Discovery of ‘yoyo’ mechanism could pave the way for generating artificial oocytes in the lab.
A potential new diagnostic marker that predicts the successful and efficient development of mammalian egg cells has been identified by scientists at the Center for Genomic Regulation (CRG) in Barcelona. The discovery could pave the way for generating artificial oocytes in the laboratory, helping researchers study the causes and treatments of infertility disorders and test the impact of drugs and chemicals on women’s reproduction. The research will be published today (May 23, 2022) in The EMBO Journal.
Humans have 23 pairs of chromosomes. Between males and females, 22 pairs are shared, with the 23rd pair being the sex chromosomes. Males have an X and a Y chromosome, while females have two X’s. This causes a potential problem for the female cellular machinery, as two active X chromosomes generate an overdose of gene products, which is fatal for developing embryos or leads to cancer in adult life. To avoid this scenario, female cells inactivate one X chromosome by turning off its genes and compacting it.
Little is known about how X-chromosome inactivation affects the development of reproductive cells. In mammals, oocytes develop from germ cells, precursor cells that migrate from early embryonic tissue to the developing gonads. Germ cells then undergo meiosis, an important chromosomal rearrangement process, which is responsible for the genetic uniqueness of each individual germ cell. Germ cells mature and eventually turn into functional sperm or oocytes.
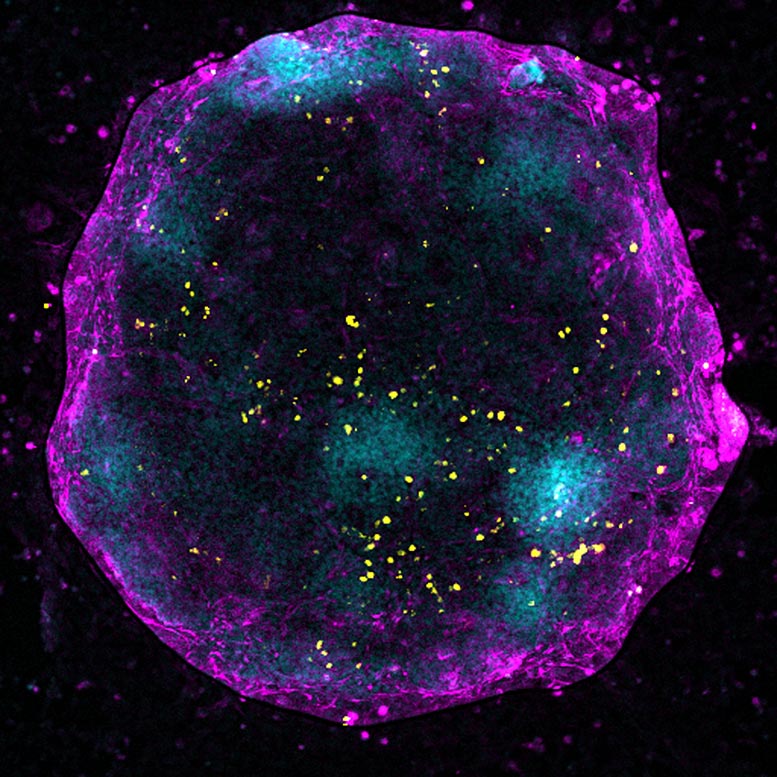
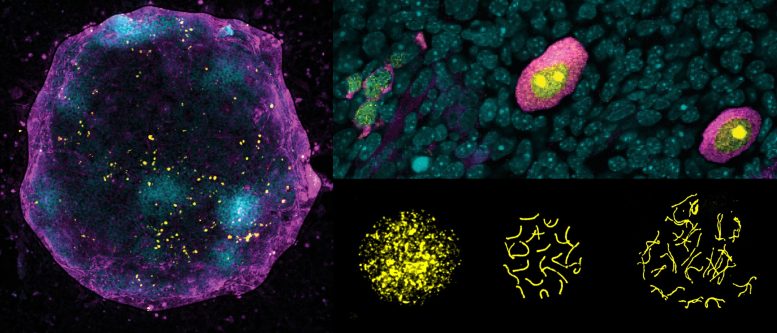
To address this question, researchers at the Centre for Genomic Regulation (CRG) built an X-chromosome reporter system (XRep), a tool that allowed them to study how the chromosome shapeshifts over time during germ cell development in vitro.
Importance of X-Chromosome ‘Yoyo’ in Germ Cell Efficiency
Using female mouse cells, the method revealed a carefully orchestrated act of X-chromosome ‘yoyo’. If one X chromosome is briefly inactivated and then reactivated, it results in germ cells being four times more efficient for entering meiosis and transforming into egg cells compared to germ cells that have never turned their X chromosome ‘off and on’ again. In comparison, germ cells that failed to inactivate the X chromosome in the first place or reactivated it too rapidly showed abnormal gene expression and cell differentiation patterns.
The study also found that cells with two active X chromosomes divided faster and easily reverted into a state of pluripotency. According to the authors, these characteristics are similar to human germ cell tumors, which come from germ cells that got lost during their migration to the gonads or failed to differentiate properly once inside the testicles and ovaries. The researchers deduced that a correct X-chromosome inactivation and reactivation sequence is an indicator of normal germ cell differentiation. The research team notes that further studies will be needed to confirm whether an abnormal X-chromosome state is a diagnostic indicator, or whether it could be a causal factor responsible for the cell abnormality.
Potential for Generating Artificial Human Oocytes
“Our findings have important implications for reproductive research because XRep allows us to assess cellular X-chromosome status in real time, helping identify and isolate germ cells with a high success rate of turning into oocytes,” says Dr. Bernhard Payer, Group Leader at the CRG and senior author of the study.
“Human oocytes have never been generated completely in vitro. Monitoring the X-chromosome state during in vitro germ cell differentiation might therefore be a way to optimize the protocol to produce high-quality human eggs in the lab. Human eggs for research are scarce and difficult to obtain because they are currently only available from egg donors and are mostly used for reproductive purposes. In vitro-generated human eggs could therefore provide an unlimited resource to study the causes and treatments of infertility disorders and as well to test the safety of drugs and chemicals for women’s reproduction,” concludes Dr. Payer.
Dr. Moritz Bauer, co-first author of the study, adds: “Our results also highlight how we need specific tools to study female cells. The vast majority of studies are performed using male cells, leading to a gender gap in scientific knowledge. We, therefore, need to stop looking at female development through the lens of male cells, which will contribute to our understanding of sex-specific disease progressions.”
Reference: “Controlled X-chromosome activation dynamics defines meiotic potential of female mouse in vitro germ cells” by Jacqueline Severino, Moritz Bauer, Tom Mattimoe, Niccolò Arecco, Luca Cozzuto, Patricia Lorden, Norio Hamada, Yoshiaki Nosaka, So I Nagaoka, Pauline Audergon, Antonio Tarruell, Holger Heyn, Katsuhiko Hayashi, Mitinori Saitou and Bernhard Payer, 23 May 2022, The EMBO Journal.
DOI: 10.15252/embj.2021109457
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.