
Two experimental drug molecules promoted myelin repair in MS disease models, pointing toward a possible future route for treating nerve damage rather than only suppressing inflammation.
Multiple sclerosis (MS) is one of the leading causes of neurological disability in young adults, affecting nearly three million people worldwide. Despite decades of research and major advances in slowing the disease, scientists have yet to develop a treatment capable of repairing the damaged nerve tissue left behind.
Now, a new doctoral thesis suggests that may finally be changing: researchers have identified two separate drug molecules that successfully triggered the regrowth of myelin, the protective coating that allows nerve cells to communicate properly.
MS is most common in Northern Europe and Canada, with rates increasing toward the northernmost latitudes. The disease occurs when the immune system mistakenly attacks myelin, gradually disrupting nerve signaling throughout the brain and spinal cord. As damage accumulates, patients can experience symptoms ranging from blurred vision and chronic fatigue to mobility problems and long-term neurological disability.
No treatment for neural damage
Today’s MS medications are designed to reduce excessive immune activity, but they do not fix the nerve damage that has already occurred. This limitation is especially serious in progressive MS, where injury builds gradually over many years.
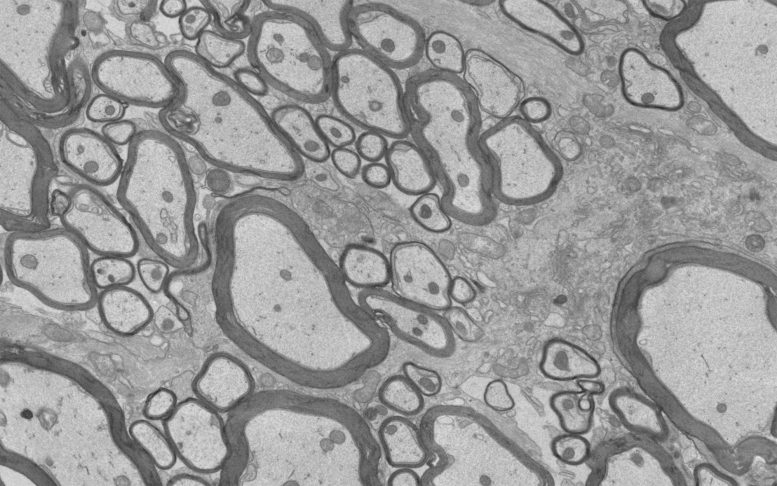
For decades, researchers have searched for ways to restart remyelination, the repair process in which damaged myelin grows back and neurons regain protection. So far, every drug candidate tested for this purpose has failed. A major obstacle is that, in later stages of MS in particular, the central nervous system develops local tissue conditions that block myelin repair.
Two solutions with the same outcome
In his doctoral thesis, Tapani Koppinen from Associate Professor Merja Voutilainen’s research group identified two separate ways to improve remyelination.
The first strategy uses a drug molecule to act on a stress response inside brain cells. In MS damaged areas, this response remains constantly overactive, which stops repair-promoting cells from carrying out their role. After this mechanism was blocked with the new drug molecule, remyelination increased significantly and happened faster in brain tissue showing MS-like damage. The study was published in the Molecular Therapy journal in February.
The second strategy targets scar tissue that develops around damaged areas and creates a physical obstacle to nerve repair. By changing the makeup of this scar tissue with another drug molecule, this approach also helped promote neuronal recovery. An article on this strategy was published in the Neuropharmacology journal in November 2025.
Although the two drugs work through completely different mechanisms, they produced strikingly similar outcomes: strong remyelination and lower neuroinflammation in disease models, that is, animal and cell tests modeling the tissue pathology of MS.
First drug that boosts remyelination requires further research
At this stage, the findings come from laboratory animals and cell models. Because human MS involves more complex tissue conditions, the drug molecules must still be tested for effectiveness in people. Brain-targeted drugs also face the challenge of the blood-brain barrier, which prevents many substances from entering the brain. Even so, the researchers showed that both molecules successfully reached the central nervous system in laboratory animals.
