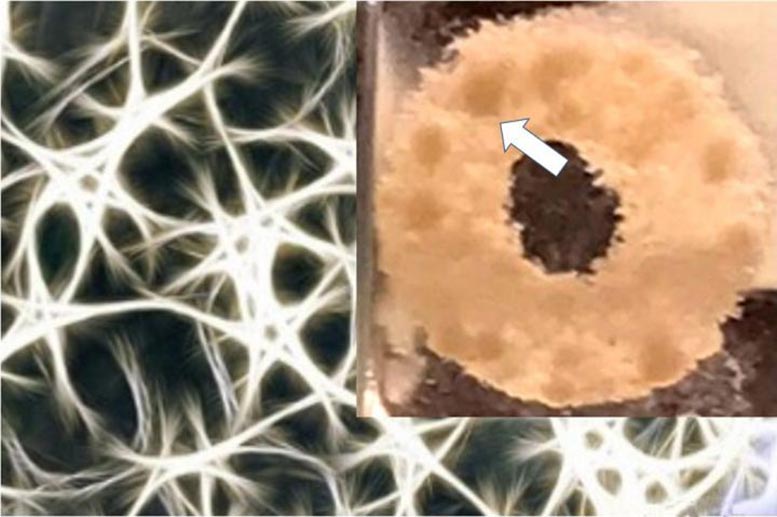
Use of brain-like extracellular matrix allows cell growth and treatment to more closely replicate physiological response.
A team of Tufts University-led researchers has developed three-dimensional (3D) human tissue culture models of pediatric and adult brain cancers in a brain-mimicking microenvironment, a significant advancement for the study of brain tumor biology and pharmacological response. The study was published on October 4, 2019, in Nature Communications.
The researchers created models that include brain-derived extracellular matrix (ECM) — the complex network of proteins and amino acids with bound sugars naturally found in the brain. The ECM not only provides support for surrounding neural tissue, but also helps to guide cell growth and development. Alterations in ECM composition have been associated with brain tumor progression, which in turn alters patterns of genetic and protein expression in the tumor cells.
Earlier studies have noted this important two-way interaction between tumor cells and the surrounding ECM, and observed that the protein composition in the ECM can either prevent or allow the further diffusion of tumor cells in the brain. In order to better understand the dynamic interactions between tumors and the ECM, the study authors developed a 3D in vitro system in which they can examine different ECM components and define their contribution to tumor development, as well as tumor response to drug treatments.
The study focused on two common types of brain tumors, both with particularly dismal prognoses — ependymoma, which occurs in young children, and glioblastoma in adults, which results in a median survival of 1-2 years post diagnosis. In an important advance, the ECM-containing 3D matrix in this study has allowed for the propagation and study of primary tumor cells taken directly from the patient, and to grow them in an environment more similar to the brain. Previous studies examined established tumor cell lines — not necessarily the tumor of interest — on 3D scaffolds or spheroids without the ECM, or spread cells out in two dimensions (plating), eliciting cell behavior not seen in their natural environment
“The power of this platform is that we can tune the composition of the ECM to find out the role of each component in tumor growth, and we can see the effect on tumor cells derived directly from the patient,” said David Kaplan, Stern Family Professor of Engineering, chair of the Department of Biomedical Engineering at Tufts’ School of Engineering and program faculty member at the Sackler School of Graduate Biomedical Sciences. “Another important feature is that we can track the 3D growth of cells with non-invasive two-photon excited fluorescence metabolic imaging via the contributions of Irene Georgakoudi’s team on the project. In other words, we can use non-invasive imaging to assess if they are viable and growing, or stressed and dying, in real-time.”
Among the findings revealed in the study was that fetal ECM, which contains higher levels of collagen, HA, and certain CSPGs, was better at supporting tumor growth than adult ECM in the 3D cultures (both fetal and adult ECMs were derived from pig brains). That result correlates with the notion that brain cancers tend to alter the ECM so its composition becomes more “fetal-like” to support their growth, according to the researchers.
Another key finding was the appearance of lipid (fat) droplets being released by the adult glioblastoma cells which may contribute to lowering the drug sensitivity of many glioblastoma cells (possibly by absorbing the drugs). This may be correlated with poor survival both in the 3D tissue model and in patients. The droplets have not been observed in vitro prior to these experiments, suggesting that this model is a robust system to study the behavior of brain tumors in the lab. The application of engineering solutions (in this case, the development of a 3D silk-based matrix) to improve the study of the brain is a collaborative effort taken on by the authors as part of the Initiative for Neural Science, Disease & Engineering (INSciDE@Tufts).
“With this platform, we have the potential to better understand what dictates the invasive behavior of brain tumors and screen drugs for their effect on tumor growth of patient-derived cells,” said Disha Sood, a graduate student in Kaplan’s lab and first author of the study. “Although it’s a preliminary notion, the ability to maintain viable cultures of patient-derived tumor cells and metabolically track them non-invasively, suggests the possibility of monitoring the cells’ behavior and drug sensitivity over time, to inform treatment decisions.”
###
Other authors contributing to the study are Dmitra Pouli, Craig Mizzoni, and Nicole Raia, graduate students in the Department of Biomedical Engineering in the School of Engineering at Tufts University; Lauren D. Black III, associate professor, and Irene Georgakoudi, professor of biomedical engineering at Tufts School of Engineering; Albert Tai and Knarik Arkun, assistant professors, and Julian Wu, associate chair of neurosurgery at Tufts Medical Center as well as a professor at Tufts University School of Medicine; Dennis Steindler, senior scientist at the Jean Mayer USDA Human Nutrition Research Center on Aging, Tufts University; Bjorn Scheffler, professor at the German Cancer Consortium (DKTK); and Min Tang-Schomer of the Jackson Laboratory in Connecticut.
This work was funded by the U.S. National Institutes of Health (NIH) P41 Tissue Engineering Resource Center grant #EB002520, NIH R01 grant #NS092847, and the NIH Research Infrastructure grant #S10 OD021624 766, and National Science Foundation Major Research Instrumentation grant #1531683, and the U.S. Department of Agriculture’s Agricultural Research Service. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Reference: “3D extracellular matrix microenvironment in bioengineered tissue models of primary pediatric and adult brain tumors” by Disha Sood, Min Tang-Schomer, Dimitra Pouli, Craig Mizzoni, Nicole Raia, Albert Tai, Knarik Arkun, Julian Wu, Lauren D. Black III, Bjorn Scheffler, Irene Georgakoudi, Dennis A. Steindler and David L. Kaplan, 4 October 2019, Nature Communications.
DOI: 10.1038/s41467-019-12420-1
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.