
The nanozymes hypothesis proposes that mineral nanoparticles drove chemical evolution on early Earth, helping transform inert matter into life through catalytic and environmental processes.
The formation of the first biopolymers on early Earth is widely seen as a critical step in the origin of life (OoL). However, exactly how life emerged from simple, nonliving chemicals (gases) remains unclear. Reconstructing these early processes is challenging, which makes it difficult to determine a complete and accurate explanation.
Over the past century, scientists have proposed many ideas to explain the OoL, most of which focused on chemical evolution on Earth or in space. Despite this progress, these theories remain incomplete and often controversial because they rely on limited experimental evidence or specific theoretical assumptions.
Examples include the metabolism-first world (FeS world), zinc world, thioester world, RNA world, and lipid world. Each provides insight into certain steps of early life formation, but none offers a complete explanation. A single, unified model that explains how life could arise from simple chemicals has not yet been established.
A New Unified Idea: The Nanozymes Hypothesis
To address these gaps, Prof. Yongdong Jin of Shenzhen University proposed the “nanozymes hypothesis.” This idea suggests that natural mineral nanozymes (MN-zymes), along with later hybrid versions formed with small organic molecules, played a central role in the emergence and evolution of life, especially during its earliest stages.
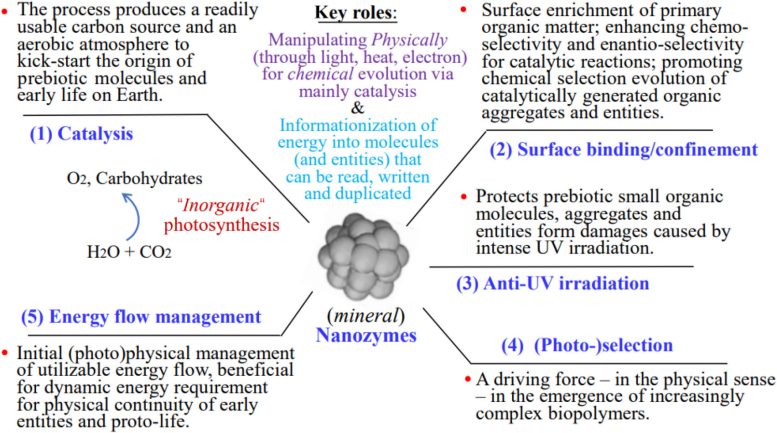
According to this hypothesis, these nanozymes helped convert simple, nonliving chemicals into early biological molecules through complex chemical and physical processes. A key mechanism is described as “inorganic photosynthesis” under early Earth conditions.
The model proposes that MN-zymes performed several essential functions, including catalysis, molecular binding and confinement, protection from ultraviolet radiation, selective reactions driven by light, and regulation of energy flow. Through these roles, they supported chemical reactions and helped organize energy into molecular forms that could be stored, copied, and used, which are fundamental requirements for life.
Earth as a Natural Chemical Laboratory
This perspective views Earth itself as a large, self-sustaining chemical system capable of generating life from entirely inorganic conditions, similar to earlier ideas about abiogenesis. In this framework, the planet functions as an “all-in-one” natural laboratory.
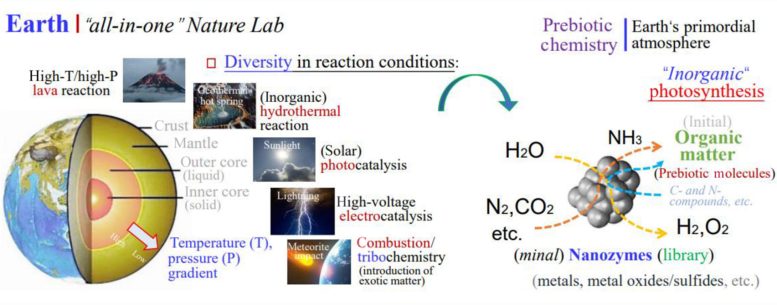
Natural variations in temperature and pressure across Earth’s interior, especially near volcanoes and geothermal systems, create ideal conditions for forming nanozymes through high-temperature and high-pressure reactions. These processes resemble methods currently used in laboratories to produce artificial nanozymes.
Over time, these early nanozymes may have evolved and self-renewed. Some eventually became incorporated into living systems, influencing both biological development and gradual changes in Earth’s environment. These changes, in turn, supported further chemical evolution and early life.
