. Substances, either natural or human-made, that open the “lock” of a receptor can spur a cell to divide, differentiate or die. By identifying which receptors perform these functions on specific cell types, and by understanding their structures and functions, scientists can design substances that act as keys to receptors, to turn them “on” or “off.”
Key to maintaining neural stem cells
Previous studies by this research team had shown that a certain “key,” the signaling protein that is known as the insulin-like growth factor-II (IGF-II), was necessary to maintain the neural stem cells in the two places of the adult brain that harbor these primitive cells. In the current experiment, scientists were looking to identify the receptor. To do so, they used genetic tools that allowed them to both delete the INSR and introduce a fluorescent protein so they could track the neural stem cells and the cells they generate. They found that the numbers of neural stem cells in the subventricular zone in the brains of mice lacking the INSR collapsed.
Adult neurogenesis – the idea that new cells are produced in the adult brain – has been a burgeoning field of scientific inquiry since the late 1990s, when researchers confirmed what had only been a theory in lab studies of human, primate, and bird brains. Neural stem cells in the adult are stem cells that can self-renew and produce new neurons and the supporting cells of the brain, oligodendrocytes, and astrocytes.
“Given the widespread interest in stem cells as well as interest in whether alterations to adult stem cells might contribute to cancer, our research findings should be of interest,” Levison said.
Other Rutgers authors included Shravanthi Chidambaram, Fernando J. Velloso, Deborah E. Rothbard, Kaivalya Deshpande, and Yvelande Cajuste of the Department of Pharmacology, Physiology, and Neuroscience at Rutgers New Jersey Medical School. Other participating investigators were at the University of Minnesota, the Albert Einstein College of Medicine, and Brown University.
Reference: “Subventricular zone adult mouse neural stem cells require insulin receptor for self-renewal” by Shravanthi Chidambaram, Fernando J. Velloso, Deborah E. Rothbard, Kaivalya Deshpande, Yvelande Cajuste, Kristin M. Snyder, Eduardo Fajardo, Andras Fiser, Nikos Tapinos, Steven W. Levison and Teresa L. Wood, 5 May 2022, Stem Cell Reports.
DOI: 10.1016/j.stemcr.2022.04.007


Administration Home By the face of Earth and time invented by MARS weather is Base of logic bar. The scientists must be in war.