Using carbon nanotubes, researchers at Rice University and Texas Children’s Hospital have created heart-defect patches that improve electrical signaling between immature heart cells.
Carbon nanotubes serve as bridges that allow electrical signals to pass unhindered through new pediatric heart-defect patches invented at Rice University and Texas Children’s Hospital.
A team led by bioengineer Jeffrey Jacot and chemical engineer and chemist Matteo Pasquali created the patches infused with conductive single-walled carbon nanotubes. The patches are made of a sponge-like bioscaffold that contains microscopic pores and mimics the body’s extracellular matrix.
The nanotubes overcome a limitation of current patches in which pore walls hinder the transfer of electrical signals between cardiomyocytes, the heart muscle’s beating cells, which take up residence in the patch and eventually replace it with new muscle.
The work appears this month in the American Chemical Society journal ACS Nano. The researchers said their invention could serve as a full-thickness patch to repair defects due to Tetralogy of Fallot, atrial and ventricular septal defects, and other defects without the risk of inducing abnormal cardiac rhythms.
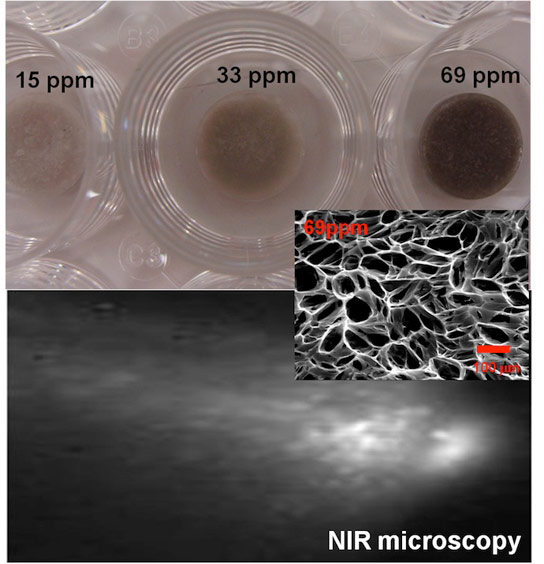
The original patches created by Jacot’s lab consist primarily of hydrogel and chitosan, a widely used material made from the shells of shrimp and other crustaceans. The patch is attached to a polymer backbone that can hold a stitch and keep it in place to cover a hole in the heart. The pores allow natural cells to invade the patch, which degrades as the cells form networks of their own. The patch, including the backbone, degrades in weeks or months as it is replaced by natural tissue.
Researchers at Rice and elsewhere have found that once cells take their place in the patches, they have difficulty synchronizing with the rest of the beating heart because the scaffold mutes electrical signals that pass from cell to cell. That temporary loss of signal transduction results in arrhythmias.
Nanotubes can fix that, and Jacot, who has a joint appointment at Rice and Texas Children’s, took advantage of the surrounding collaborative research environment.
“This stemmed from talking with Dr. Pasquali’s lab as well as interventional cardiologists in the Texas Medical Center,” Jacot said. “We’ve been looking for a way to get better cell-to-cell communications and were concentrating on the speed of electrical conduction through the patch. We thought nanotubes could be easily integrated.”
Nanotubes enhance the electrical coupling between cells that invade the patch, helping them keep up with the heart’s steady beat. “When cells first populate a patch, their connections are immature compared with native tissue,” Jacot said. The insulating scaffold can delay the cell-to-cell signal further, but the nanotubes forge a path around the obstacles.
Jacot said the relatively low concentration of nanotubes — 67 parts per million in the patches that tested best — is key. Earlier attempts to use nanotubes in heart patches employed much higher quantities and different methods of dispersing them.
Jacot’s lab found a component they were already using in their patches – chitosan – keeps the nanotubes spread out. “Chitosan is amphiphilic, meaning it has hydrophobic and hydrophilic portions, so it can associate with nanotubes (which are hydrophobic) and keep them from clumping. That’s what allows us to use much lower concentrations than others have tried.”
Because the toxicity of carbon nanotubes in biological applications remains an open question, Pasquali said, the fewer one uses, the better. “We want to stay at the percolation threshold, and get to it with the fewest nanotubes possible,” he said. “We can do this if we control dispersion well and use high-quality nanotubes.”
The patches start as a liquid. When nanotubes are added, the mixture is shaken through sonication to disperse the tubes, which would otherwise clump, due to van der Waals attraction. Clumping may have been an issue for experiments that used higher nanotube concentrations, Pasquali said.
The material is spun in a centrifuge to eliminate stray clumps and formed into thin, fingernail-sized discs with a biodegradable polycaprolactone backbone that allows the patch to be sutured into place. Freeze-drying sets the size of the discs’ pores, which are large enough for natural heart cells to infiltrate and for nutrients and waste to pass through.
As a side benefit, nanotubes also make the patches stronger and lower their tendency to swell while providing a handle to precisely tune their rate of degradation, giving hearts enough time to replace them with natural tissue, Jacot said.
“If there’s a hole in the heart, a patch has to take the full mechanical stress,” he said. “It can’t degrade too fast, but it also can’t degrade too slow, because it would end up becoming scar tissue. We want to avoid that.”
Pasquali noted that Rice’s nanotechnology expertise and Texas Medical Center membership offer great synergy. “This is a good example of how it’s much better for an application person like Dr. Jacot to work with experts who know how to handle nanotubes, rather than trying to go solo, as many do,” he said. “We end up with a much better control of the material. The converse is also true, of course, and working with leaders in the biomedical field can really accelerate the path to adoption for these new materials.”
Seokwon Pok, a Rice research scientist in Jacot’s lab, is the lead author of the paper. Co-authors are research scientist Flavia Vitale, graduate student Omar Benavides, and former postdoctoral researcher Shannon Eichmann, all of Rice. Pasquali is chair of Rice’s Department of Chemistry and a professor of chemical and biomolecular engineering, of materials science and nanoengineering, and of chemistry. Jacot is an assistant professor of bioengineering at Rice, director of the Pediatric Cardiac Bioengineering Laboratory at the Congenital Heart Surgery Service at Texas Children’s, and an adjunct assistant professor at Baylor College of Medicine.
The National Institutes of Health, the Welch Foundation, and Texas Children’s Hospital supported the research.
Reference: “Biocompatible Carbon Nanotube – Chitosan Cardiac Scaffold Matching the Electrical Conductivity of the Heart” by Seokwon Pok, Flavia Vitale, Shannon L. Eichmann, Omar M. Benavides, Matteo Pasquali and Jeffrey G. Jacot, 18 September 2014, ACS Nano.
DOI: 10.1021/nn503693h
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.