Gut bacteria brew all sorts of chemicals, but we don’t know what most of them do. A new study suggests that one such compound, previously linked to cancer, may serve as a bizarre weapon in microbial skirmishes.
Some gut bacteria have a spooky superpower: they can reanimate dormant viruses lurking within other microbes.
This viral awakening unleashes full-blown infections that destroy the virus-carrying cells, Howard Hughes Medical Institute Investigator Emily Balskus’s lab first published as a preprint on bioRxiv.org and later in the journal Nature on February 23, 2022. A cryptic molecule called colibactin can summon the killer viruses from their slumber, they found.
Microbes often generate noxious compounds to attack one another within the cramped quarters of the gut. But among these chemical weapons, colibactin appears unusual, says Balskus, a chemical biologist at Harvard University. “It doesn’t directly kill the target organisms, which is what we normally think of bacterial toxins doing within microbial communities.” Instead, colibactin tweaks microbial cells just so, activating latent – and lethal – viruses tucked away in some bacteria’s genomes.
Humans have long sought out the potent compounds that microbes produce. “We know a lot about their chemical properties, we purify them in the lab, and we use them as medicine, including antibiotics,” says Breck Duerkop, who studies bacterial viruses at the University of Colorado School of Medicine.
But why bacteria make these compounds and what effects they have on neighboring organisms are open-ended questions, says Duerkop, who was not involved in this research. He calls Balskus’s team’s new work “one step in the right direction.”
Chemical Dark Matter
Scientists have known for years that colibactin can wreak havoc on human cells. Research by Balskus and many others has shown that the compound damages DNA, which can lead to colorectal cancer. But establishing a connection between this compound and disease proved particularly formidable.
In 2006, a French team reported that mammalian cells that encountered the gut bacteria E. coli suffered fatal damage to their DNA. The researchers linked this damage to a cluster of E. coli genes encoding machinery for building a complex molecule. Dubbed colibactin, the molecule was extraordinarily difficult to study. After many tries, researchers simply couldn’t isolate it from the E. coli making it.
Colibactin is one of many ephemeral compounds that scientists suspect microbes make. Like invisible particles of dark matter in space, this “chemical dark matter” requires creative means to study. As part of her exploration of the gut’s microbial chemistry, Balskus uses indirect approaches to examine these elusive molecules.
Over the past 10 years, her team has probed colibactin by studying the microbial machinery that manufactures it. She and her colleagues have pieced together colibactin’s structure and determined that it damages DNA by forming errant connections within the double helix.
Building off this work, scientists elsewhere uncovered a definitive link to cancer: the molecule’s distinctive fingerprints appear in genes known to drive colorectal tumor growth.
A Role for Viruses
Balskus’s most recent colibactin study got its start with another disease: COVID-19. Like many other labs, hers had to rearrange things to reduce physical contact among researchers. As part of the reshuffling, postdoc Justin Silpe and graduate student Joel Wong ended up working near one another for the first time. Their conversations led them and Balskus to wonder how colibactin affected other microbes in a crowded gut.
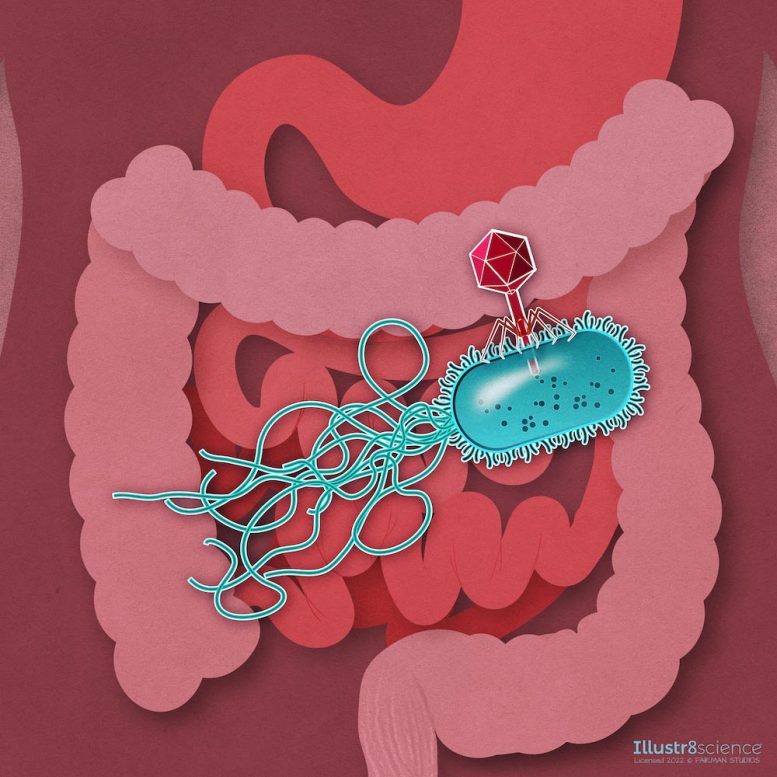
Early on, they found that exposing colibactin-producing bacteria to non-producers had little effect, suggesting that, on its own, the molecule isn’t particularly deadly. Silpe and Wong weren’t sure if colibactin, a large, unstable molecule, could even enter bacterial cells to damage their DNA. They wondered if a third party — bacteria-infecting viruses — might be involved. Hardly more than bits of genetic information, these viruses can slip into bacteria’s DNA and lie quietly in wait. Then, once triggered, they cause an infection that blows up the cell like a landmine.
“We always suspected that bacteria made this toxin to target other bacteria in some way.”
Emily Balskus, HHMI Investigator at Harvard University
When the researchers grew colibactin producers alongside bacteria carrying such latent viruses, they saw the number of viral particles spike, and the growth of many virus-containing bacteria drop. That suggested the molecule sparked a surge in active, cell-killing infections. Colibactin does actually enter bacteria and damage DNA, the team showed. That damage sounds a cellular wake-up bell that rouses the viruses.
Many microbes appeared equipped to protect themselves against colibactin. Balskus’s lab identified a resistance gene encoding a protein that neutralizes the compound in a wide variety of bacteria.
Though colibactin clearly has a dangerous side, it may serve as more than just a lethal weapon, Balskus says. For example, both DNA damage and awakened viruses can also induce genetic changes, rather than death, in neighboring bacteria, potentially benefiting colibactin producers.
Balskus’s team’s discoveries suggest that cancer may be collateral damage caused by whatever else colibactin-producing bacteria are doing. “We always suspected that bacteria made this toxin to target other bacteria in some way,” she says. “It didn’t make sense from an evolutionary perspective that they acquired it to target human cells.”
Next, Balskus plans to investigate how the compound alters the community of microbes in the gut — which ones disappear and which thrive after exposure to the compound. “The key to preventing cancer may be understanding the effects colibactin has on the microbe community and how its production is controlled,” she says.
Reference: “The bacterial toxin colibactin triggers prophage induction” by Justin E. Silpe, Joel W. H. Wong, Siân V. Owen, Michael Baym and Emily P. Balskus, 23 February 2022, Nature.
DOI: 10.1038/s41586-022-04444-3
bioRxiv
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
This should all point to the obvious that we originated from bacteria.