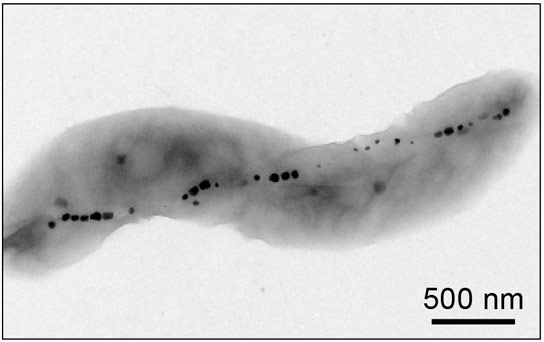
A typical electron microscope image of a bacterium; magnetic nanoparticles inside the bacterium appear as black spots. A new technique has been developed that is capable of studying living cells at comparable spatial resolutions by taking advantage of the presence of these magnetic particles. Credit: Nature
In a new study, researchers present a method for resolving the magnetic structure of living biological specimens at a sub-cellular level.
Magnetic field measurement techniques have long enabled scientists to probe the internal structure of biological and material samples. For example, magnetic resonance imaging (MRI) provides information about the structure and function of tissue inside opaque biological specimens. A current limitation of such biologically compatible magnetic imaging techniques is that their resolution is insufficient to resolve the internal structure of cells, which are instead typically studied using optical or electron microscopes.
In the current issue of the journal Nature, Harvard-Smithsonian Center for Astrophysics (CfA) scientists David Le Sage, David Glenn, and Ron Walsworth, together with their collaborators, present a method for resolving the magnetic structure of living biological specimens at a sub-cellular level. In their study, they use a particular variety of bacteria that naturally produces an internal chain of magnetic nanoparticles. They place these live bacteria onto a diamond surface that has been modified to contain crystal defects that interact with magnetic fields and light. By shining a laser beam onto the surface and measuring the pattern of the light emitted by the defects using an optical microscope, they are able to record images of the magnetic field pattern present at the diamond surface with a spatial resolution that is on the order of the wavelength of light. The technique is a spinoff from the group’s laser technology research on behalf of astronomical research, including exoplanet detection and radio interferometry.
These first results demonstrate the promise of the technique to probe magnetically the internal structure of living cells. This technique could be used to shed new light on the properties and life cycles of these magnetic bacteria, and it points the way toward more sophisticated probes of a wide range of other biologically interesting systems.
Reference: “Optical magnetic imaging of living cells” by D. Le Sage, K. Arai, D. R. Glenn, S. J. DeVience, L. M. Pham, L. Rahn-Lee, M. D. Lukin, A. Yacoby, A. Komeili and R. L. Walsworth, 24 April 2013, Nature.
DOI: 10.1038/nature12072








Be the first to comment on "Magnetic Imaging of Living Cells with Sub-Cellular Spatial Resolution"