Proteins communicating through the DNA molecule constitute a newly discovered genetic “switch.”
Proteins can communicate through DNA, conducting a long-distance dialogue that serves as a kind of genetic “switch,” according to Weizmann Institute of Science researchers. They found that the binding of proteins to one site of a DNA molecule can physically affect another binding site at a distant location, and that this “peer effect” activates certain genes. This effect had previously been observed in artificial systems, but the Weizmann study is the first to show it takes place in the DNA of living organisms.
A team headed by Dr. Hagen Hofmann of the Chemical and Structural Biology Department made this discovery while studying a peculiar phenomenon in the soil bacteria Bacillus subtilis. A small minority of these bacteria demonstrate a unique skill: an ability to enrich their genomes by taking up bacterial gene segments scattered in the soil around them. This ability depends on a protein called ComK, a transcription factor, which binds to the DNA to activate the genes that make the scavenging possible. However, it was unknown how exactly this activation works.

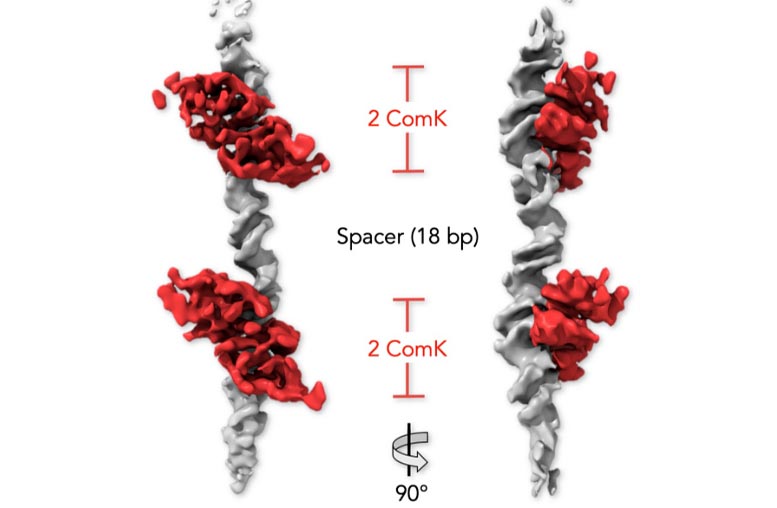
Staff Scientist Dr. Gabriel Rosenblum led this study, in which the researchers explored the bacterial DNA using advanced biophysical tools – single-molecule FRET and cryogenic electron microscopy. In particular, they focused on the two sites on the DNA molecule to which ComK proteins bind.
DNA Transmits Physical Signals Like a Communication Cable
They found that when two ComK molecules bind to one of the sites, it sets off a signal that facilitates the binding of two additional ComK molecules at the second site. The signal can travel between the sites because physical changes triggered by the original proteins’ binding create tension that is transmitted along the DNA, something like twisting a rope from one end. Once all four molecules are bound to the DNA, a threshold is passed, switching on the bacterium’s gene scavenging ability.
“We were surprised to discover that DNA, in addition to containing the genetic code, acts like a communication cable, transmitting information over a relatively long distance from one protein binding site to another,” Rosenblum says.
Specific Conditions for Signal Transmission
By manipulating the bacterial DNA and monitoring the effects of these manipulations, the scientists clarified the details of the long-distance communication within the DNA. They found that for communication – or cooperation – between two sites to occur, these sites must be located at a particular distance from one another, and they must face the same direction on the DNA helix. Any deviation from these two conditions – for example, increasing the distance – weakened the communication. The sequence of genetic letters running between the two sites was found to have little effect on this communication, whereas a break in the DNA interrupted it completely, providing further evidence that this communication occurs through a physical connection.
Knowing these details may help design molecular switches of desired strengths for a variety of applications. The latter may include genetically engineering bacteria to clean up environmental pollution or synthesizing enzymes to be used as drugs.
“Long-distance communication within a DNA molecule is a new type of regulatory mechanism – one that opens up previously unavailable methods for designing the genetic circuits of the future,” Hofmann says.
Reference: “Allostery through DNA drives phenotype switching” by Gabriel Rosenblum, Nadav Elad, Haim Rozenberg, Felix Wiggers, Jakub Jungwirth and Hagen Hofmann, 20 May 2021, Nature Communications.
DOI: 10.1038/s41467-021-23148-2
The research team included Dr. Nadav Elad of Weizmann’s Chemical Research Support Department; Dr. Haim Rozenberg and Dr. Felix Wiggers of the Chemical and Structural Biology Department; and Jakub Jungwirth of the Chemical and Biological Physics Department.
Dr. Hagen Hofmann is the incumbent of the Corinne S. Koshland Career Development Chair in Perpetuity.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
The facts are facts, but the interpretation is a bit SciFi. There is no DNA-molecule “communication” in this case, but a well-know structural change interpretation. If there are 4 protein binding sites in a relatively moderate proximity (i.e., within up to ~5000 base-pairs), all 8 DNA sites (4×2 as 1 for each DNA strand) have the ability to self-pair as well as most-likely can form complex 3-4-more strands binding complexes. As normal, these pairings “dissolve” when proteins are bound utilizing the same hydrogen bonds that thereby leads to more or all protein binding-sites become available to protein attachment. That’s all – no long-distance “communication” in fact and nothing uncommon other than what are known principles for the past 40 years.