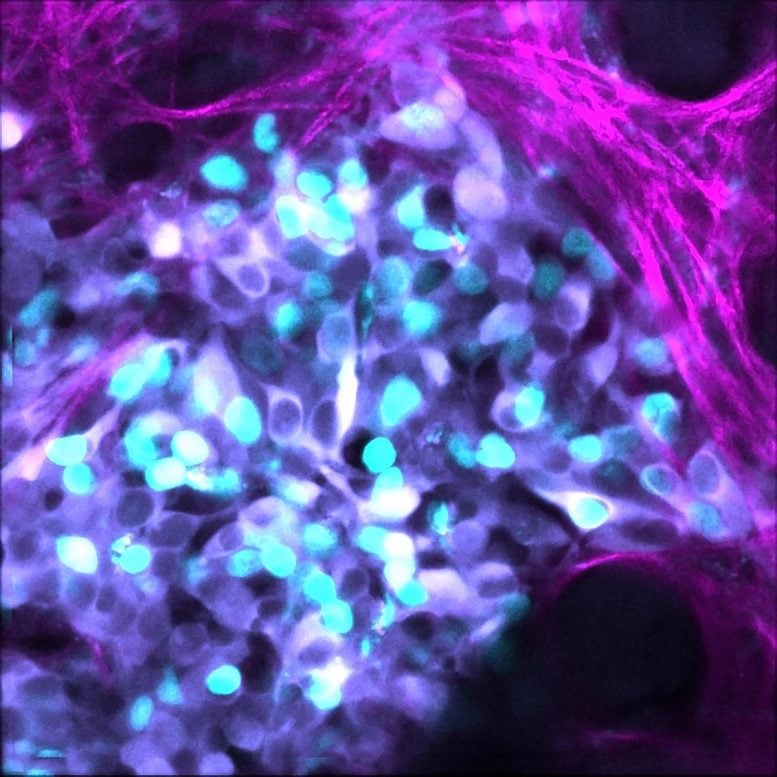
Inherent ‘noise’ in the system of gene expression in tumor cell death, is another tool cancer cells use to resist chemotherapy treatment.
Cancer cells have an innate randomness in their ability to respond to chemotherapy, which is another tool in their arsenal of resisting treatment, new research led by the Garvan Institute of Medical Research shows.
Understanding why some tumor cells become resistant to chemotherapy is a core challenge in cancer research, as chemotherapy is still a frontline treatment for most cancers.
The new research shows that tumor cells from neuroblastoma – cancer that develops in the body’s ‘fight or flight’ sympathetic nervous system – can move between states of responding, or not, to chemotherapy.
“We showed there is ‘noise’ in the process of cell death, which is what happens to cancer cells with chemotherapy treatment – and that this inherent noise, or randomness, in the system of gene expression is an important aspect of chemoresistance,” says Associate Professor David Croucher, Head of the Network Biology Lab at Garvan.
About 15% of people with neuroblastoma don’t respond to chemotherapy treatment.
“Our findings suggest that genetics don’t account for everything; other layers of regulation and other mechanisms of tumor progression can also underpin drug response, so we need to consider them,” says Dr. Sharissa Latham, co-lead author on the study.
The team showed that once neuroblastoma cells reach a state of resisting chemotherapy, they can’t go back, suggesting there is a small window where treatment could work on a tumor cell before it’s locked in.
“Combining chemotherapy with drugs that target this noise within tumors may have the best results as a first-line treatment after diagnosis, before tumors lock into a state of resistance,” says Associate Professor Croucher. This flips on its head the typical protocol for clinical trials in cancer where a new treatment is given to patients who have exhausted all other treatment options.
The new study is published in the journal, Science Advances.
