
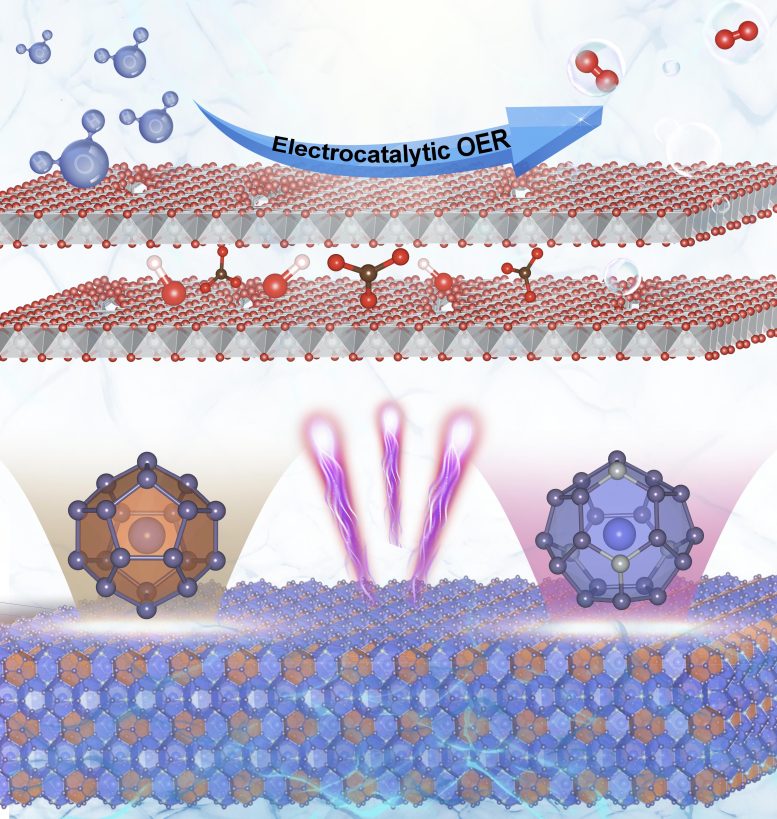
Experiments at BESSY II show that during electrolysis, the structure breaks down into ultrathin nickel sheets, exposing the active catalytic centers to the electrolyte.
Hydrogen can be produced through the electrolysis of water. When the electricity for this process comes from renewable sources, the resulting hydrogen is carbon-neutral. This “green” hydrogen is considered a key component of the future energy system and is also needed in large quantities as a raw material for the chemical industry.
Electrolysis relies on two main reactions: hydrogen evolution at the cathode and oxygen evolution at the anode (OER). However, the oxygen evolution reaction tends to slow down the overall process. To accelerate hydrogen production, researchers must develop more efficient and durable catalysts for the OER step.
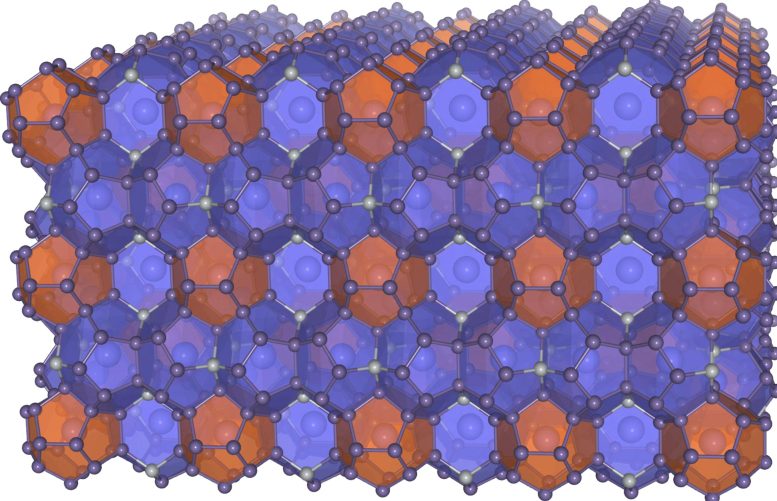
Clathrates, a structure built of cages
Currently, nickel-based compounds are regarded as effective and affordable catalysts for the alkaline oxygen evolution reaction. This is the focus of Dr. Prashanth Menezes and his team.
“The contact between the active nickel centers and the electrolyte plays a crucial role in the efficiency of a catalyst,” explains Menezes. In conventional nickel compounds, this contact surface is relatively limited. “We therefore wanted to test whether nickel-containing samples from the fascinating class of materials known as clathrates could be used as catalysts,” he says.
The materials are made of Ba8Ni6Ge40 and were produced at the Technical University of Munich. Like all clathrates, they are characterised by a complex crystalline structure of polyhedral cages, in this case, formed by germanium and nickel, enclosing barium.
This structure gives clathrates special properties that make them interesting as thermoelectrics, superconductors, or battery electrodes. However, until now, no research group had considered of investigating clathrates as electrocatalysts.
Experiments at universities and BESSY II
The electrochemical measurements showed that the Ba₈Ni₆Ge₄₀ as a catalyst exceeded the efficiency of nickel-based catalysts at a current density of 550 mA cm⁻², a value also used in industrial electrolysis. The stability was also remarkably high: even after 10 days of continuous operation, the activity did not decrease significantly.

The team used a combination of experiments to find out why the material is so remarkably well suited. At BESSY II, they studied the samples using in situ X-ray absorption spectroscopy (XAS), while basic structural characterization were carried out at the Freie and Technische Universität Berlin.
From cage to sponge
Their analysis showed that the Ba8Ni6Ge40 particles in the aqueous electrolyte undergo a structural transformation under an electric field: germanium and barium atoms dissolve out of the former three-dimensional framework.
“The germanium and barium atoms make up almost 90 % of the clathrate starting material, and they are completely washed out, leaving behind highly porous, sponge-like nanolayers of the remaining 10 % nickel that offer a maximum surface area,” says Dr. Niklas Hausmann from Menezes’ team. This transformation brings more and more catalytically active nickel centres into contact with the electrolyte.
“We were actually surprised by how well these samples work as OER catalysts. We expect that we can observe similar results with other transition metal clathrates and that we have discovered a very interesting class of materials for electrocatalysts,” says Menezes.
Reference: “Ba-Ni-Ge Clathrate Transformation Maximizes Active Site Utilization of Nickel for Enhanced Oxygen Evolution Performance” by Ziliang Chen, Hongyuan Yang, J. Niklas Hausmann, Stefan Mebs, Viktor Hlukhyy, Holger Dau, Matthias Driess and Prashanth W. Menezes, 26 March 2025, Angewandte Chemie International Edition.
DOI: 10.1002/anie.202424743
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.