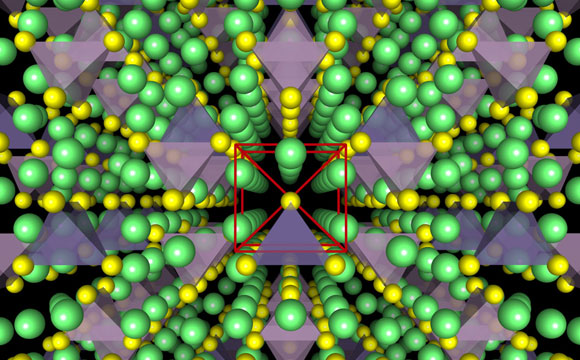
Illustrations show the crystal structure of a superionic conductor. The backbone of the material is a cubic-like arrangement of sulphur anions. Lithium atoms are depicted in green, sulfur atoms in yellow, PS4 tetrahedra in purple, and GeS4 tetrahedra in blue. Researchers have revealed the fundamental relationship between anion packing and ionic transport in fast lithium-conducting materials. Credit: Yan Wang
Engineers from MIT and Samsung have developed an approach for a solid electrolyte that could greatly improve both battery lifetime and safety, while providing a significant boost in the amount of power stored in a given space.
If you pry open one of today’s ubiquitous high-tech devices — whether a cellphone, a laptop, or an electric car — you’ll find that batteries take up most of the space inside. Indeed, the recent evolution of batteries has made it possible to pack ample power in small places.
But people still always want their devices to last even longer, or go further on a charge, so researchers work night and day to boost the power a given size battery can hold. Rare, but widely publicized, incidents of overheating or combustion in lithium-ion batteries have also highlighted the importance of safety in battery technology.
Now researchers at MIT and Samsung, and in California and Maryland, have developed a new approach to one of the three basic components of batteries, the electrolyte. The new findings are based on the idea that a solid electrolyte, rather than the liquid used in today’s most common rechargeables, could greatly improve both device lifetime and safety — while providing a significant boost in the amount of power stored in a given space.
The results are reported in the journal Nature Materials in a paper by MIT postdoc Yan Wang, visiting professor of materials science and engineering Gerbrand Ceder, and five others. They describe a new approach to the development of solid-state electrolytes that could simultaneously address the greatest challenges associated with improving lithium-ion batteries, the technology now used in everything from cellphones to electric cars.
The electrolyte in such batteries — typically a liquid organic solvent whose function is to transport charged particles from one of a battery’s two electrodes to the other during charging and discharging — has been responsible for the overheating and fires that, for example, resulted in a temporary grounding of all of Boeing’s 787 Dreamliner jets, Ceder explains. Others have attempted to find a solid replacement for the liquid electrolyte, but this group is the first to show that this can be done in a formulation that fully meets the needs of battery applications.
Solid-state electrolytes could be “a real game-changer,” Ceder says, creating “almost a perfect battery, solving most of the remaining issues” in battery lifetime, safety, and cost.
Costs have already been coming down steadily, he says. But as for safety, replacing the electrolyte would be the key, Ceder adds: “All of the fires you’ve seen, with Boeing, Tesla, and others, they are all electrolyte fires. The lithium itself is not flammable in the state it’s in these batteries. [With a solid electrolyte] there’s no safety problem — you could throw it against the wall, drive a nail through it — there’s nothing there to burn.”
The proposed solid electrolyte also holds other advantages, he says: “With a solid-state electrolyte, there’s virtually no degradation reactions left” — meaning such batteries could last through “hundreds of thousands of cycles.”
The key to making this feasible, Ceder says, was finding solid materials that could conduct ions fast enough to be useful in a battery.
“There was a view that solids cannot conduct fast enough,” he says. “That paradigm has been overthrown.”
The research team was able to analyze the factors that make for efficient ion conduction in solids, and home in on compounds that showed the right characteristics. The initial findings focused on a class of materials known as superionic lithium-ion conductors, which are compounds of lithium, germanium, phosphorus, and sulfur, but the principles derived from this research could lead to even more effective materials, the team says.
The research that led to a workable solid-state electrolyte was part of an ongoing partnership with the Korean electronics company Samsung, through the Samsung Advanced Institute of Technology in Cambridge, Massachusetts, Ceder says. That alliance also has led to important advances in the use of quantum-dot materials to create highly efficient solar cells and sodium batteries, he adds.