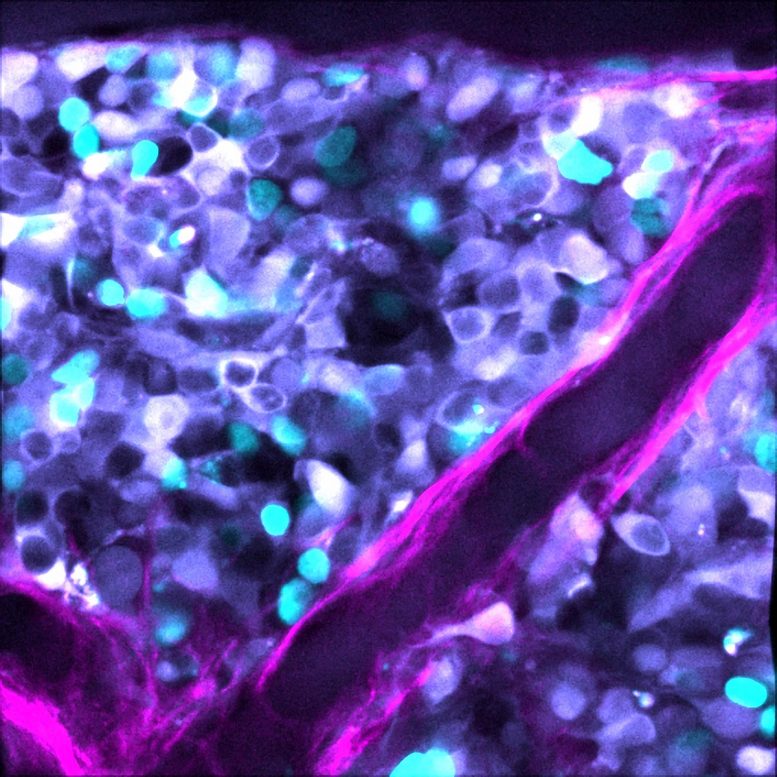
Noise in the Tumor System
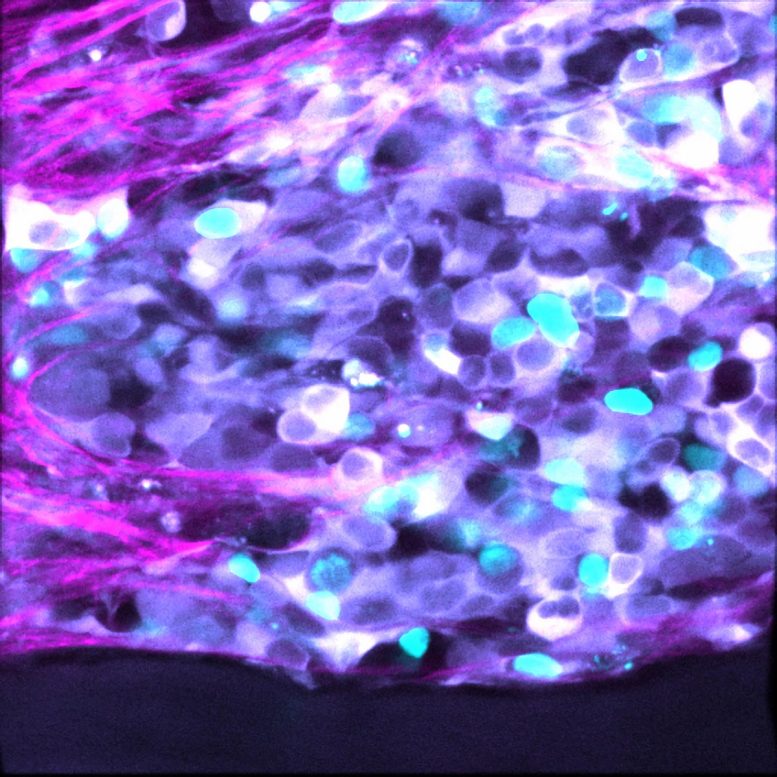
The researchers used mathematical modeling to narrow down the ‘noise’ signals in the pathways of cell death in neuroblastoma tumors. They then applied that to patient cell samples, using cutting-edge imaging to look at single cells, en masse, to visually isolate the cells that didn’t respond to treatment.
The found a marker for resistance – a set of proteins involved in the process of cell death, known as apoptosis.
“We wanted to figure out what underlies that randomness. What is it about those cells and can anything be manipulated to make them respond,” says Dr. Latham.
The team identified certain classes of approved drugs that might be combined with chemotherapy to stabilize expression of the genes involved in cell death, or by changing the innate threshold that may tip a tumor cell into a resistant state.
The next step is to start progressing the work to clinical trial.
Reference: “Memory of stochastic single-cell apoptotic signaling promotes chemoresistance in neuroblastoma” by Jordan F. Hastings, Sharissa L. Latham, Alvin Kamili, Madeleine S. Wheatley, Jeremy Z. R. Han, Marie Wong-Erasmus, Monica Phimmachanh, Max Nobis, Chiara Pantarelli, Antonia L. Cadell, Yolande E. I. O’Donnell, King Ho Leong, Sophie Lynn, Fan-Suo Geng, Lujing Cui, Sabrina Yan, Joanna Achinger-Kawecka, Clare Stirzaker, Murray D. Norris, Michelle Haber, Toby N. Trahair, Frank Speleman, Katleen De Preter, Mark J. Cowley, Ozren Bogdanovic, Paul Timpson, Thomas R. Cox, Walter Kolch, Jamie I. Fletcher, Dirk Fey and David R. Croucher, 3 March 2023, Science Advances.
DOI: 10.1126/sciadv.abp8314
This research was supported by Cancer Institute NSW, NHMRC, Cancer Council NSW, NBCF and the Medical Research Future Fund.
Associate Professor David Croucher is a Conjoint Professor at St Vincent’s Clinical School, Faculty of Medicine and Health, UNSW Sydney. Dr. Sharissa Latham is a Conjoint Lecturer at St Vincent’s Clinical School, Faculty of Medicine and Health, UNSW Sydney.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.