A blue-light-powered iron catalyst just replaced rare metals — and unlocked a milestone in precision drug synthesis.
Photocatalysts are materials that trigger chemical reactions when exposed to light. In modern organic chemistry, metal-based photocatalysts are especially valuable because they are stable and can be fine-tuned by adjusting the ligands attached to the central metal atom. These ligands influence how the catalyst behaves and what kinds of molecules it can help build.
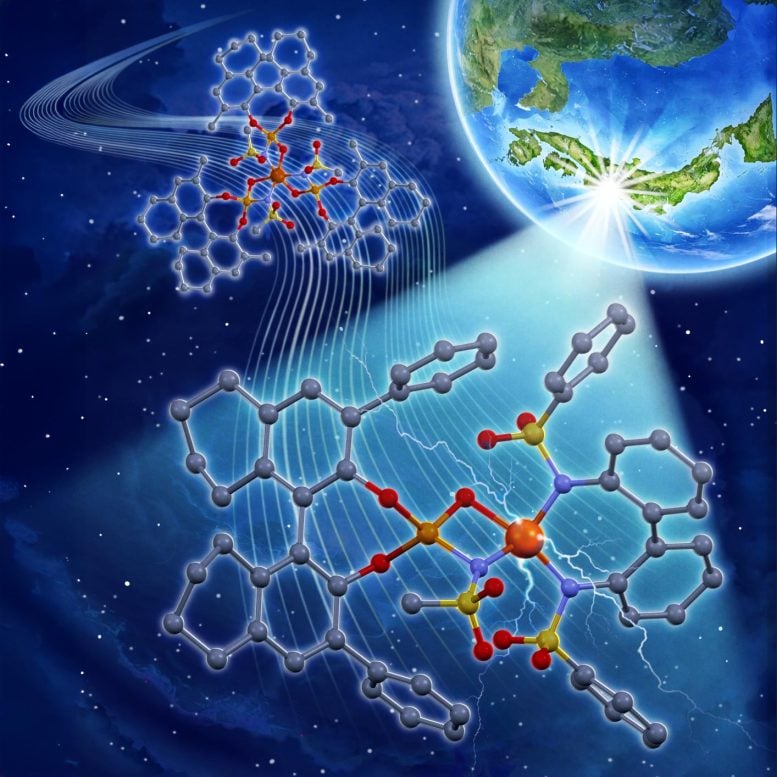
Common photocatalyst metals such as ruthenium and iridium work well but are rare and costly. To address this, researchers at Nagoya University in Japan previously introduced an iron-based alternative. However, that earlier system depended on large quantities of expensive chiral ligands, which act as structural guides that determine the three-dimensional shape of the final chemical product.
In a new study published in the Journal of the American Chemical Society, the team reports a redesigned iron catalyst that cuts chiral ligand use by two-thirds. The catalyst also operates under energy-efficient blue LED light, making the process more practical and potentially more sustainable.
Using this improved system, the scientists completed the asymmetric total synthesis of (+)-heitziamide A. This natural compound, found in medicinal plants, is known to suppress respiratory bursts. The work was carried out by Professor Kazuaki Ishihara, Assistant Professor Shuhei Ohmura, and graduate student Hayato Akao at Nagoya University’s Graduate School of Engineering.
Iron Photocatalyst Design Improves Efficiency
In their 2023 work, the group developed an iron photocatalyst that incorporated three chiral ligands per iron atom. Yet only one of those ligands actually influenced enantioselectivity, meaning much of the material was not contributing to the desired three-dimensional control. That made the system less efficient than it could be.
The newly engineered catalyst takes a different approach. It pairs an inexpensive achiral bidentate ligand with a chiral ligand to form a specific iron(III) salt structure. The chiral ligand directs the three-dimensional arrangement of the product, while the achiral bidentate ligand adjusts and enhances the catalyst’s overall activity.
With this design, the team achieved a highly controlled radical cation (4 + 2) cyclization. In this reaction, two molecular components join to create a six-membered ring. The method allows chemists to construct 1,2,3,5-substituted adducts, structural patterns frequently seen in natural products such as heitziamide A.
“The new catalyst design represents the definitive form of chiral iron(III) photoredox catalysts,” stated Ohmura, one of the study’s corresponding authors. “We believe this achievement marks a significant milestone in advancing iron-based photocatalysis.”
First Asymmetric Synthesis of (+)-Heitziamide A
Although scientists have previously reported laboratory synthesis of heitziamide A, they had not achieved the total asymmetric synthesis of its naturally occurring enantiomer.
By using blue light to activate the iron photocatalyst and carefully controlling six membered ring formation, the researchers successfully completed the first total asymmetric synthesis of (+)-heitziamide A. The results suggest that employing the mirror image version of the catalyst could also produce (-)-heitziamide A, making it possible to selectively generate either enantiomer.
Implications for Drug Synthesis and Green Chemistry
This new iron photocatalyst provides a way to build complex molecules, including pharmaceutical precursors, using abundant iron and low energy blue LEDs instead of scarce rare metals.
“Achieving the first-ever asymmetric total synthesis of (+)-heitziamide A using this catalytic reaction is a remarkable accomplishment,” stated Ishihara, the study’s other corresponding author. “Several additional bioactive substances can be accessed through total synthesis, with enantioselective radical cation (4 + 2) cycloaddition serving as a key step. We intend to publish follow-up papers on the asymmetric total synthesis of these compounds in the near future.”
Reference: “A Rational Design of Chiral Iron(III) Complexes for Photocatalytic Asymmetric Radical Cation (4 + 2) Cycloadditions and the Total Synthesis of (+)-Heitziamide A” by Hayato Akao, Shuhei Ohmura and Kazuaki Ishihara, 8 January 2026, Journal of the American Chemical Society.
DOI: 10.1021/jacs.5c20243
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
I understand the premise but I would think cryosynthesis would make more sense because it can be modified as the said therom could potentially get most bacteria to grow. if you could use the cryosynthesis to modify the said light structure it could be studied and form said antibacteria or be used for both, growing bacteria for ailments and a manor which breaks down and hybrid or adapted cell. should be modifyable