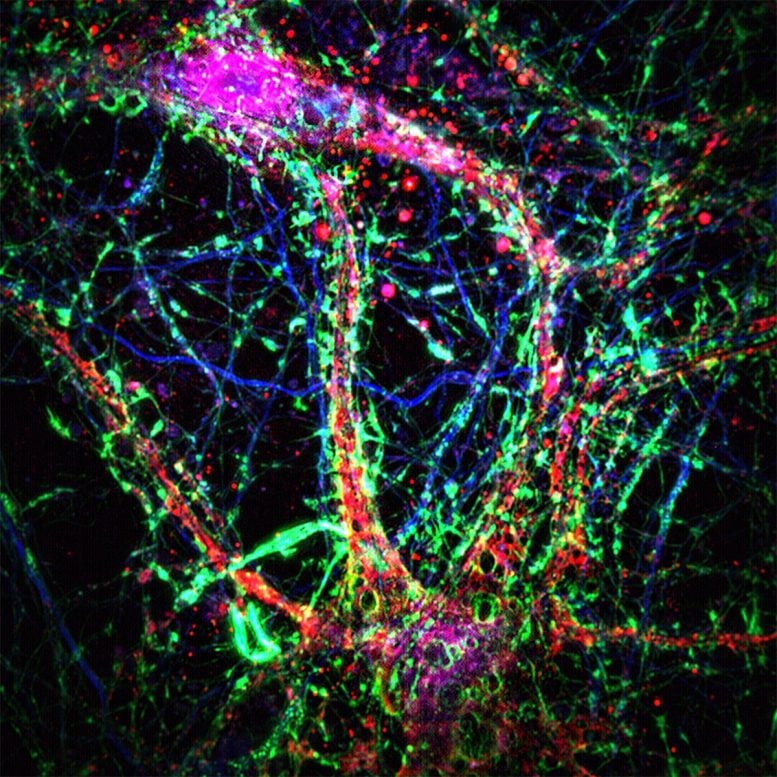
Johns Hopkins scientists found that neurons form tiny nanotube networks to transport toxins like amyloid-beta.
The discovery shows how brain cells might clear waste but also unintentionally spread Alzheimer’s-related proteins.
Brain’s Hidden Tube Network Revealed
Researchers at Johns Hopkins Medicine have discovered how mammalian brains form intricate tube-like networks that move toxic substances in and out of brain cells, much like the pneumatic tube systems once used in offices and stores to send items between locations.
The study, published on October 2 in Science and funded by the National Institutes of Health, used genetically engineered mice and advanced imaging tools to observe these microscopic structures. The researchers believe the discovery could shed new light on the biological processes behind Alzheimer’s disease and other neurodegenerative disorders, potentially leading to future treatment strategies.
The Brain’s Secret Transport System
During their experiments, the scientists observed the formation of nanotubes that appeared to help neurons remove harmful small molecules, including amyloid-beta, a protein known for forming sticky clumps that mark the brains of people with Alzheimer’s disease.
“Cells have to get rid of toxic molecules, and by producing a nanotube, they can then transmit this toxic molecule to a neighboring cell,” says corresponding author Hyungbae Kwon, associate professor of neuroscience at the Johns Hopkins University School of Medicine. “Unfortunately, this also results in spreading harmful proteins to other areas of the brain.”
Highways for Information and Harm
By using powerful microscopes and live-cell imaging, the team watched as neurons created thin, finger-like extensions between their dendrites, the branching structures that connect brain cells. These “dendritic nanotubes” seemed to act as narrow channels that allowed small, toxic molecules to move from one neuron to another.
“The long and thin column-like structures of these dendritic nanotubes help transfer information quickly from neuron to neuron,” says Kwon. “These nanotubes can transport calcium, ions or toxic molecules, and are ideal for sending information to cells that are far away.”
Early Clues to Alzheimer’s
Computational models of the process the researchers documented in the new experiments mimic the process of “early amyloidosis,” the team reports, and “uncover a nanotubular connectivity layer in the brain” that goes beyond usual communication between brain cells.
Kwon says these findings could help researchers better understand how to develop treatments for neurodegenerative conditions, including Alzheimer’s disease.
Seeing Nanotubes in Action
In their experiments, the authors took small tissue samples from the brains of normal lab mice and used high-powered microscopes to characterize the function and structure of nanotubes within neurons. New microscope technology enabled the Johns Hopkins scientists to see the nanotube structures in fine detail and observe how they transferred materials between brain cells.
The scientists then compared brain tissue samples from the normal mice to brain tissue samples from mice genetically engineered to develop the hallmark amyloid buildup of Alzheimer’s disease.
Nanotubes and Alzheimer’s Progression
The researchers say the mice with Alzheimer’s disease had an increased number of nanotubes in their brains at three months old, when the mice were symptom-free, as compared with normal mice of the same age. At six months of age, the number of nanotubes in normal mice and those with Alzheimer’s disease began to equalize.
By taking a closer look at human neurons (sampled with permission from a publicly available electron microscopy database), the scientists identified nanotubes with similar morphology forming between neurons in the same way that the laboratory mice developed them.
Future Experiments
In future experiments, Kwon says, the team will focus on whether larger-scale nanotube networks exist in cell types other than neurons in the brain. Eventually, he intends to design an experiment in which researchers create a nanotube to see how it affects the state of cells.
With such knowledge, Kwon says, there’s the possibility of one day dialing up or down nanotube production to protect the brain.
Toward Targeted Treatments for Neurodegeneration
“When designing a potential treatment based on this work, we can target how nanotubes are produced — by either increasing or decreasing their formation — according to the stage of the disease,” Kwon says.
Reference: “Intercellular communication in the brain through a dendritic nanotubular network” by Minhyeok Chang, Sarah Krüssel, Laxmi Kumar Parajuli, Juhyun Kim, Daniel Lee, Alec Merodio, Jaeyoung Kwon, Shigeo Okabe and Hyung-Bae Kwon, 2 October 2025, Science.
DOI: 10.1126/science.adr7403
Funding for this research was provided by the National Institutes of Health (DP1MH119428 and R01NS138176).
Additional researchers who conducted the study are Minhyeok Chang, Sarah Krüssel, Juhyun Kim, Daniel Lee, Alec Merodio, and Jaeyoung Kwon from Johns Hopkins; and Laxmi Kumar Parajuli and Shigeo Okabe from the University of Tokyo, Japan.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
The word “could” really does a lot of heavy lifting in the articles on popular science, to convince us, peasants, that science is churning out discoveries hand over fist on a daily basis.
Unfortunately, in practice “could” almost never translates to “will” and “does”. It just brings more funding to the people in white coats.