Researchers have successfully regenerated skeletal and connective tissue, although the new tissue was not perfectly formed. The result demonstrates a critical step forward in limb regeneration.
For centuries, scientists have viewed the inability to regrow lost body parts as a major biological limit for humans and other mammals. Salamanders and some other animals can regenerate entire limbs, but people usually heal serious injuries by forming scar tissue.
New research from the Texas A&M College of Veterinary Medicine and Biomedical Sciences (VMBS) suggests that this limitation may not be absolute. The ability to regenerate tissue may still exist in mammals, but it may be hidden within the body’s ordinary healing response.
“Why some animals can regenerate and others, particularly humans, can’t is a big question that has been asked since Aristotle,” said Dr. Ken Muneoka, a professor in the VMBS’ Department of Veterinary Physiology & Pharmacology (VTPP). “I’ve spent my career trying to understand that.”
In a study published in Nature Communications, Muneoka and his colleagues describe a new two-step treatment that triggered the regrowth of bone, joint structures, and ligaments. The regenerated tissues were not perfectly formed, but the researchers say the method could have near-term value for reducing scar formation and improving tissue repair after amputations.
Redirecting the body’s natural response
When mammals are injured, the body usually responds through fibrosis. In this process, fibroblast cells quickly seal the wound and create scar tissue. That rapid closure helps protect the body, but it also prevents missing structures from being rebuilt.
In animals that can regenerate, such as salamanders, similar cells gather into a blastema. This temporary structure acts as a foundation for new tissue growth.
“It’s as if these cells can move in two different directions,” Muneoka said. “They could either make a scar or make a blastema. Our research focused on redirecting the behavior of fibroblasts already present at the injury site.”
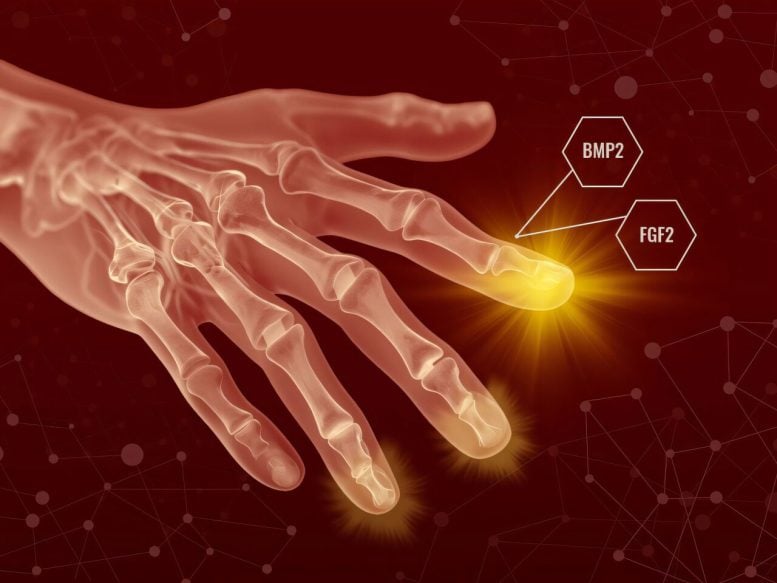
To find out whether mammalian healing could be pushed toward regeneration, the researchers created a sequential treatment using two growth factors that are already well studied.
The first step was to apply fibroblast growth factor 2 (FGF2) after the wound had closed. This allowed the body to finish its usual healing process before the researchers “changed what happens next,” Muneoka said.
FGF2 encouraged the formation of a blastema-like structure, which normally does not appear in mammals after this kind of injury. Several days later, the researchers applied a second treatment, bone morphogenetic protein 2 (BMP2), which prompted those cells to begin building new tissue structures.
“This is really a two-step process,” Muneoka said. “You first shift the cells away from scarring, and then you provide the signals that tell them what to build.”
Challenging assumptions about regeneration
One major takeaway from the study is that regeneration may not require adding outside stem cells, a common goal in many regenerative medicine strategies.
“You don’t have to actually get stem cells and put them back in,” Muneoka said. “They’re already there — you just need to learn how to get them to behave the way you want.”
Dr. Larry Suva, a VTPP professor who contributed to the study, said the work changes how scientists view the boundaries of mammalian healing.
“The cells that we thought to be unprogrammable, in fact are,” Suva said. “The capacity is not absent — it’s just obscured.”
The researchers also found that cells could be redirected to form structures outside their original position. This process, known as positional re-specification, is important during development.