“The goal is to enable the molecules we have developed to reach clinical trials, which could one day produce the first drugs that enhance remyelination in MS. In the meantime, our findings can help in investigating the pathogenic mechanisms of MS that inhibit remyelination,” Koppinen says.
References:
“Modulation of the unfolded protein response with a C-terminal fragment of MANF facilitates recovery in models of multiple sclerosis” by Tapani K. Koppinen, Carolina R. Reyes, Jinhan Nam, Aastha Singh, Shibajee Mandal, Liam Beckett, Alba Montedeoca, Tuomas A.E. Kallionpää, Maria Lindahl, Francisco J. Rivera and Merja H. Voutilainen, 11 October 2025, Molecular Therapy.
DOI: 10.1016/j.ymthe.2025.10.023
“Modulation of the unfolded protein response with a C-terminal fragment of MANF facilitates recovery in models of multiple sclerosis” by Tapani K. Koppinen, Carolina R. Reyes, Jinhan Nam, Aastha Singh, Shibajee Mandal, Liam Beckett, Alba Montedeoca, Tuomas A.E. Kallionpää, Maria Lindahl, Francisco J. Rivera and Merja H. Voutilainen, 11 October 2025, Molecular Therapy.
DOI: 10.1016/j.ymthe.2025.10.023
Disclosure: Tapani Koppinen, MSc (Pharmacy), will defend his doctoral thesis entitled ‘Enhancing remyelination by overcoming extrinsic and intrinsic inhibitory factors’ on 8 May 2026 at 13.00 at the Faculty of Pharmacy, University of Helsinki., Finland The public defence will take place at Biocenter 2, room 2041, Viikinkaari 5.
Professor Robin Franklin, FRS, from the University of Cambridge will serve as the opponent and Associate Professor Merja Voutilainen, the thesis supervisor, as the custos.
The thesis is also available in electronic form through the Helda repository.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
21 Comments
thanks
THE NEW CURE FOR “MULTIPLE-SCELEROISIS” IS ACCUMLATED (150% NATURAL-ABSTARCTION) AN ADHEVISE OF COLLECTIONS INTO THE NEXT GENEARTIONS OF LIQUID (ANTIBOTICS BEYOND INCREMETNTS ARE VALID INTO INCREASEDLEVELS BOTH RED-BLOOD CELL (DNA RE-DEFINES & WHITE CELLS (IMMUNITY-SCREEN) (MATHAEMATCIAL-QUOTIONS (REALITY OF CLIENTS IN THE PROCESS EFFECTIVE BY AMOUNTS OF RATIO(S) THESE CELLS ARE INTO THESE STATES OF RESTATED FORMS A NEW PROVEN (BIO-GENETICS)(BIO-ANATOMY)(BIO-PHYISOLOGY) THE 50% OF PROCESS OF THESE COLLECTIONS ARE DEVELOPED BY (BIO-STATISICS) ACCELLERATED-VALUES OF TEHN NEW THREE MINERALS FOUNDED “BANANAS-EXTRACT”FROZEN/DEHEATED AT ROOM CAPACITY (TIME-FRAMES OF 15-20MINUTES STATES A SERIES OF COLLECTIONS (Z-G-E CELLS)DEICISTS A WONDERFUL ISSUES OF CURES COMBINATIONS IN ALOE VERA REDUCES THE VALUES OF NEW PERCENTAGE (EXCEEDS: 100%) FOR GENERATIONS ELIMINATIONS OF THE BODY TO BECOME REPAIRED NOT REPLACED:THE COMBINATIONS REPLACES (VITAMINS BEING INTERWINED WITH MINERALS &CHEMSITRY”SOME” ‘(POINT)’REPLACES A SIMPLE FACTORS AREAS OF DESCRIPITONS ARE DECREASED BY EVERY-TYPES OF CURES ARE IST OWN INFINTY TO COLLECTIONS OF MEASUREMENTS OF ALL SERIES OFTEN )”REDUCED-FUNDAMNENTALS ARE COMBINATIONS COMPLETED-PROCESS OF REALTIY THE 100% IS BEYOND PER-INDIVISUALS AND ENERGY IS COMBINATIONS “MIGHTY”ARE COMMON-ISSUES OF REASONING DUE TO THESE QUESTIONS SOURCES ARE REALITY NEVER-THE-LESS & ASSUMPTIONS INTO THESE QUESTIONS INNER-CODES ARE INTO THESE QUESTIONS MEASURES ARE COLLECTIONS OF DOUBTS ARE DESCRIPITONS MASSIVELY ARE THE FUTURE (BIO-MEDICINE)(BIO-PSYCHOLOLGY)(BIO-SURREGRY AND MASSIVE DESCREASED AND DEPENDS INTO THESE QUESTIONS ARE COMMON-ISSUES ACCUMLATES INTO THESE COLLECTIONS REDUCED BY EACH COLLECTIONS
