
Promising 2D materials may face an unexpected obstacle at the atomic scale: a tiny gap at their interfaces.
For decades, the steady shrinking of electronic components has powered faster, more efficient technology. Engineers now hope to push that trend even further with 2D materials, atomically thin sheets that promise unprecedented control at the smallest scales.
But a new study from TU Wien shows that many of these once promising materials may not work as expected. The issue is not just the material itself. It is also how it interacts with other components. When a 2D material is paired with an insulating layer, a tiny gap forms between them, significantly reducing performance. Identifying which materials avoid this problem could help the semiconductor industry steer clear of costly dead ends.
It’s not just the material—it’s the interface
“For many years, researchers have quite rightly been fascinated by the remarkable electronic properties of novel 2D materials such as graphene or molybdenum disulfide,” says Prof. Mahdi Pourfath, who carried out the research together with Prof. Tibor Grasser at TU Wien’s Institute for Microelectronics. “What is often overlooked, however, is that a 2D material alone does not make an electronic device. We also need an insulating layer—usually an oxide. And this is where things become more complicated from a materials science perspective.”
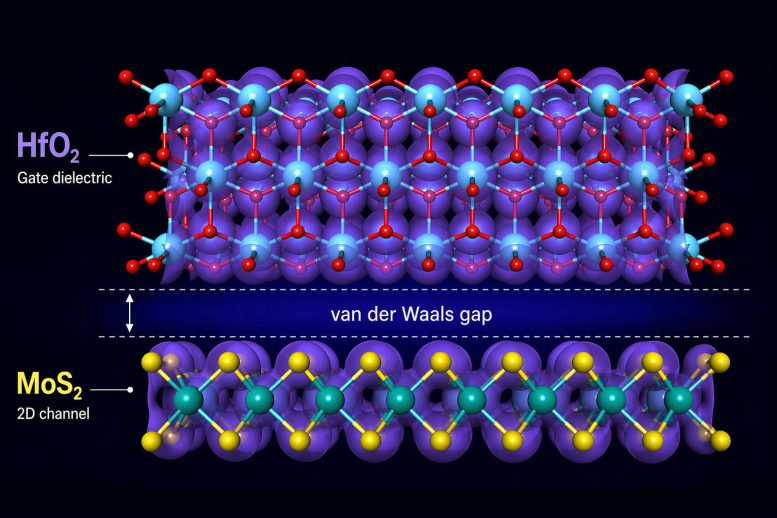
At their core, transistors operate by switching a semiconductor between conducting and nonconducting states. In this case, the semiconductor can be a 2D material. A gate electrode controls this switching, but it must be separated from the material by an insulating layer.
Mind the gap!
To achieve precise control and allow for extremely compact devices, this insulating layer must be as thin as possible. However, atomic-scale analysis reveals a problem that has often been overlooked.
“In many combinations of 2D materials and insulating layers, the bonding between them is relatively weak,” explains Grasser. “They are held together only by so-called van der Waals forces, which provide only a weak attraction between the semiconductor and the insulator. As a result, the two layers do not come into close contact—there is always a gap between them.”
This gap is incredibly small, about 0.14 nanometers (0.0000000055 inches), thinner than a single sulfur atom, yet it has a major effect on performance. For comparison, a SARS-CoV-2 virus is about 700 times larger. “This gap weakens the capacitive coupling between the layers. No matter how good the intrinsic properties of the materials may be, the gap can become the limiting factor. As long as it exists, it imposes a fundamental limit on how far these devices can be miniaturized.”
The solution: “zipper” materials
“If the semiconductor industry wants to succeed with 2D materials, the active layer and the insulating layer must be designed together from the very beginning,” emphasizes Mahdi Pourfath. One potential solution is the use of “zipper materials,” in which the semiconductor and insulator interlock. Instead of being loosely held together by van der Waals forces, they form a stronger bond that removes the gap.
“Our work is good news for the semiconductor industry,” says Tibor Grasser. “We can predict which materials are suitable for future miniaturization steps—and which are not. But if one focuses only on the 2D materials themselves, without considering the unavoidable insulating layers from the outset, there is a risk of investing billions in an approach that simply cannot succeed for fundamental physical reasons.”