Abundance and Formation of Natural Nanozymes
Mineral nanoparticles are widespread on Earth. Each year, thousands of terragrams (Tg) (1 Tg = 1012 g) of these particles circulate through ecosystems. Many display enzyme-like behavior and are found in oceans, soils, the atmosphere, and freshwater systems, where they play key roles in biogeochemical cycles.
Recent findings suggest that nanozymes can form more easily than previously thought. They may develop through processes such as mineral weathering in charged water microdroplets or exposure to ultraviolet light. Sunlight and lightning can also drive reactions that produce both nanozymes and a wide range of prebiotic molecules on Earth’s surface.
The “Au World” and Specialized Nanozyme Roles
The hypothesis also highlights a potential role for gold nanoparticles (AuNPs), referred to as the “Au world.” These particles may have been especially effective nanozymes and contributed significantly to early chemical evolution, even though their importance has largely been overlooked.
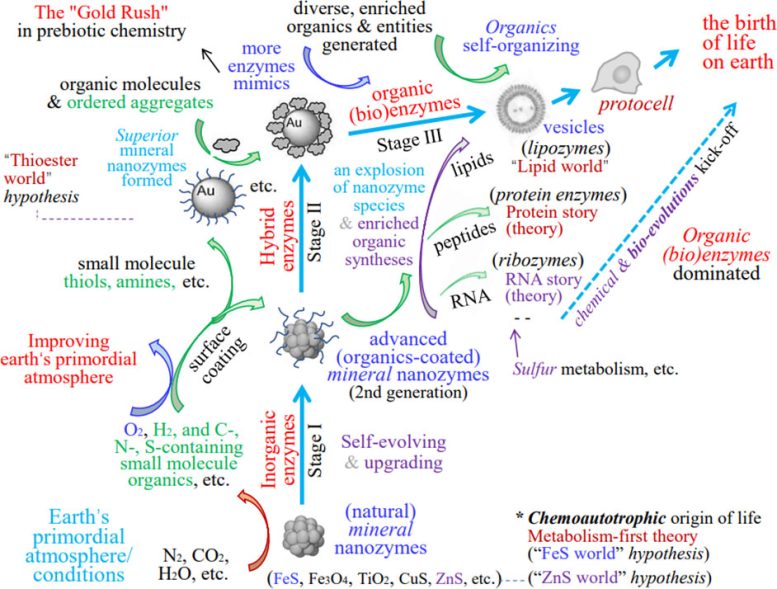
Although free AuNPs are unlikely to remain stable without protective coatings, they could exist on mineral surfaces or in stabilized forms once small organic molecules such as thiols and amines became available. These conditions may have allowed them to participate in early chemical processes.
Key Conditions for Life and Broader Implications
The hypothesis also outlines several essential conditions for the emergence and survival of early life-related molecules. These include wet–dry cycles, amphiphilic behavior, self-assembly and organization, catalytic activity similar to protoenzymes, and stabilizing interactions between molecules.
Beyond nanozymes, the framework addresses broader questions about the origin of life. These include the role of water and surface structures, cooperation and co-evolution among early molecules, and physical factors such as the origin of molecular chirality.
Overall, the nanozymes hypothesis aims to resolve long-standing questions about how life began on Earth. It offers a more integrated explanation and may encourage further research into the role of nanozymes in early biological evolution.
Reference: “On the Origin of Life on Earth: The Nanozymes Hypothesis, and More” by Yongdong Jin, 9 December 2025, Research.
DOI: 10.34133/research.1025
This study was funded by the National Natural Science Foundation of China.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
5 Comments
The flintstones hypno
Abeogenesis should consider the fact that life is heat engine that works due to the temperature difference between the surface of Earth and the Cosmic Background Radiation. When Earth was colder than BCR, nanozymes would have caused formation of biomolecules that store energy. By the time, temperatures became equal, a huge build up of biomass would have formed. When BCR became sufficiently cold, life, that is, biomolecules that can replicate itself using stored energy, would have emerged in thousands of locations. Emergence of life is deterministic. Chance acts as spoiler. My paper is available at academia.edu.
1st of all the universe consists of physics laws chemical rxns etc among chemistry in the universe u shud stop looking at life as the grandest in the universe, frm the beginning of the universe there jv been multitude of chemical rxns and what happened on earth 2 4m life is just one of the multitude of chemical rxns that occur so these chemical rxns will continue 2 occur indefinitely in the universe and 4m different things(outcomes)
What is a terragram and why is it 1012 grams?
nice