
Human eggs can remain viable for decades by dialing down their internal recycling systems, new research shows.
Studying over 100 freshly donated eggs, scientists discovered that slowing protein breakdown helps minimize harmful byproducts that could damage DNA. The eggs also perform a last-minute “spring cleaning” before ovulation, ejecting waste and rearranging key components to preserve quality.
How Human Eggs Stay Dormant for Decades
Human eggs are among the body’s most enduring cells, often remaining inactive for decades until they are called upon. New research published in The EMBO Journal reveals that as these cells mature, they intentionally reduce the activity of their internal waste disposal machinery. This slowdown appears to be an evolutionary adaptation aimed at keeping metabolism low and minimizing cellular damage over the long term.
“By looking at more than a hundred freshly donated eggs, the largest dataset of its kind, we found a surprisingly minimalist strategy that helps the cells stay pristine for many years,” says Dr. Elvan Böke, corresponding author of the study and Group Leader at the Centre for Genomic Regulation (CRG) in Barcelona.
Women are born with one to two million immature eggs, a supply that gradually declines to a few hundred by the time of menopause. Each egg must remain in good condition for as long as 50 years before it has the chance to participate in reproduction. The findings shed light on how these cells manage to maintain their integrity for so long.
Waste Management: The Power-Saving Secret
Inside every cell, protein recycling is a vital housekeeping task. Lysosomes and proteasomes handle this job, breaking down old or damaged proteins. However, this process consumes energy and can produce reactive oxygen species (ROS), harmful molecules capable of damaging DNA and cell membranes. The researchers did not directly measure ROS in this study, but they propose that by reducing the pace of protein recycling, eggs limit ROS production while still carrying out enough basic maintenance to stay alive.
This discovery aligns with the team’s earlier research, published in 2022, which found that human oocytes intentionally bypass a key metabolic reaction to further cut down on ROS generation. Together, the two studies indicate that eggs employ multiple strategies to lower metabolic activity and protect themselves from damage for as many years as possible.
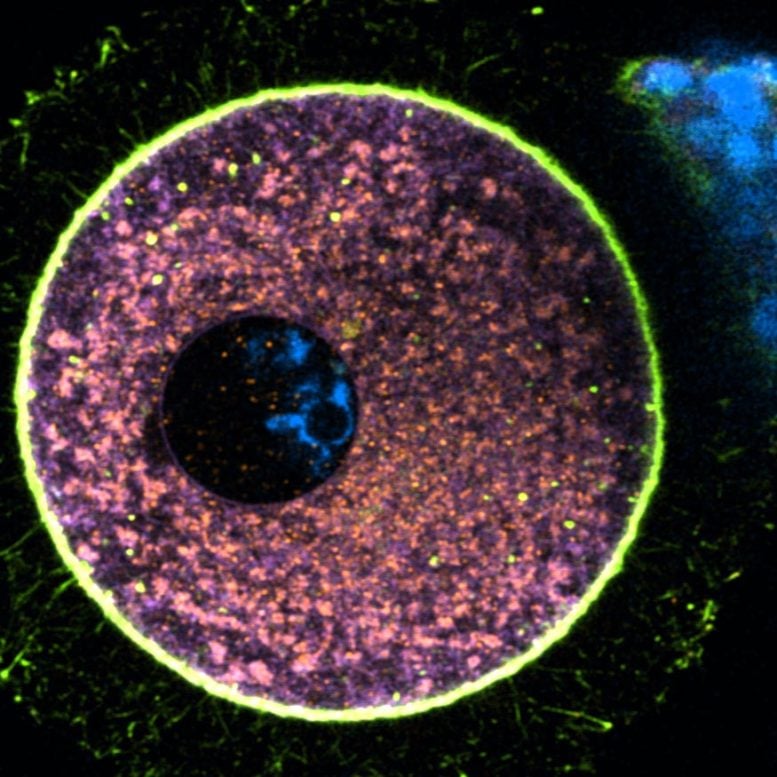
Live Imaging Unlocks Egg Behavior
The discovery was made possible by collecting over 100 eggs from 21 healthy donors aged 19–34 at Dexeus Mujer, a Barcelona fertility clinic, 70 of which were fertilization-ready eggs and 30 still-immature oocytes. Using fluorescent probes, they tracked lysosome, proteasome, and mitochondrial activity in live cells. All three readouts were roughly 50 percent lower than in the eggs’ own surrounding support cells and fell even further as the cells matured.
Live imaging showed the eggs literally jettisoning lysosomes into the surrounding fluid during the last hours before ovulation. At the same time, mitochondria and proteasomes migrated to the cell’s outer rim. “It’s a type of spring cleaning we didn’t know human eggs were capable of,” says first author Dr. Gabriele Zaffagnini.
The research is the largest-scale study of healthy human eggs collected directly from women. Most laboratory research to date has relied on eggs that have been ripened artificially in a dish, yet such in-vitro-matured oocytes often behave abnormally and are linked to poorer IVF results.
The study could lead to new strategies to improve success rates for the millions of IVF cycles attempted worldwide each year. “Fertility patients are routinely advised to take random supplements to improve egg metabolism, but evidence for any benefit for pregnant outcomes is patchy,” says Dr. Böke.
“By looking at freshly-donated eggs, we’ve found evidence to suggest the opposite approach, maintaining the egg’s naturally quiet metabolism, could be a better idea for preserving quality,” she adds.
The team now plans to examine eggs from older donors and failed IVF cycles to see whether throttling the activity of cellular waste disposal units falters with age or disease.
Reference: “The proteostatic landscape of healthy human oocytes” by Gabriele Zaffagnini, SoléMiquel, Juan Manuel Duran, Nikolaos P Polyzos and Elvan Böke, 16 July 2025, The EMBO Journal.
DOI: 10.1038/s44318-025-00493-2
Never miss a breakthrough: Join the SciTechDaily newsletter.