A new study uncovers a molecular modification method for converting CO2 into valuable chemical resources using a platinum surface.
Copper-based (Cu) materials are widely recognized for their efficiency in converting CO2 into valuable hydrocarbons via the CO2 reduction reaction (CO2RR). However, their stability, particularly in acidic environments, needs significant improvement. In contrast, metallic platinum (Pt) demonstrates excellent stability under both acidic and alkaline conditions. However, its high activity in the hydrogen evolution reaction (HER) hinders its effectiveness in CO2RR applications.
To address these challenges, composite materials incorporating metal-doped molecules offer a promising solution. These modified molecules can be securely retained at the interface, forming a unique structure that enhances the metal interface properties. This configuration not only increases the contact between reactants and active sites but also optimizes the adsorption strength of critical intermediates, ultimately improving catalytic performance.
A Molecular Doping Strategy with Pt Nanocrystals
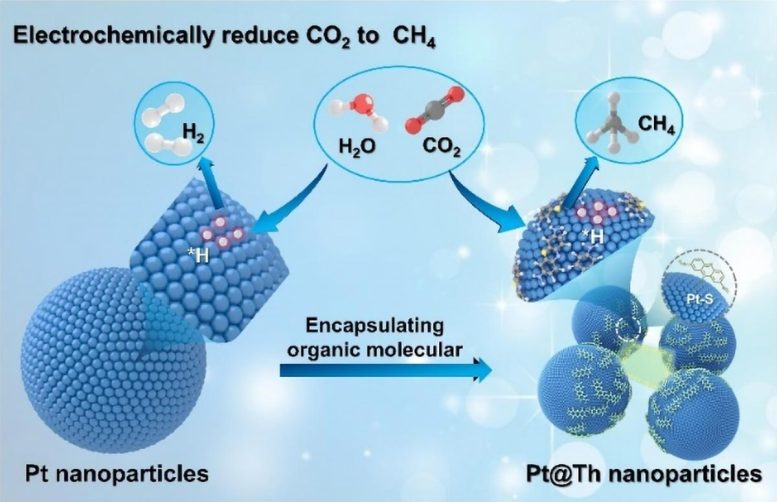
Professor He’s team employed a molecular doping strategy to encapsulate a substantial number of thionine (Th) molecules within Pt nanocrystals, resulting in the formation of PtNPs@Th catalysts. These entrapped Th molecules are securely confined around the Pt surface, significantly altering the catalytic activity of metallic Pt. As a result, the conventional HER activity of Pt-based materials is largely suppressed, while the CO2RR performance is markedly improved in both strongly acidic (pH = 1) and weakly acidic (pH = 4.2) electrolytes.
Importantly, due to the robust corrosion resistance of metallic Pt, the PtNPs@Th catalyst can maintain catalytic stability in acidic media for over 100 hours. The combination of theoretical calculations and in-situ characterization has confirmed that the synergistic effect of Th molecules and Pt facilitates the production of CH4 from CO2RR. This research opens new avenues for the application of molecularly decorated reaction interfaces in diverse electrocatalysis.
Reference: “Molecular modification enables CO2 electroreduction to methane on platinum surface in acidic media” by Hengpan Yang, Huizhu Cai, Deliang Li, Yan Kong, Shangzhao Feng, Xingxing Jiang, Qi Hu and Chuanxin He, 19 November 2024, National Science Review.
DOI: 10.1093/nsr/nwae361
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
You don’t want to encapsulate carbon. We breathe Oxygen supplied by plants that breathe carbon. You cut carbon, you cut plants, you cut Oxygen, at term. Ya get it? You’ve been sold a bill of goods by political manipulators – so that money is shifted for political reasons – but to reasons that are actually detrimental for ecology.
CO2 is already useful in its current state. No need to convert it, especially not with the help of expensive and hard to obtain materials.