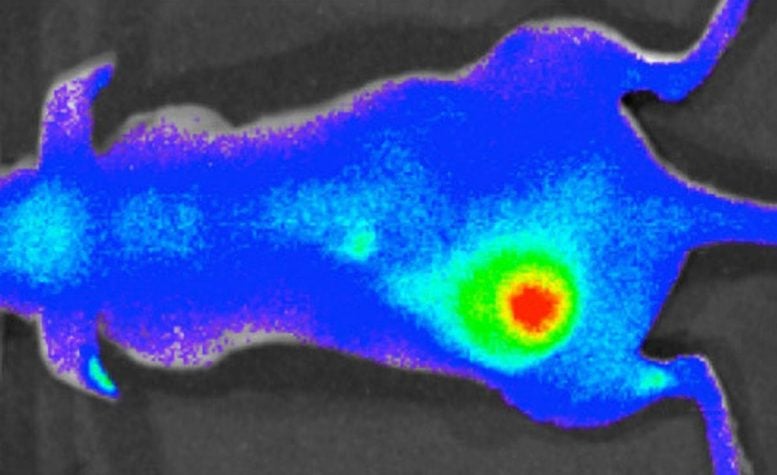
A team of researchers at Yale University has developed a therapeutic molecule that both targets the acidic microenvironments of tumors and penetrates cells to deliver a therapeutic cargo.
MicroRNAs may be tiny — as few as 20 genetic letters, compared to 3 billion in the DNA of a human — but they play a major role in biology, helping to determine which genes are expressed or silenced. In the last 10 years, researchers at Yale and elsewhere have shown they play a major role in formation and spread of tumors.
However, their potential as a target for cancer therapy has not been realized because of a daunting problem that has held back clinical applications of gene therapy: How can you target minute pieces of genetic material locked safely inside the membranes of billions of cells?
Now a multi-disciplinary team of Yale researchers has solved the problem by designing a therapeutic molecule that both targets the acidic microenvironments of tumors and penetrates cells to deliver a therapeutic cargo. The new delivery system effectively killed advanced tumors in mice, the team reports in the November 17 issue of the journal Nature.
“This strategy opens up a new pathway to therapy, not just for the treatment of cancer but for a host of other diseases as well,” said Donald Engelman, a co-author of the paper and the Eugene Higgins Professor of Molecular Biophysics and Biochemistry at Yale.
Reference: “MicroRNA silencing for cancer therapy targeted to the tumour microenvironment” by Christopher J. Cheng, Raman Bahal, Imran A. Babar, Zachary Pincus, Francisco Barrera, Connie Liu, Alexander Svoronos, Demetrios T. Braddock, Peter M. Glazer, Donald M. Engelman, W. Mark Saltzman and Frank J. Slack, 17 November 2014, Nature.
DOI: 10.1038/nature13905
The findings were the result of an extensive collaboration between labs of Engelman, Peter Glazer, Mark Saltzman and Frank Slack.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Hi very interested in your research 😊 thank you 😊
Why on earth would you use a photo of a dog?