SOX9, a transcription factor crucial for stem cell fate, can trigger cancer when reactivated in adult epidermal stem cells. Researchers found that SOX9 functions as a pioneer factor, unlocking chromatin and reprogramming cell identity. Their study offers new therapeutic insights into SOX9-driven cancers.
In the early stages, every stem cell is presented with a decisive selection. For example, during the development of skin, the embryonic epidermis starts off with a single layer of epidermal progenitor cells. These cells have to decide whether to transform into a mature epidermal cell or to opt for becoming a hair follicle cell. This critical decision, known as the fate switch, is regulated by the transcription factor known as SOX9. When SOX9 is expressed by the progenitor cell, it leads to the development of hair follicle cells. In its absence, the cell develops into an epidermal cell.
But there is a dark side to SOX9, as it has been associated with some of the most lethal cancers across the globe, such as lung, skin, head and neck, and bone cancer. In the context of skin, certain aberrant adult epidermal stem cells might unexpectedly activate SOX9, regardless of their chosen path—and never turn it off, kickstarting a process that ultimately activates cancer genes.
Scientists have never fully understood how this doomed outcome ensues at a molecular level. But now Rockefeller researchers have revealed the mechanisms behind this malignant turn of events. SOX9, it turns out, belongs to a special class of proteins that govern the transfer of genetic information from DNA to mRNA. That means it has the ability to pry open sealed pockets of genetic material, bind to previously silent genes within, and activate them. They published their results in Nature Cell Biology.
“Our discovery provides new insights into how cancer derails a stem cell’s carefully tuned decision-making process, thereafter making it impossible for it to make normal tissue,” says Elaine Fuchs, head of the Robin Chemers Neustein Laboratory of Mammalian Cell Biology and Development. “It also illuminates new SOX9-activated genes as potential therapeutic targets.”
Rare Keys to Gene Expression
Our genome is not an open book. In fact, it’s more like a library filled with a few billion books that are mostly under lock and key—the majority of genetic material actually lies silent within non-coding and tightly bound packets of DNA cordoned off by histone proteins in a closed state. Together the DNA and histones form what’s called closed chromatin. The genes that are packaged into this cloistered material are inaccessible to the transcription proteins, or factors, that would help it to express the genes within.
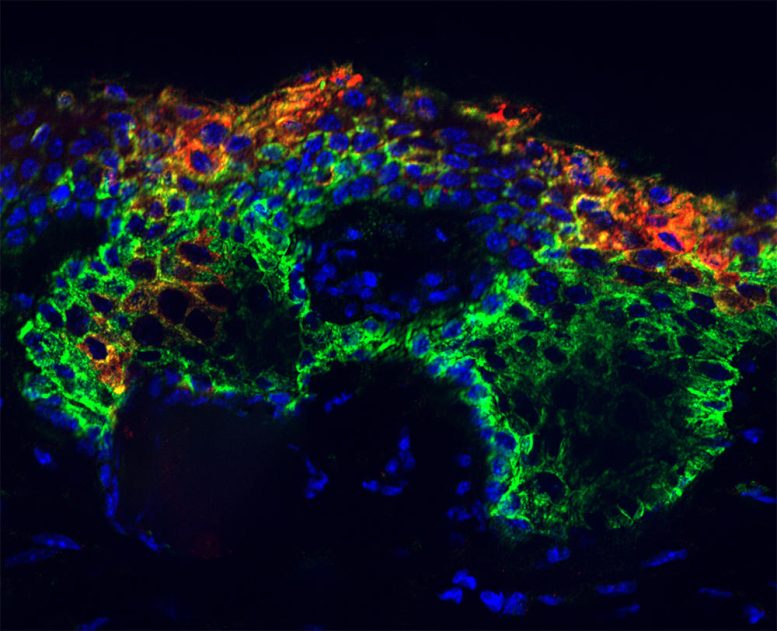
But there are a few rare keys that aren’t simply transcription factors. These “pioneer factors” can unlock those genetic packets. They possess the superpower to peer inside the closed chromatin and recognize binding sites within. They then recruit other transcription factors to help them pry open the closed chromatin and bind to receptor sites on the nucleosome, which reprograms the chromatin and activates new genes.
This normally happens during the early stages of development, when a stem cell’s fate is yet to be determined. In adult skin, SOX9 is normally associated with maintaining the identity of adult hair follicle stem cells. It’s normally suppressed in adult epidermal stem cells. But that’s not the case when it comes to basal cell and squamous cell carcinomas.
“In the disease context, SOX9 gets reactivated in adult epidermal stem cells,” says Yihao Yang, first author of the study.
How this process might unfold step by step has been unknown, Yang says. “Reprogramming in vitro happens really fast—within 48 hours. With such a short time window, it’s hard to get a good resolution on the sequence of events.”
The SOX9 Swap
To find out, the researchers engineered mice that contained a copy of SOX9 that could be activated in their adult epidermal stem cells when the mice were fed doxycycline, a drug that induced the transgenic SOX9.
“In adult tissues, choices that were easily made in embryogenesis are tightly suppressed so that adult stem cells stick to their dedicated task,” explains Fuchs.
Unleashing SOX9, however, turned out to be a potent influencer, progressively reprogramming the epidermal stem cells to new fates. “By only expressing this single SOX9 transcription factor,” Yang says, “we were able to induce basal cell carcinoma-like structures by week six. By week 12, we started to see lesions that resembled human basal cell carcinoma.”
Simultaneously, they tracked the epigenetic process going on behind the scenes. In the first two weeks, SOX9 turned off the epidermal stem cell genes. Reversing their normal state, they began to turn on hair follicle stem cell genes.
Seeking the mechanism, the researchers discovered that to achieve this fate switch, SOX9 hijacked the nuclear machinery from the active epidermal genes and brought this stolen equipment to the silent hair follicle genes. It then enlisted other transcription factors to pry open the closed chromatin bind to the silent genes within, turning them on.
“When SOX9 could not be regulated, the stem cells failed to make hair but instead just kept proliferating and activating several new transcription factors, eventually leading to a basal cell carcinoma state,” Fuchs says.
This complicated, identity-shifting back-and-forth was only possible because SOX9 is a pioneer factor, Yang says. “Only a pioneer factor has the ability to access closed chromatin,” he points out.
Because SOX9 is overly active in many of the deadliest cancers worldwide, the researchers aim to look for ways to intervene in its role in proliferating these cells. “By identifying how SOX9’s interacting proteins and its target genes change during malignancy, we hope to make inroads into unearthing new drug targets for these cancers,” Fuchs says.
Reference: “The pioneer factor SOX9 competes for epigenetic factors to switch stem cell fates” by Yihao Yang, Nicholas Gomez, Nicole Infarinato, Rene C. Adam, Megan Sribour, Inwha Baek, Mélanie Laurin and Elaine Fuchs, 24 July 2023, Nature Cell Biology.
DOI: 10.1038/s41556-023-01184-y
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.