Reference: “Device-scaling constraints imposed by the van der Waals gap formed in two-dimensional materials” by Mahdi Pourfath and Tibor Grasser, 16 April 2026, Science.
DOI: 10.1126/science.aeb2271
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
THE FOLLOWING DUE TO THEORIES N THE DISCOVERIES TO COMPLETE THE PYTHEPOEM THEORY IS REVISED FORTHE NEW WORLD FOR ALL THE MORE INCREASED THEORIES ARE VALID INCLUDES A SIMPLE REPETIVE COMMUNICATIONS VARIOUS INCRASED RESULTS I (SCIENCE/MATH/HISTORY/RELIGION/WORLD-LANGUAGES & TELECOMUNICATIONS(WORLD-TECHNOLOGY)INTO THESE COMPRENSIONS ARE DESCRIPTIONS NOT ISSUES ARE DEFINED AREAS OF ALL OF THESE COLLECTONS ARE DEFINED AND REVISED INTO THESE QUESTIONS ARE VALID.
More punctuation, maybe!
Can’t name it Bloch hand-waving v. smol.
Add another layer having the same gap before removing the resine.
Connect the top layer using chemistry and electricity on a side and the other of the gap.
Add your isolation layer leaving a lower layer gap.
(Removing the resine after adding the isolation layer may not work…)
No discrimination over names (not bad mahdi !). With my best wishes for no discrimination over color.
‘THE NEW TYPES OF”PHASES/(BIO-TECHNOLOGY)’; ANY THEORTCIAL-STATUS INTO THE FOLLOWING ISSUES ARE INTO THESE INCREASED AMOUNTS OF “ALL EMPHASIS-COORDINATIONS” : A CONCEPT OF THREE NEW STATES (HOT)(COLD)(NEUTRAL) EACH ELEMENTS INCLUDES ISOTOPES/IONIC BOND(CELL-CONTAINS ARE NEURAL NOT THE NEW EVIDENT-CIRCUMSTANCES OF THE THREE ATOME OF POSTIVE & NEGATIVE CHARGES ARE PROVEN TO EXCEED THE LIMITS OF ALL SERIES INTO:”A SIMPLE TYPES OF CHANGES A FORMS EACH QUESTIONS A RATIO(S) INTO THESE FOLLOWING FACTORS: A XCOMPLETED THOUGHT -PROCESS COMMON ISSUES IN COLLECTIONS ARE (BIO-/TRI-/ANTI/)THEORIES ARE NEW ELEMENTS ARE ‘NEVER-THE-LESS’INTO ALL PROCESS IN THE FOLLOWING INTO THE FEATURES DUE TO ANAYLSIS OFTEN REALITY THE FEATURES NOT VIRTUES ARE NOT (-)/(+) ARE REVERSED CREATES A SIMPLE S “MATHAEMATICAL-COMPUATIONS”THEORTICAL CONCEPTS” ARE DEVELOPEMENTAL ISSUES OF ” 2″-STATES OF ((BIO-LIQUID)) INTO THE VIRTUES OF SEQUENCES INTO THEORIES INTHE LEVELS OF GENERILZATIONS ARE INTO RE- “VISIONS NOT FANATASY IN “THE VALUES OF THE CONCEPTS ARE ((MOLECULAR-BIOLOGY))”ANY CONCERNS :A WORLD INTO THE FOLLOWING COMPREHENSIONS ARE SERIES INTO THESE AREAS (BIO)”EMPHASIS)(BIO-TECHNOLOGY) ARE SUBJECTS COMMUNATIONS-LEVELS INCREASED BY VALUES (“quoted) AND REVISED INTO THESE COLLECTIONS OF THEORIES IN(BIO-VIRTUES CONCEPTS ACCUMLATES A SOURCES INTO THE FOLLOWING ISSUES AND SERIES ARE COMBINATIONS REVERSE SEQUENCES ARE SIMPLE INTO THE FEATURES-REVERSED INNER-CODES NONE-THE LESS INTO COLLECTION BECOME DEVISE AND SERIES ARE COMPREHENSIONS ARE QUESTIONS WITHOUT COMPLETED THEORIES & WIHTOUT REVERSED QUESTIONS OPPOSITES SIMPLE REVERSED INTO ISSUES OF COLLECTIONS