University of Oklahoma Medicine, University of Oklahoma Health Sciences Center and Oklahoma Medical Research Foundation file for FDA Emergency Use Authorization for COVID-19 detection test utilizing fluidigm technology.
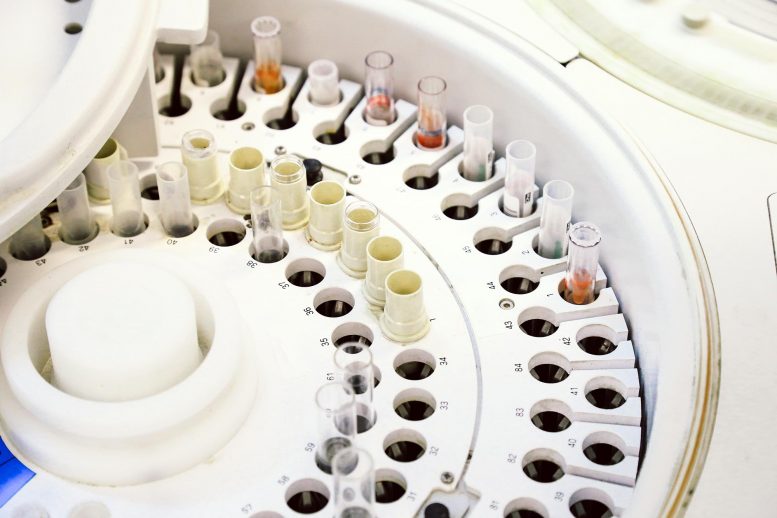
OU Medicine, the OU Health Sciences Center and the Oklahoma Medical Research Foundation collaborated to create a new test for COVID-19 using technology and reagents from Fluidigm Corporation, an innovative biotechnology tools provider. The test is intended for large-scale testing of patients across the OU Medicine healthcare system, with the capacity to test 180,000 samples over the next 90 days.
“We believe this greatly increased number of tests will serve as a turning point in our battle against the COVID-19 pandemic.” — Chuck Spicer, FACHE, President and CEO of OU Medicine, Inc.
High-complexity laboratories like OU Medicine’s that are certified by CLIA (Clinical Laboratory Improvement Amendments) are eligible to create their own diagnostic test for COVID-19, according to new guidance issued by the U.S. Food and Drug Administration. OU Medicine has applied for Emergency Use Authorization for the test from the FDA.
“We are grateful for the collaboration and leadership of the researchers at OU Health Sciences Center and OMRF. This partnership with three Oklahoma healthcare leaders and Fluidigm makes it possible for OU Medicine to offer this unique testing capability to our state,” said Chuck Spicer, FACHE, President and CEO of OU Medicine, Inc. “We believe this greatly increased number of tests will serve as a turning point in our battle against the COVID-19 pandemic.
“Readily accessible and rapid testing for COVID-19 not only expedites treatment for patients who test positive, but it allows healthcare providers to be routinely tested to ensure their safety and that of everyone they encounter,” Spicer added. “Increased testing will also allow more patients without COVID-19 to access specialty care at OU Medicine, including surgeries, procedures and other diagnostic testing.”
The testing platform will produce results quickly – in about six hours – which will allow physicians to promptly begin treatment on patients who test positive, and will help public health officials gain a more accurate picture of the spread of the virus in Oklahoma.
OMRF’s experience with Fluidigm’s microfluidics technology and its Fluidigm Biomark™ HD system was a crucial component in the creation and validation of the test. The FAA Civil Aerospace Medical Institute in Oklahoma City also provided a Biomark™ HD system for use in the testing. Microfluidics technology generates more data and uses a fraction of limited testing reagents per sample compared with more traditional technology.
“Solutions that offer scale and high-speed processing are critically important in ramping up testing capabilities for COVID-19,” said OMRF President Stephen Prescott, M.D. “We anticipate that the test will generate several thousand test results per day, and it comes online at a particularly critical time for COVID-19 patients in Oklahoma.”
Rapid, high-capacity testing will continue to be critical for contact tracing – determining how many people have been exposed to an infected person. Testing will remain fully functional for as long as patients and the residents of Oklahoma need to be tested. That capability will help the state determine when and how Oklahomans can begin returning to work and their daily lives following the peak of the pandemic.
“The remarkable volume of testing made available through the tireless work of our OU Health Sciences Center researchers and partners at OU Medicine, OMRF, and Fluidigm is, simply put, game-changing,” said OU Interim President Joseph Harroz Jr. “As this innovative testing platform ramps into full capacity, we will be able to ensure Oklahoma has consistent testing for the current environment and for possible future outbreaks, as well as demographic and geographic sampling that can help contain further spread.”
Because Fluidigm technology employs a different approach than other testing platforms, OU Medicine is able to benefit from its speed and scale capabilities.
“Since the beginning of the COVID-19 outbreak, adequate resources for testing have been a serious challenge for healthcare systems around the world,” said Chris Linthwaite, President and CEO of Fluidigm. “Speed, scale and automation are critical components to delivering timely results.
“We are excited to support OU Medicine, the Oklahoma Medical Research Foundation and the OU Health Sciences Center in this incredibly important program,” Linthwaite added. “In parallel with this scaling of testing capacity, Fluidigm products are providing vital information on immune response to COVID-19 exposure. This information is essential for evaluation of patient management approaches and effective vaccine and therapeutic candidate development. The COVID-19 testing program underway in Oklahoma City positions their community at the forefront of pandemic preparation efforts in the global fight against this virus.”







Thank you for the reporting on the potential of this research and development effort. I’d note that reports like this one would be much more valuable if characteristics such as sensitivity, specificity, positive predictive value, and negative predictive value were included. Successfully producing a test is, of course, a valuable endeavor but not all tests are created equal. When reporting on such advances, having enough information to interpret how well a test performs is of critical importance.