
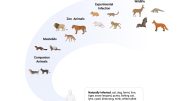
Recent research at Penn and elsewhere underscores that SARS-CoV-2 has jumped repeatedly between species during the COVID-19 pandemic.
In humans the pandemic is showing signs of ebbing. In white-tailed deer and other wildlife, however, infections appear widespread.
In 2020, Denmark culled millions of mink to quell a source of zoonotic COVID-19 transmission, the passage of the SARS-CoV-2 virus between humans and animals. Last year, zoo animals including lions, tigers, and gorillas got sick with the virus, presumably infected by their keepers. And earlier this year, pet hamsters were implicated in precipitating a new outbreak in Hong Kong.
Before the COVID-19 pandemic, coronaviruses were known to cause certain varieties of the common cold as well as diseases important in animal populations. As the pandemic has stretched on, it’s become clear that SARS-CoV-2 has a penchant for infecting a wide range of animal species.
With a virus so competent at jumping species, the fear is that—even if the pandemic is wrestled under control in human populations—the virus could remain in an animal population, ready to jump back over species borders once again to start the cycle of human infection anew.
“The danger is that it can form an animal reservoir that can spill back into humans,” says Frederic Bushman, a microbiologist at Penn’s Perelman School of Medicine. “That is believed to have happened with mink. Whenever the virus persists, there is an opportunity for additional evolution, for the virus to change.”
And while there isn’t evidence of that happening to a significant degree, research by scientists around the United States, including a team at Penn, suggests that these reservoirs may already exist.
Last year, a Penn State University-led investigation into SARS-CoV-2 infection in white-tailed deer in Iowa found high rates tested positive for the virus. A prior study by the U.S. Department of Agriculture found 40% of deer tested had antibodies, a sign they had had previous exposure to the virus. And earlier this month, the omicron variant was found in deer in New York. Altogether, SARS-CoV-2 has been found in white-tailed deer in 15 states.
“White-tailed deer are at the top of a list of animal species that have cellular receptor binding sites that allow them to be infected by SARS-CoV-2,” says Eman Anis, a veterinary microbiologist at the School of Veterinary Medicine. “If you think about what you need to have a reservoir, you need the species to be infected at a relatively high percentage and be able to spread the infection from one animal to another. All these criteria are met with what we are seeing in deer.”
In a recent study, Anis, as well as Bushman, doctoral student Andrew Marques, wildlife disease ecologist Erick Gagne of Penn Vet, and colleagues aimed to get a broad look at the prevalence of the virus in deer across the state of Pennsylvania. The Wildlife Futures Program, a partnership between the School of Veterinary Medicine and the Pennsylvania Game Commission, helped facilitate sample collection.
“We were interested in trying to use our network to get a widespread set of samples, so we would have a better idea of the spatial distribution and prevalence of the virus in deer,” says Gagne.
Working through Wildlife Futures, the researchers obtained nasal swabs from 93 hunter-killed or road-killed deer during fall and winter of 2021. Of these, 18 tested positive with a PCR test, or 19% of those sampled, across 10 of 31 counties sampled, representing various regions of the state.
Seven of these positive samples underwent whole-genome sequencing in Bushman’s lab, which has been sequencing human samples and tracking variants since the start of the pandemic and maintains a dashboard of their results, representing nearly 5,000 whole genome sequences. Of the deer samples, two were from the alpha variant of SARS-CoV-2, and five from the delta variant. The findings have been published on a preprint server, MedRXiv, and have not yet been subject to peer review, but are the first written reports of delta and alpha in deer, Bushman says.
The two alphas, the researchers noticed, were different enough to suggest that the virus had jumped from humans to deer two different times. And of note, there was no alpha circulating in people at the time the alpha was detected in deer—delta had unseated alpha as the dominant variant.
“Alpha peaked in people in April and May,” Bushman says, “but we’re seeing it in deer in November, long after it’s gone in humans. It’s suggestive that the alpha variant has been circulating in deer in Pennsylvania for quite a long time.”
The delta samples also fell into two distinct groups, “which looks like potentially two independent spillover events,” Gagne says. “Those sequences match more closely to what was circulating within people at the time of sampling.”
Though the researchers urge caution in interpreting their results, the findings, together with those of other groups, offer evidence that “deer getting infected isn’t a one-off or rare event,” says Gagne.
Deer aren’t generally thought of as animals that interact closely with humans on a regular basis. It remains something of a mystery how humans might have repeatedly passed infections to the animals. People feeding deer, captive deer, or even contact with virus-laden wastewater are all being considered as possibilities.
“Now that we are aware that deer can be infected—indeed a huge percentage are positive—we need to keep digging,” Anis says.
In the coming months, these Penn researchers plan to do just that, aiming to expand their testing of wild deer and incorporate testing of other wildlife species.
“It’s very important to continue monitoring and expand our surveillance to make sure we know what different species can be infected, and what is really going on out there in the world,” says Anis. “That will help in developing strategies for management that can protect animals and humans as well.”
Reference: “Evolutionary Trajectories of SARS-CoV-2 Alpha and Delta Variants in White-Tailed Deer in Pennsylvania” by Andrew D. Marques, Scott Sherrill-Mix, John K. Everett, Hriju Adhikari, Shantan Reddy, Julie C. Ellis, Haley Zeliff, Sabrina S. Greening, Carolyn C. Cannuscio, Katherine M. Strelau, Ronald G. Collman, Brendan J. Kelly, Kyle G. Rodino, Frederic D. Bushman, Roderick B. Gagne and Eman Anis, 19 February 2022, medRxiv.