THESE INNER-CORE ARE THE FOLLOWING-CIRCUMSTANCES OF (BIO-INFINITY)(BIO-REALITY)(BIO-SPACE) AND
(BIO-GENETICS) THE FEATURES A SIMPLE DESCRIPLTIOSN MUST BE CONNECTED BY EACH VARIABLES OF DOUBTS AND DESCRIPTIONS ENERGY-LEVELS ARE VALUES OF COMPLETES INTO THESE COMBINATIONS NOT DECREASED SERIES ARE COMMON-ISSUES ASSUMPTIONS AMOUNTS NOT MASSIVE DECLINED ERRROS IN ALL (BIO-EVOLTIONS AND FOR SIDE-EFFECTS (DIZZINESS/CONSIPATION/HEADACHES & DIAHRRIES.THESE QUESTIONS ARE VALUES OF DECRIPTIONS INTO THE REDUCTIONS OF ALL SIMPLE ISSUES:
I have Peripheral Neuropathy with Edema in my Toes & Anklets which is very Painfull developed after Long COVID Inflammation.
What is the best treatment to cure same.
How do I get this treatment for nerve damage repair
How can I get more information on this Please help me
Holy hell what a mess of unintelligible babble.
Can this concept be applied to injury of the myelin around the trigeminal nerve? I have trigeminal neuralgia from a root canal procedure. If this could work to repair the injuried myelin it would be wonderful. Has this been explored?
I have secondartbprogressive MS, please keep me posted.
I have ppms and am losing my ability to walk i would like you to consider trying this breakthrough on me
Can this be applied to other nerve disorders and diseases? If so which ones? Is there hope for future studies into different nerve issues?
I have to make a statement regarding the 2nd paragraph above. “Multiple sclerosis (MS) is one of the leading causes of neurological disability in young adults, affecting nearly three million people worldwide.”
MS is not a disease the just affects the young. I was diagnosed at age 55 with RRMS.
Me 2 at 50 with PPMS
I am now 80 years old and was diagnosed with MS when I reached my 50’s.Now I am dealing with sciatic nerve pain along with managing MS problems. Yes I am still getting around without a cane or wheelchair , the fatigue is unbelievable.
The two references at the end are duplicates.
Sounds like this would be great for Fiber Myalgia and Lupus as well.
Thanks to the researcher and his supervisor for this excellent research. I hope it can finish as a solution to cure MS.
Read the article and the studies provided at the bottom.
Does this have any relationship to myelin sheath damage for diabetic peripheral neuropathy?
Could this also possibly help with
Parkinson’s that would Great .
If you have any more information about this please let me know I have a dear friend that has MS and
my Aunt has Parkinson’s
Fat lot of good it does when you neglect to identify the remylineating molecules. Some of are DESPERATE and need relief NOW.
How soon will drugs be out to regenerate myelin for MS please answer my question