The enzyme complex ATP synthase is one of the most important molecular machines in the cell. It produces adenosine triphosphate (ATP), which serves as the energy source for almost all essential cellular processes. ATP production and availability can also be useful for biotechnological processes outside the cell. Researchers from the Max Planck Institute of Biophysics in Frankfurt, the Ludwig Maximilian University (LMU) in Munich, and the Imperial College London have now developed a technique, which allows them to switch ATP synthase on and off using azopolyphenols and light.
Polyphenols are found naturally in black grapes, peanuts, berries, and red wine. They are aromatic compounds with bioactive properties, and include dyes, flavoring substances, and tannins. These phytochemicals are reputed to have a range of beneficial nutritional effects, including antioxidant and anti-inflammatory effects, and are even in some cases said to help prevent cancer. Polyphenols can also have bactericidal effects, enabling them, for example, to inhibit tooth decay.
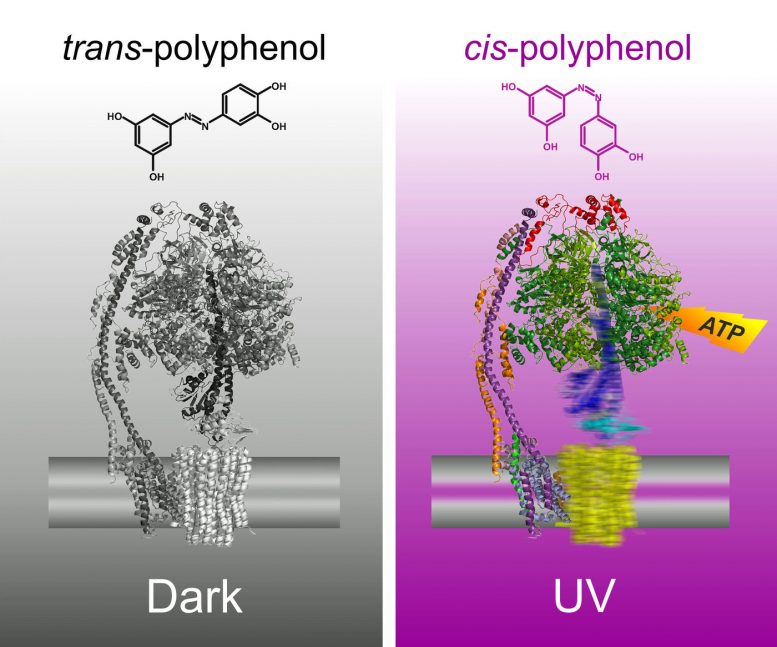
It turns out, however, that these unusual chemicals can do even more: “We have synthesized azopolyphenols that can be switched between the cis and trans forms using ultraviolet light,” explains Felix Hartrampf from the Department of Chemistry at LMU. “We have exploited this property to enable us to turn ATP synthase on and off at will. In the dark, the azopolyphenols adopt the trans form, which blocks ATP synthase activity. If exposed to UV light, they switch to the cis form, reactivating ATP synthase,” explains Bianca Eisel from the Max Planck Institute of Biophysics.
The ability to change the azopolyphenol conformation using light could also be exploited to block other target proteins. In addition, the ability to turn energy generation on and off in living cells could offer a simple way to control specific energy-dependent biotechnological processes. “The results of our work offer an elegant, light-controlled way to provide energy-rich ATP to chemical or biotechnological processes and lays the foundations for the development of further light-activated compounds, which could in the future even be used within the cell,” explained Project Leaders Dirk Trauner (LMU and New York University) and Thomas Meier (Max Planck Institute of Biophysics and Imperial College London).
Reference: “Reversible Optical Control of F1Fo-ATP Synthase Using Photoswitchable Inhibitors” by Bianca Eisel, Felix W.W. Hartrampf, Thomas Meier and Dirk Trauner, 1 January 2018, FEBS Letters.
DOI: 10.1002/1873-3468.12958
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.