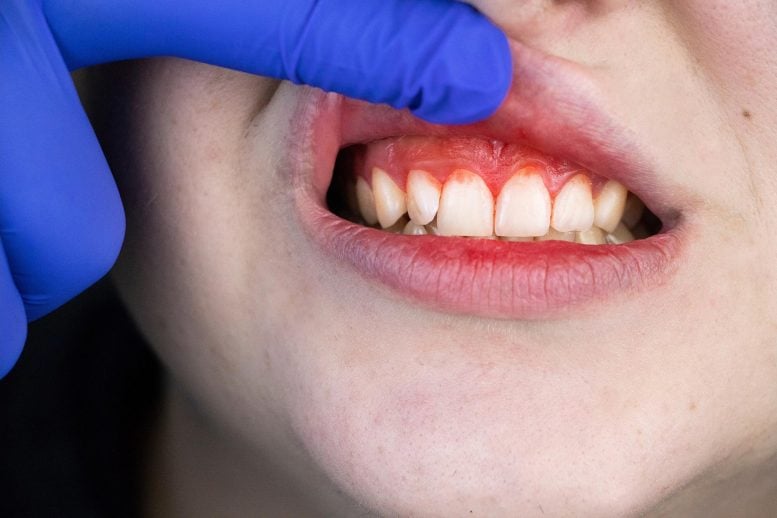
Researchers at Tufts University School of Dental Medicine believe that targeting the bacteria that cause periodontal disease could slow the progression of Alzheimer’s.
The bacteria Fusobacterium nucleatum (F. nucleatum) often flourishes in periodontal disease. It has an influence on the jawbone and gums, and if left untreated, can lead to unstable teeth and tooth loss. F. nucleatum has been connected in recent years to a variety of disorders, including premature birth and colorectal cancer.
Now, a new study from Tufts University researchers and associates reveals a connection between F. nucleatum and Alzheimer’s disease. Their findings were published in the journal Frontiers in Aging Neuroscience.
“In this study, our lab is the first to find that Fusobacterium nucleatum can generate systemic inflammation and even infiltrate nervous system tissues and exacerbate the signs and symptoms of Alzheimer’s disease,” says Jake Jinkun Chen, professor of periodontology and director of the Division of Oral Biology at Tufts University School of Dental Medicine. The first author of the paper is Hongle Wu, who was a postdoctoral fellow in the Chen Lab at the time of the study.
According to Chen, a professor in the Department of Developmental, Molecular and Chemical Biology at the School of Medicine and Graduate School of Biomedical Sciences and a trained pathologist, F. nucleatum can also cause severe generalized inflammation, which is a symptom of many chronic diseases such as Type 2 diabetes and Alzheimer’s disease.
By targeting F. nucleatum, Chen and his colleagues hope to slow the spread and progression of at least two epidemics: periodontal disease, which affects 47% of American adults over the age of 30, and Alzheimer’s disease, which currently affects 6.5 million Americans but is expected to reach over 14 million by 2060.
F. nucleatum and Immune Cells in the Brain
The latest research, done in mice, shows that F. nucleatum results in abnormal proliferation of microglial cells, which are immune cells in the brain that normally remove damaged neurons and infections and help maintain the overall health of the central nervous system. This over-supply of microglial cells also created an increased inflammatory response, the researchers found. Chronic inflammation or infection is believed to be a key determinant in the cognitive decline that occurs as Alzheimer’s disease progresses.
“Our studies show that F. nucleatum can reduce the memory and thinking skills in mice through certain signal pathways. This is a warning sign to researchers and clinicians alike,” Chen says.
Possible links between periodontal disease and Alzheimer’s have been posited by scientists in the past. While the new research does not show that F. nucleatum-related periodontal disease leads directly to Alzheimer’s disease, the new study suggests that periodontal disease caused by F. nucleatum and left untreated or poorly treated could exacerbate symptoms of Alzheimer’s disease, Chen believes. Conversely, treating periodontal disease effectively in those who have early-stage Alzheimer’s could potentially slow Alzheimer’s progression.
“Testing for bacterial load and degree of symptoms could one day become a way to measure the effects of F. nucleatum and manage treatment to slow the progression of both periodontal disease and Alzheimer’s,” Chen says.
Their research also suggests potential drug targets that could specifically quench the local as well as systemic inflammation caused by F. nucleatum in a periodontal environment.
Oral Infections and Systemic Diseases
More broadly, Chen and colleagues are targeting their translational research at blocking the pathways between periodontal disease and not only Alzheimer’s, but other diseases linked to inflammation, including Type 2 diabetes.
Only 2% of mRNA is translated into proteins. Ninety-eight percent of RNA is “noncoding” and traditionally has been considered as no more than “junk genes.” But increasingly scientists, including Chen, are uncovering important functions they possess.
In particular, his lab is focused on two noncoding RNAs. One—microRNA—regulates the production of proteins in cells. Another—lncRNA—performs other functions to regulate gene expression and could ultimately be used to treat atherosclerosis (hardening of the arteries) as well as periodontal disease, diabetes, cancer, and diabetic bone disease.
Studies by Chen’s lab have shown that a molecule called microRNA-335-5P can inhibit damage done by periodontal pathogens. The molecule could also have a robust effect in targeting the pathological molecules produced in the brain that lead to Alzheimer’s.
“MicroRNA in general suppresses gene expression and could stop the production of certain proteins. MicroRNA-335-5P, in particular, could target three ‘bad’ genes—DKK1, TLR-4, and PSEN-1—all believed to be related to Alzheimer’s disease,” he says.
His lab has also designed a small molecule called adipoAI, which has strong anti-inflammatory properties. Chen hopes to begin a clinical trial soon to study whether it is effective in treating a range of inflammatory diseases including Type 2 diabetes, Alzheimer’s disease, and periodontal disease.
“Your mouth truly is the gateway to your body,” he concludes.
Reference: “The Periodontal Pathogen Fusobacterium nucleatum Exacerbates Alzheimer’s Pathogenesis via Specific Pathways” by Hongle Wu, Wei Qiu, Xiaofang Zhu, Xiangfen Li, Zhongcong Xie, Isabel Carreras, Alpaslan Dedeoglu, Thomas Van Dyke, Yiping W. Han, Nadeem Karimbux, Qisheng Tu, Lei Cheng and Jake Chen, 23 June 2022, Frontiers in Aging Neuroscience.
DOI: 10.3389/fnagi.2022.912709
The study was funded by the National Institutes of Health.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
8 Comments
Is it just me, or has there been an unusually large amount of Alzheimer’s articles of late?
I agreed with you, Sam.
So your saying gum disease will cause all this really what if people cant get dental cleanings ect no INS r money to do treatments on teeth gums? dental INS out ragouios as the work done on teeth..
Tons to f Alzheimer’s articals lately because so many of use are having signs of it at an early age. I’m 30 and am needing to see a Dr about it I have a tbi and feel as if I have early signs of it
I think you published an article about relationship
between gum problems and dementia. How can I get a
copy of this article?
I would like to get a copy of an article you recently
published on the relationship of gum problems and
dementia.
How can I get a copy of an article which you
recently published about the link between gum
problems and dementia?
How can I obtain a copy of an article which you
recently published about the link between gum
problems and dementia?