DLB is a relative newcomer to the dementia landscape. It is frequently confused by doctors with Alzheimer’s disease, but also with Parkinson-related dementia (PDD), which may afflict a significant number of Parkinson’s patients, up to 80%, on average 10 years after the onset of Parkinson’s disease.
DLB was identified in 1910, when a German-born American neurologist, Frederic Henry Lewy, observed in the autopsied brains of Parkinson’s and dementia patients “clumps” of an unusual protein (Alzheimer’s brains also present protein clumps, but the proteins involved are different). However, only recently has Dementia with Lewy bodies been recognized as a common type of dementia. “It is the second most frequent cause of degenerative dementia in elderly people (15 to 25% of cases at autopsy)” after Alzheimer’s, writes an international team — including scientists from the Champalimaud Centre for the Unknown (CCU) in Lisbon, Portugal — in a new study published on February 5th 2021 in the Journal of Neurology, Neurosurgery and Psychiatry (a publication of the British Medical Journal group).
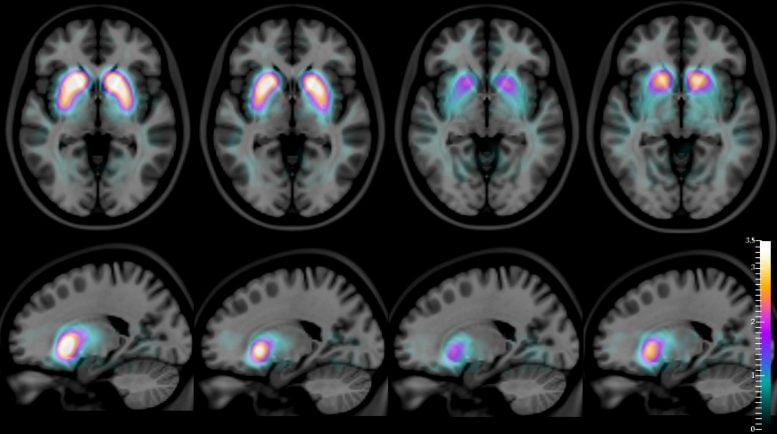
For several years, says Durval Costa — lead author of the new study and head of the Radiopharmacology Lab at the Champalimaud Experimental Clinical Research Programme — it was hypothesized that the use of an imaging technique called SPECT (single-photon emission computed tomography), combined with the intravenous injection of a radioactive compound, [123I]FP-CIT, should allow the distinction of dementia with Lewy bodies from Alzheimer’s disease. This is due to the fact that [123I]FP-CIT binds to dopamine transporters (dopamine is a neurotransmitter) located on the membrane of dopamine-producing neurons which are very abundant in a specific part of the brain called the striatum. Since the striatal dopamine-producing neurons are depleted in dementia with Lewy bodies (just as they are in Parkinson’s disease), but not in Alzheimer’s disease, it was natural to assume that the pattern of distribution of this compound in the brains of patients, revealed by its radioactive emissions (captured by a special camera), would then allow doctors to visually, and in a quantitatively accurate way, distinguish DLB from AD.
The results now presented by the team have been more than twenty years in the making. “The imaging data was acquired around 1996-1999,” explains Francisco Oliveira, who works in Durval Costa’s Lab and is the first author of the new paper. “These patients were followed from the time of their initial clinical diagnosis (and image collection) to that of their death — in some cases, for about 20 years.”
Part of the same team had already published preliminary results in 2002 in the same journal that has now released the new quantitative results. But at the time, the post-mortem material of the patient cohort was not yet fully available, so they had not been able to compare the in vivo diagnosis with the autopsy reports in a sufficient number of patients. “We did not have all the data then,” notes Durval Costa. “Now we do.”
And, for the first time, the autopsies confirm the imaging data with very high accuracy: the images obtained enable not only the differentiation of DLB from AD, but also of DLB from Parkinson’s disease (whose corresponding [123I]FP-CIT distribution patterns are also different from one another). Going back to Robin Williams’ excruciating final years, it is noteworthy that his doctors eventually gave him a diagnosis of Parkinson’s disease, although he and his wife were certain that something else was very wrong with him.
“To be able to quantitatively make these distinctions between diseases is crucial,” says Francisco Oliveira. “These diseases may present overlapping symptoms, which makes the clinical diagnosis difficult in some cases and gives rise to a considerable percentage of misdiagnoses. Studies have shown that, frequently, patients with DLB are clinically diagnosed as AD.” This could lead to patient mismanagement. The care of Alzheimer’s disease patients is different from patients suffering from DLB. Patients with DLB are very sensitive to certain types of medications that should be avoided as they lead to faster deterioration and death.
“Our findings may have a significant impact both for patients and caregivers,” adds Francisco Oliveira. “In addition, the selection of patients for clinical trials can now be done with more accurate biomarkers.” The scientists also hope that, in the future, their quantitative technique may also help to differentiate PD patients with dementia (PDD) from patients with DLB, as they may benefit from different treatment strategies.
References:
“I FP-CIT SPECT in dementia with Lewy bodies, Parkinson’s disease and Alzheimer’s disease: a new quantitative analysis of autopsy confirmed cases” by Francisco P M Oliveira, Zuzana Walker, Rodney W H Walker, Johannes Attems, Joana C Castanheira, Ângelo Silva, Carla Oliveira, Sofia Vaz, Mariana Silva and Durval C Costa, 5 February 2021, JNNP (Journal of Neurology, Neurosurgery and Psychiatry).
DOI: 10.1136/jnnp-2020-324606
“The terrorist inside my husband’s brain” by Susan Schneider Williams, 26 September 2016, Neurology.
DOI: 10.1212/WNL.0000000000003162
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
I take salons. What can I take to replace a satin that won’t affect my brain on a negative way?
What are doctors replacing salon Sawi???t8ins I 65r