
The discovery that inert helium can form bonds with iron may reshape our understanding of Earth’s history.
Researchers from Japan and Taiwan have made a groundbreaking discovery, demonstrating for the first time that helium—long considered chemically inert—can bond with iron under extreme pressure. Using a laser-heated diamond anvil cell, they observed this unexpected interaction, suggesting that vast amounts of helium may be present in the Earth’s core. This finding challenges long-held theories about the planet’s internal structure and history and could provide new insights into the primordial nebula from which our solar system originated.
Volcanic eruptions primarily release rocks and minerals, but they can also emit traces of a rare gas known as primordial helium. Unlike the more common isotope, helium-4 (⁴He), which consists of two protons and two neutrons and is continuously produced by radioactive decay, primordial helium—helium-3 (³He)—contains only one neutron and is not formed on Earth. Its presence offers valuable clues about the planet’s deep interior and its connection to cosmic origins.
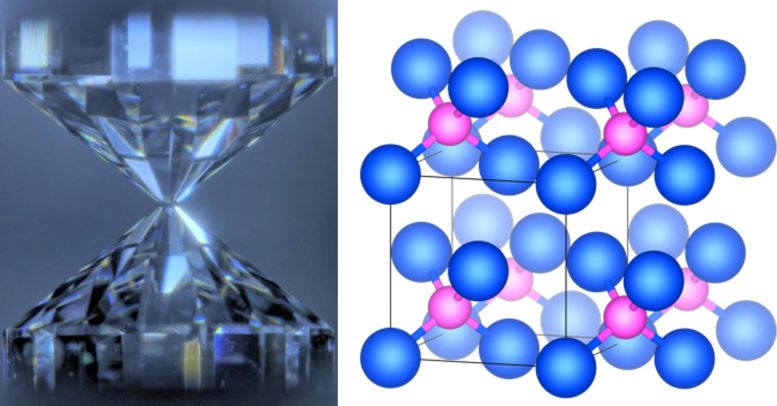
Given the occasionally high 3He/4He ratios found in volcanic rocks, especially in Hawaii, researchers have long believed there are primordial materials containing 3He deep within the mantle. However, graduate student Haruki Takezawa and members of Professor Kei Hirose’s group from the University of Tokyo’s Department of Earth and Planetary Science have now challenged this view with a new take on a familiar experiment: crushing things.
Simulating Earth’s Extreme Conditions
“I have spent many years studying the geological and chemical processes that take place deep inside the Earth. Given the intense temperatures and pressures at play, experiments to explore some aspects of this environment must replicate those extreme conditions. So, we often turn to a laser-heated diamond anvil cell to impart such pressures on samples to see the result,” said Hirose. “In this case, we crushed iron and helium together under about 5-55 gigapascals of pressure and at temperatures of 1,000 kelvins to nearly 3,000 kelvins. Those pressures correspond to roughly 50,000-550,000 times atmospheric pressure and the higher temperatures used could melt iridium, the material often used in car engine spark plugs due to its high thermal resistance.”
Previous studies have shown only small traces of combined iron and helium, in the region of seven parts per million helium within iron. But in this case, they were surprised to find the crushed iron compounds contained as much as 3.3% helium, about 5,000 times higher than previously seen. Hirose suspects this is at least in part due to something novel about this particular set of experiments.
“Helium tends to escape at ambient conditions very easily; everyone has seen an inflatable balloon wither and sink. So, we needed a way to avoid this when taking our measurements,” he said. “Though we carried out the material syntheses under high temperatures, the chemical-sensing measurements were done at extremely cold, or cryogenic, temperatures. This way prevented helium from escaping and allowed us to detect helium in iron.”
This finding has implications for understanding Earth’s origins. The presence of helium in the core suggests the young Earth likely captured some gas from the solar nebula of hydrogen and helium that surrounded the early solar system. This could also mean that some of Earth’s water may have come from hydrogen in this ancient gas, offering a new perspective on the planet’s early development.
Reference: “Formation of Iron-Helium Compounds under High Pressure” by Haruki Takezawa, Han Hsu, Kei Hirose, Fumiya Sakai, Suyu Fu, Hitoshi Gomi, Shiro Miwa and Naoya Sakamoto, 25 February 2025, Physical Review Letters.
DOI: 10.1103/PhysRevLett.134.084101
Funding: This work was supported by JSPS grant 21H04506 to K.H.; H.H. is supported by National Science and Technology Council of Taiwan under grants NSTC 113-2116-M-008-010, 112-2112-M-008-038, and 111-2112-M-008-032.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Mind-blowing! If helium can bond with iron under extreme conditions, who knows what other ‘inert’ elements might be hiding in plain sight under the Earth’s surface?
We tend to think of the Earth’s interior as being well understood, but discoveries like this remind us that we are only beginning to scratch the surface. If helium can chemically bond with iron at high pressures, could there be other exotic compounds forming deep within the Earth that we’ve never even considered?