Research indicates that regulatory T cells (Tregs) might stabilize mood and prevent depression, with their depletion linked to increased anxiety and cognitive issues in Alzheimer’s models.
Regulatory T cells—known as the workhorses of the body’s immune system—may also play a role in stabilizing mood, a study suggests. The transcription factor Forkhead box P3 (Foxp3) controls the production of regulatory T cells (Tregs). While Tregs primarily regulate the adaptive immune system, research indicates they may also influence mood. Notably, reduced Foxp3 expression has been linked with major depressive disorders.
Experimental Findings on Tregs and Behavior
Giulio Maria Pasinetti and colleagues tested a line of lab mice whose Tregs can be temporarily depleted on standard tasks designed to measure depression and anxiety in the rodents. Treg-depleted mice were more likely to hide in darkness, moved less, and gave up on self-preservation actions more easily—suggesting that Treg-depleted mice were more anxious and depressed than control mice. These neurobehavioral changes in Treg-depleted mice were reversed after restoration of Foxp3-expressing cells, and Treg-restored mice were more similar to controls than Treg-depleted mice were.
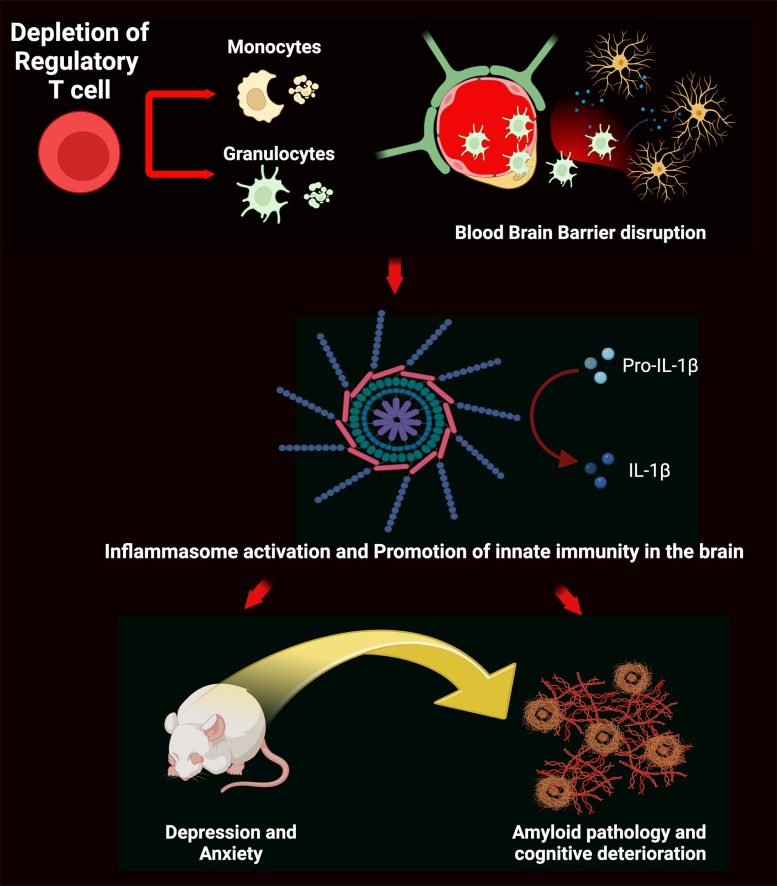
Tregs in Neurological Disorders
Additionally, mice bred to model Alzheimer’s disease showed cognitive impairments when their Tregs were depleted. The authors posit that Treg depletion causes proliferation of peripheral immune cells, some of which can cross the blood-brain barrier into the brain and cause inflammatory responses in the hippocampal formation. This transient activation of innate immunity in the brain can cause anxiety, depression, or Alzheimer’s disease-type cognitive deterioration, according to the authors.
Reference: “Transient anxiety-and depression-like behaviors are linked to the depletion of Foxp3-expressing cells via inflammasome in the brain” by Eun-Jeong Yang, Md Al Rahim, Elizabeth Griggs, Ruth Iban-Arias and Giulio Maria Pasinetti, 22 August 2023, PNAS Nexus.
DOI: 10.1093/pnasnexus/pgad251
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.