This solid-state electrolyte has other, unexpected side benefits: While conventional lithium-ion batteries do not perform well in extreme cold, and need to be preheated at temperatures below roughly minus 20 degrees Fahrenheit, the solid-electrolyte versions can still function at those frigid temperatures, Ceder says.
The solid-state electrolyte also allows for greater power density — the amount of power that can be stored in a given amount of space. Such batteries provide a 20 to 30 percent improvement in power density — with a corresponding increase in how long a battery of a given size could power a phone, a computer, or a car.
The team also included MIT graduate student William Richards and postdoc Jae Chul Kim; Shyue Ping Ong at the University of California at San Diego; Yifei Mo at the University of Maryland; and Lincoln Miara at Samsung. The work is part of an alliance between MIT and the Samsung Advanced Institute of Technology focusing on the development of materials for clean energy.
Reference: “Design principles for solid-state lithium superionic conductors” by Yan Wang, William Davidson Richards, Shyue Ping Ong, Lincoln J. Miara, Jae Chul Kim, Yifei Mo and Gerbrand Ceder, 17 August 2015, Nature Materials.
DOI: 10.1038/nmat4369









I would like one of the old flip phones that you carried in a holster on your hip, with the newer batteries. Recharging once a month with plenty of power when in remote locations would be great. That pull up antenna has got to have more gain than the ones embedded in todays phones. If not, I am sure that could be remedied quickly. Just a thought.
I still have a flip phone, and I carry it in a holster type container that hooks on my belt. No pull up antenna, though. Seriously, I think for actually making and receiving voice calls, a flip phone with hard key buttons is far superior to any so-called smartphone with a touch screen. A lot more rugged and less expensive, too. More battery life? – Well, it wouldn’t hurt.
All these fantastic ground-breaking revolutionary battery technologies I have read about over the last couple of years and I haven’t seen a SINGLE ONE come to market.
In 2006 Mit stated it had a battery that would fully charge in minutes .
We never saw it … 2009 Northwestern University also claims a battery recharge in minutes … Never saw that one as well … Why … Simple … Quick recharging battery technology would relate to electric vehicles thus making them the choice over gasoline , and then bye bye big oil … That can’t happen so those batteries will never see the light of day .
….Or a single one ever mentioned again twice. We’ll never see it again. 2) solid electrolyte is exactly what you don’t want if you want to do away with charging. Exactly what they were trying to get rid of with nano flow cell. They have liquid electrolyte that can be swapped in minutes so you don’t have to wait for charging. Oh so now we’re going too go back to solid unswapable electrolyte that we have to wait for it to charge. And that’s ‘breakthrough’. Duh! I think it’s a cover up. A cover up of the nano flow cell instantly swappable electrolyte. google Quantino
Didn’t you hear? The battery companies bought the patents and killed them.
Well, joking aside, they all seem like incremental improvements, not really ground-breaking innovations.
I work for an advanced materials company that supplies the battery industry. We are pushing every day to improve battery life, and batteries as a whole. No one is buying patents to suppress battery advancements in battery tech, haha.
The salt water electrolyte batteries that support 100% depth of discharge (lithium can’t) with 3000-5000 discharge cycles ARE on the market. And they cost less/kWh than the Tesla home batteries. Aquion Energy sells them.
You can say “over the last 30+ years” and it’s still true. Maybe I’m getting old and cynical, but I read this stuff with only passing interest now.
So when will they be available, who will build them, how much will the cost, is the electrolyte expensive. Is the material for the electrolyte expensive. Why didn’t this article cover these topics?
I’ll bet Energizer, Rayovac, et al., make this disappear, like the Big Three did to the Tucker automobile. This could be an industry wrecker.
Like congress trying to get the printer companies charging more for their ink than Chanel does for it’s top perfume. That faded away without a whimper…
all these so called “path-breaking” research in batteries with fancy names like MIT and Stanford etc. involving few Asian scientists seem to be a lot of bs and hogwash that pops up every day….battery technology real breakthrough isn’t something that has happened and god knows when it will…as said above just some incremental advancement, fact of the matter is scientists have no clue how to make things work in a practical sense and their main intent is publications..