In practical terms, that means cells that would normally help form one body region can be guided to rebuild a different structure after injury.
Imperfect but complete regrowth
The regenerated structures did not perfectly match the original anatomy. Even so, the researchers were able to restore all the main components removed during amputation, including bone, tendon, ligament, and joint tissue.
The regrowth included both skeletal and connective tissues arranged in a way that reflected the natural structure.
“We regenerated what you would expect to see at that level of injury,” Muneoka said. “The structures are there — just not in a perfect form.”
The study also showed that regeneration depends on several biological pathways rather than one simple mechanism, suggesting that rebuilding tissue is a more complex process than activating a single switch.
Potential applications in human healing
The work is still at an early stage, but it could have more immediate relevance for improving wound repair.
Instead of aiming first to regrow entire body parts, the researchers think the approach may initially help reduce scarring and promote better tissue healing.
“People should start thinking about using these signals during the healing process,” Muneoka said. “Even shifting the response slightly away from scarring could have real benefits.”
Because BMP2 is already FDA-approved for some medical uses, and FGF2 is being tested in multiple clinical trials, the path toward clinical investigation may be more accessible than it would be for entirely new treatments.
A new direction for regenerative medicine
The study points to a different way of thinking about mammalian regeneration. Rather than being completely lost, the ability may still be present but inactive.
“This changes the way we think about what’s possible,” Suva said. “Once you show that regeneration can be activated, it opens the door to asking entirely new questions.”
For Muneoka, those questions have shaped decades of research and now have a stronger experimental basis.
“Regenerative failure in mammals can be rescued,” he said. “Now we have a model to begin figuring out how.”
Reference: “Digit regeneration in mice is stimulated by sequential treatment with FGF2 and BMP2” by Ling Yu, Mingquan Yan, Katherine Zimmel Scaturro, Osama Qureshi, Yu-Lieh Lin, Benjamin B. Bartelle, C. Addison Smith, Daniel Osorio Hurtado, James J. Cai, Lindsay A. Dawson, Regina Brunauer, Larry J. Suva, Manjong Han, Connor P. Dolan and Ken Muneoka, 17 April 2026, Nature Communications.
DOI: 10.1038/s41467-026-72066-8
The research is funded by W911NF-06-1-0161 from DARPA to KM, W911NF-09-1-0305 from the US Army Research Center to KM, R01HD116825 to CPD, R01AG081812 to LAD, the John L. and Mary Wright Ebaugh Endowment Fund at Tulane University to KM, and Texas A&M University to KM.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
12 Comments
I’ve always wondered why my hands can heal from cuts w/o scars but other parts of my body do not have this capability. I have cut my hands many times over my decades and yet when I look at my hands, there is no scarring from any of the cuts.
Please email me about regenerative medication to stop fibrosis and then to generate proper cells in the liver because I have hep b, and stage one fibrosis and I am taking vemlidy daily, and I heard that TheraVacB wil be available in America in 2028
HELLO EDITOR OF THIS ARTICLE…THIS MAY HINT AT THE “HAND GESTURES AND FINGER MUDRAS OF THE BUDHHISTS AND CHINESE PRACTICES TOWARDS SELF HEALING IF DONE MINDFULLY,ALSO A HINT TOWARDS REIKI HEALING.
JUST A THOUGHT PROVOKNG ARTICLE.
Very interesting, I am too old to benefit from this but wish the best to Moneuka and others like him
We have forgotten how to use our inner resources
I have clearly demonstrated this time and time again
At that time the eyes of the blind will be opened, And the ears of the deaf will be unstopped. 6 At that time the lame will leap like the deer, And the tongue of the speechless will shout for joy. For waters will burst forth in the wilderness, And streams in the desert plain. Isa. 35
Under God’s Kingdom. Hate to disappoint all the sciencers…
Don’t forget that the next time you go to the hospital.
So in essence are they saying that if the scab was kept from forming that more of the wound (and I am thinking road rash to a really bad cut) would regenerate correctly leaving less scar tissue? That could be promising for those injuries in places like on joints etc. which can also be painful because of the amount of scar tissue.
So in essence are they saying that if the scab was kept from forming that more of the wound (and I am thinking road rash to a really bad cut) would regenerate correctly leaving less scar tissue? That could be promising for those injuries in places like on joints etc. which can also be painful because of the amount of scar tissue.
Why did you type it twice bro?
maybe this will all occur through the work of the scientists. The solution exists/ they just have to find it..
I have clearly demonstrated this time and time again