New research from Yale University shows that a drug that blocks or inhibits the AIM2 pathway could potentially limit the deleterious side effect of chemotherapy or radiotherapy on cancer patients.
High doses of radiation from cancer treatment can cause severe damage to cells and tissues, resulting in injury to bone marrow and the gastrointestinal tract. The consequences can be fatal. Yet researchers do not fully understand how exposure to radiation triggers this damage at the molecular level.
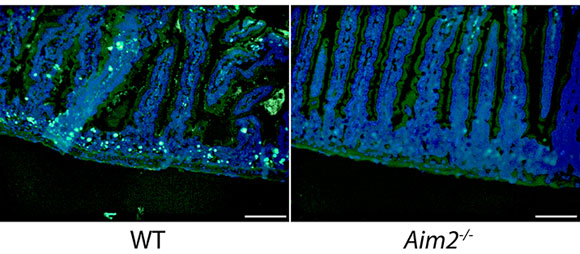
Led by Yale professor of immunobiology Richard Flavell, an international team of researchers studied the radiation response using animal models. They identified a novel mechanism of radiation-induced tissue injury involving a protein called AIM2, which can sense double-strand DNA damage and mediate a special form of cell death known as pyroptosis.
They observed that in animals lacking AIM2, both the gastrointestinal tract and bone marrow were protected from radiation. While the role of AIM2 as a sensor that detects infectious threats to the body was known, this study is the first to describe the protein’s function in the detection of radiation damage to the chromosomes in the nucleus, said the researchers.
When a cell receives a high dose of radiation, the DNA is broken into pieces, which can be joined together again. However, this aberrant rejoining of chromosomal fragments can lead to chromosomal abnormalities and cancer. Flavell and his team believe that when this chromosomal damage is inflicted, the AIM2 pathway is activated in order to kill the cell to avoid the deleterious consequences of these chromosomal translocations, such as those commonly seen in cancer cells.
For this reason, the cells that accumulate this chromosomal damage are dangerous to the person or animal and are therefore killed by this AIM2 pathway. This pathway is beneficial to the person or animal under normal circumstances because it eliminates dangerous cells, but when a high dose of radiation is given the pathway is detrimental because it leads to bone marrow and digestive tract injury.
These findings suggest that a drug that blocks or inhibits the AIM2 pathway could potentially limit the deleterious side effect of chemotherapy or radiotherapy on cancer patients, said the researchers.
Reference: “The DNA-sensing AIM2 inflammasome controls radiation-induced cell death and tissue injury” by Bo Hu, Chengcheng Jin, Hua-Bing Li, Jiyu Tong, Xinshou Ouyang, Naniye Malli Cetinbas, Shu Zhu, Till Strowig, Fred C. Lam, Chen Zhao, Jorge Henao-Mejia, Omer Yilmaz, Katherine A. Fitzgerald, Stephanie C. Eisenbarth, Eran Elinav and Richard A. Flavell, 11 November 2016, Science.
DOI: 10.1126/science.aaf7532
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.