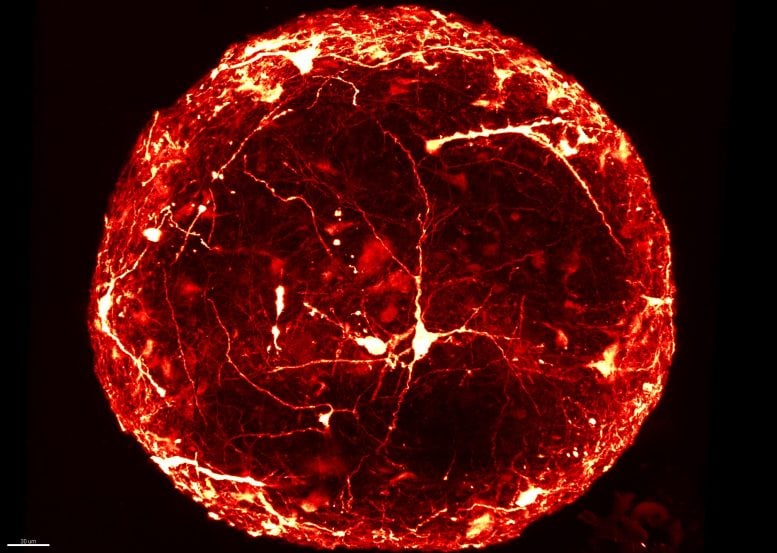
A seven-month-old 3D ‘mini-brain’ imaged with a confocal microscope to reveal the structure of individual neurons throughout. Credit: HEPIA
Revealing details of the internal structure of ‘mini-brains’ could help accelerate drug studies and may offer alternatives to some animal testing.
‘Mini-brains’ are pin-head-sized collections of several different types of human brain cells. They are used as a tool, allowing scientists to learn about how the brain develops, study disease, and test new medicines. Personalized ‘mini-brains’ can be grown from stem cells generated from a sample of human hair or skin and could shed light on how brain disease progresses in an individual and how this person may respond to drugs.
Research published today by a team of scientists and engineers from HEPIA and the Wyss Center for Bio and Neuroengineering, in the journal Frontiers in Bioengineering and Biotechnology, has revealed the detailed internal anatomy of ‘mini-brains’, for the first time.
“Despite advances in growing ‘mini-brains’, it has been difficult to understand in detail what is going on inside – until now,” said Professor Adrien Roux from the Tissue Engineering Laboratory, HEPIA, senior author on the paper.
“Typically, to look inside a ‘mini-brain’, we slice it thinly and view it on a slide under a microscope. This is a slow process that can damage the sample. Now, for the first time, we have produced high-resolution 3D images of single neurons within intact ‘mini-brains’, revealing their remarkable complexity,” added Dr. Subashika Govindan, lead author on the paper, who carried out the work at HEPIA and is now Wellcome DBT early career fellow at the Indian Institute of Technology Madras (IITM).
The team combined a novel technique for labeling individual neurons with a method to make the whole sample completely transparent.
Leveraging the Wyss Center’s microscopy capabilities, the team developed a state-of-the-art custom module, including a bespoke sample holder and sensitive imaging detectors, for capturing 3D images of entire intact ‘mini-brains’, without slicing them. They were then able to visualize and analyze the 3D morphology of specific neurons and their anatomical distribution inside the ‘mini-brains’.
Dr. Laura Batti, Microscopy Facility Manager at the Wyss Center said: “Human ‘mini-brains’ have a life span of more than a year and, with our new ability to visualize them in more detail, we can envision benefits such as reducing some animal testing.”
The new approach could also enable imaging of large numbers of ‘mini-brains’, making it suitable for high-throughput screening for drug discovery or toxicity testing. It is reproducible and cost-effective and could potentially help accelerate personalized medicine studies.
Reference: “Mass generation, neuron labeling, and 3D imaging of minibrains” by Subashika Govindan, Laura Batti, Samira F. Osterop, Luc Stoppini and Adrien Roux, 7 January 2021, Frontiers in Bioengineering and Biotechnology.
DOI: 10.3389/fbioe.2020.582650








Be the first to comment on "Peeking Inside Human “Mini-Brains” to Understand the Brain, Study Disease and Test New Medicines"