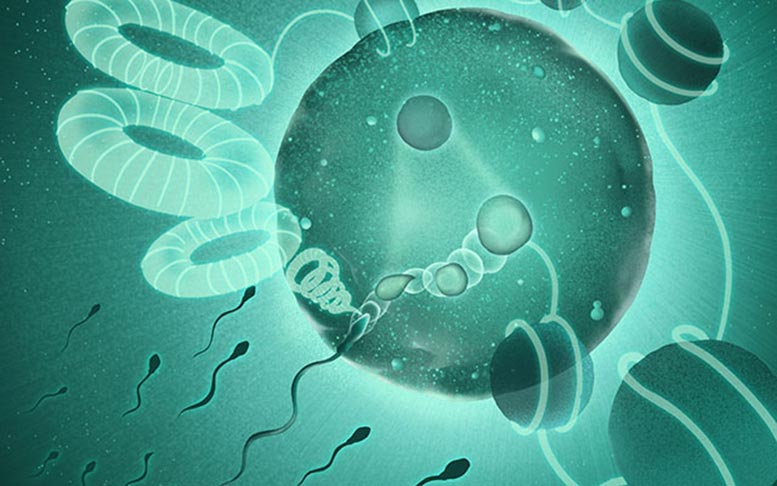
University of California San Diego (UCSD) researchers discovered the enzyme SPRK1’s role in reorganizing the paternal genome during the first moments of fertilization — a finding that might help explain infertility cases of unknown cause.
A sperm enters an egg, an embryo develops, and eventually a baby is born. But back up a second — how does the mother’s half-genome actually merge with the father’s half-genome to form one new human genome? Turns out researchers don’t really know that much about these relatively brief, yet crucial, incipient moments in fertilization.
Researchers at the University of California San Diego School of Medicine have discovered that the enzyme SPRK1 leads the first step in untangling a sperm’s genome, kicking out special packing proteins, which opens up the paternal DNA and allows for major reorganization — all in a matter of hours.
The study was published on March 12, 2020, in the journal Cell.
“In this study, we were simply interested in answering a fundamental question about the beginning of life,” said senior author Xiang-Dong Fu, PhD, Distinguished Professor in the Department of Cellular and Molecular Medicine at UC San Diego School of Medicine. “But in the process we’ve uncovered a step that might malfunction for some people, and contribute to a couple’s difficulty conceiving. Now that we know SPRK1 plays a role here, its potential part in infertility can be further explored.”
Sperm can be up to 20 times smaller than a normal cell in the body. And while sperm carry only half as much genetic material as a regular cell, it needs to be folded and packaged in a special way in order to fit. One way nature does this is by replacing histones — proteins around which DNA is wound, like beads on a necklace — with a different type of protein called protamines.
Fu’s team has long studied SPRK1 for a completely different reason: its ability to splice RNA, an important step that enables the translation of genes to proteins. They previously showed that SPRK1 is over-activated in colon cancer, and they developed inhibitors to dampen the enzyme.
But back in 1999, shortly after Fu published a paper that first described the enzyme’s role in RNA splicing, a research group in Greece noted similarities in the sequence of amino acid building blocks that make up SPRK1 substrates (the proteins upon which the enzyme acts) and protamine. Fu thought about it for years, but didn’t have the expertise and tools to study sperm development. In 2015, Lan-Tao Gou, PhD, was interviewing for a position as postdoctoral researcher when Fu realized that with Gou’s experience in spermatogenesis, he finally had the right person for the job.
“I said to Lan-Tao, let’s do something nobody else is doing. I have a theory and you have the expertise,” Fu said. “So we borrowed the equipment we needed and leveraged the core facilities we have here at UC San Diego.
“And, surprisingly, everything we tried supported our hypothesis — SRPK1 leads a double life, swapping protamines for histones once sperm meets egg.”
According to Fu, SPRK1 most likely started out playing this role in early embryogenesis, then later evolved the ability to splice RNA. In this way, SPRK1 gets to stick around even when it’s no longer needed for embryogenesis.
Fu, Gou, and team next want to determine the signals that instruct sperm to synchronize with the maternal genome.
“We have a ton of new ideas now,” Fu said. “And the better we understand every step in the process of spermatogenesis, fertilization and embryogenesis, the more likely we are to be able to intervene when systems malfunction for couples struggling with reproductive issues.”
Reference: “Initiation of Parental Genome Reprogramming in Fertilized Oocyte by Splicing Kinase SRPK1-Catalyzed Protamine Phosphorylation” by Lan-Tao Gou, Do-Hwan Lim, Wubin Ma, Brandon E. Aubol, Yajing Hao, Xin Wang, Jun Zhao, Zhengyu Liang, Changwei Shao, Xuan Zhang, Fan Meng, Hairi Li, Xiaorong Zhang, Ruiming Xu, Dangsheng Li, Michael G. Rosenfeld, Pamela L. Mellon, Joseph A. Adams, Mo-Fang Liu and Xiang-Dong Fu, 12 March 2020, Cell.
DOI: 10.1016/j.cell.2020.02.020
Additional study co-authors include: Do-Hwan Lim, Wubin Ma, Brandon E. Aubol, Yajing Hao, Jun Zhao, Zhengyu Liang, Changwei Shao, Xuan Zhang, Fan Meng, Hairi Li, Michael G. Rosenfeld, Pamela L. Mellon, Joseph A. Adams, UC San Diego; Xin Wang, Xiaorong Zhang, Ruiming Xu, Dangsheng Li, and Mo-Fang Liu, Chinese Academy of Sciences.
This research was funded, in part, by the National Institutes of Health (grants GM052872, GM067969, HG004659, P30DK063491, P30CA023100, P42ES010337 and K99HD094901).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Good finding. Fund for the research well utilized and viable for future’s biological disease and skeletal development control.
Amazing to read about such research cooperation ! It is also very telling that so many of these scientists are Chinese or have Asian names at least – you are all so smart & I for one appreciate the huge contribution to the world that this group is doing ! That intensity in education of small Asian children is paying off in amazing ways for the planet !