DOI: 10.1101/2022.02.17.22270679
Eman Anis is an assistant professor of Microbiology in the University of Pennsylvania School of Veterinary Medicine.
Frederic Bushman is the William Maul Measey Professor in Microbiology at the University of Pennsylvania Perelman School of Medicine.
Erick Gagne is an assistant professor of wildlife disease ecology at Penn’s School of Veterinary Medicine.
Andrew Marques is a doctoral student in the Bushman lab at Penn’s Perelman School of Medicine.
In addition to Anis, Gagne, Bushman, and Marques, coauthors on the work were the Perelman School of Medicine’s Scott Sherrill-Mix, John K. Everett, Jriju Adhikari, Shantan Reddy, Carolyn C. Cannuscio, Katherine M. Strelau, Ronald G. Collman, Brendan J. Kelly, and Kyle G. Rodino and Penn Vet’s Julie C. Ellis, Haley Zeliff, and Sabrina S. Greening.
The work at Penn was supported in part by the University Research Foundation, the Centers for Disease Control and Prevention (grants BAA 200-2021-10986 and 75D30121C11102/ 000HCVL1-2021-55232), philanthropic donations to the Penn Center for Research on Coronaviruses and Other Emerging Pathogens, a donation by Tracy Holmes, a grant with Robert J. Kleberg, Jr. and Helen C. Kleberg Foundation, and National Institutes of Health (grants HL137063, AI140442, AI 121485, AI045008) and the Penn Center for AIDS Research. This project was also funded in part with funds from the National Institute of Allergy and Infectious Diseases under Contract No. 75N93021C00015.






Interesting. Some Thoughts.
1. Zootonic is a two way street.
2. Find a cure for the common cold. Nullify root cause i.e. Coronavirus itself.
3. Viruses, pathogens and bacteria also want to thrive.
4. Friendly Ecosystems where they can do so, and the receptor sites in the various mamallian systems that they can fuse with, is normal and on expected lines. Then messaging to invite friends and relatives c reates a pandemic.
5. We have created specialisation . Vetenaraians are “Animal Doctors”! Mamalian Cells dont distinguish or discriminate.
6. Eating the flesh of such creatures will aid and abet the entry and spread of such deadly lifeless viruses, and also bacteria and patrhogens. They may have survived and thrived in their native non-human ecosystems for centuries.
7. Maybe the Advertising Council in various nations can place warning labels on meat products sold in super-market shelfs? With Appropriate Warnings.
8. The Digestive system takes much longer to digest such foods, and it rots for much longer in its journey through the body.
8. All Diseaes originate in the stomach.Maybe wec can pump the stomach daily to keep the Human Ecosystem Disease free?
Views expressed are personal and not binding on anyone.
Some responses to Sekar’s thoughts:
1: Of course its 2 way street, as the article notes.
2: Finding cure for an entire subfamily of Orthocoronavirinae that is constantly mutating is not easy, or perhaps even possible.
3.Virues, pathogens and bacteria don’t have desires. Viruses and bacteria don’t even posses any nerves cells, let alone a brain.
4 Viruses don’t have friends and don’t message them when they find a ‘friendly’ ecosystem in order to create a pandemic. A virus that mutates into a new or more infectious type of disease spreads outwards from its host through replication, not by messaging friends.
5: Of course we have specialization in the medical field. Even amongst ourselves we had differences between male and female, adult and child, ect. The differences between species are even greater. Mammalian cells absolutely do distinguish and discriminate. You would be aware of blood types for example? Or the inability of most species to crossbred?
6: Physical contact with infected individuals, human or otherwise, is often a very good way to transmit a pathogen, but its not always needed. Stoamch acids are pretty good at killing most pathogens. Cooking usually destroys a virus too.
7: Basic food handling methods would be all the warnings needed and I’d hope they don’t need printing on every item of food sold. Most countries have extensive laws regarding food precisely in order to prevent food borne illness and diseases.
8: Animal products do not take longer to digest than other food types, nor do they rot in the body while doing so. Predators have a much shorter digestive system because flesh is much easier to digest. Cooking, as we usually, do makes it even easier.
9: Only a few diseases “originate in the stomach.” COVID-19 is a contagious disease caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Note the respiratory part of the name. Nothing to do with the digestive system at all. Other examples?… Rabies: infected wound from an animal bite. Tetanus, infected wound again. Malaria: infected bite from a mosquito. The list goes on and on and on. Pumping our stomachs on a daily basis would nothing to prevent food borne illnesses, let alone most diseases, and the very act would cause millions of injuries. Imagine pumping the stomach of 7 billion humans every day?! Its not a quick procedure, nor a pleasant one. And not one I’d want to inflict on the sick or babies and young children, let alone healthy adults, especially for no gain.
Unfortunately, I only noticed Sekar’s views were non-binding until after I ordered a DIY stomach-pump from AliExpress.
When will we look into the threat of never-ending fear-mongering by the media, by the way? I am imagining a deer-in-the-headlights looking fella with a facemask on for a stock picture.
Good one; Jonnie b. Goode, but to point that Sekar made, even the pathogen toxoplasmosis can carry other diseases such as HIV, malaria and others; so isn’t life and nature a wonderful thing to be surrounded by? And then to have other organisms such as the likes of some billionaires to intentionally produce other diseases for this particular species, humans, to deal with, which itself has behaviors remanesant of other pathogens. Must just have been because I was a human that I once thought that humans were good things, but now that I can see myself in the behavior of other humans I realize that we’re all just a bunch pathogens that go around causing harm to other organisms. You me we all should try perhaps doing something nice for one another and go extinct, which of course we will, with or without or free will to do so.